Research Article
Screening of Florida Native Green Microalgae as a Potential Source of Biodiesel
Priyanka Narendar1, Krishnaswamy Jayachandran1 and Miroslav Gantar2*
1Department of Earth and Environment, Florida International University, MM Campus, 11200 SW 8th St. Miami, Fl. 33199
2Department of Biological Sciences, Florida International University, MM Campus, 11200 SW 8th St. Miami, Fl. 33199
*Corresponding author: Miroslav Gantar, Department of Biological Sciences, Florida International University, MM Campus, 11200 SW 8th St. Miami, Fl. 33199, E-mail: gantarm@fiu.edu
Article Information: Submission: 23/04/2015; Accepted: 25/05/2015; Published: 28/05/2015
Copyright: © 2015 Miroslav Gantar, et al. This is an open access article distributed under the Creative Commons
Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the
original work is properly cited.
Abstract
Twenty seven strains of green algae from the Florida aquatic habitats were screened for their ability to accumulate lipids. In addition, Botryococcus
braunii was used as the reference strain. This preliminary screening for lipid content was performed with laboratory grown algal cultures using the fluorescent
dye Nile red. The lipid accumulation was followed during the course of cultivation as well as under conditions of nitrogen and phosphorus deprivation. Of
all strains tested, an increased lipid accumulation was found in five strains (Coelastrum sp. strain 46-4 and strain 108-5, Chlorococcum sp. strain64-12,
Dactylococcus sp. strain 64-10 and Stigeoclonium sp. strain 64-8) and they were used for further studies. A linear relationship between algal biomass yield
(culture age) and lipid accumulation was observed in most strains tested (Coelastrum sp. strain 46-4 and strain 108-5, Chlorococcum sp. strain 64-12,
and Stigeoclonium sp. strain 64-8). Significantly higher lipid accumulation under nitrogen deprivation was observed in only two strains while five strains
had elevated lipid accumulation under phosphorus deprivation. Apparently, nutrient deprivation conditions affect different strains differently. Three strains
contained more than 50% of lipid of dry biomass (Coelastrum 108-5, Chlorococcum 64-12 and Stigeoclonium 64-8) and therefore were identified as potential
organisms to be used in algae-based biodiesel technology.
Keywords
Biofuel; Green algae; Lipids; Nile red
Introduction
The need for renewable energy sources is increasing rapidly as
result of rising global population and industrialization. Diminishing
of fossil fuel reserves, and increasing concerns about energy security
and climate change have led to an increased interest in biofuel
research [1]. One form of biofuels, which is currently receiving much
attention, is biodiesel. Current sources of commercial biodiesel
include soybean and palm oil, animal fat and cooking oil waste [2].
The advantage of using biodiesel is that it is a renewable fuel, nontoxic
and biodegradable [3]. However, despite the benefits that
biodiesel commercialization can provide, the economic aspect of its
development at large-scale is limited by the high cost of vegetable oil
[4], resulting in a search for alternative sources of feedstock.
Microalgae are a promising alternative sources for biodiesel [5,6].
The algae-for-fuel concept started in the 1970s [7] and has recently
been re-invigorated with steady increases in energy prices [2]. Using algae for biofuels have several advantages over conventional crops
including high growth rates and oil production per area unit [8,9] and
they can be grown in regions of non-arable land, thereby avoiding
competition with food crops [10]. Algae also use less water than
the traditional oilseed crops and contribute to sequestration of the
greenhouse gas carbon-dioxide (CO2) [11,12].
Different kind of lipids, hydrocarbons and other oils are produced
by different algal species [13]. Some algae can accumulate up to 80%
of lipids by weight of dry biomass [14], however the suitability of
lipids for fuel conversion depends on species characteristics [1].
In order to develop a technology of producing biodiesel from
algae, a selection of a productive organism adapted to local conditions
is crucial. Since green algae are ubiquitous and easy to grow they
represent the major groups of algae within lipid-producing species
[2]. Florida is considered a suitable location for mass cultivation of
algae and therefore screening of native green algal strains for their
ability to accumulate lipids has great potential. In this work the cell lipid accumulation was determined in cultures of different strains of
green algae under varying culture conditions.
Materials and Methods
Organisms and culture conditions:
Twenty-seven green algal strains (Chlorophyta) that are part of
a culture collection at Florida International University were used in
the study (Table 1). Taxonomic identification of the strains to the
genus level was based on morphological characteristics [15]. As a
reference strain, we used Botryococcus braunii, which was purchased
from University of Texas, Austin. Algal cultures were grown in BG11
medium [16] which was buffered with MES buffer(Sigma-Aldrich) at
pH 7.20.The cultivation was carried out in laboratory conditions on a
shaker with agitation speed of 150 rpm under cool white light (30 μΕ
m-2 s-1) and temperature of 24°C.Growth rate:
The growth rate determination was based on dry weight and
chlorophyll measurements. For the dry weight measurements, five
milliliters (three replicas) of samples were filtered onto pre-weighed
GF/C (Whatman) filter papers. Filters were dried in an oven at 60°C
to a constant weight and dry weight determined. In the time course
experiment (Figure 1) the growth rate was determined using changes
in chlorophyll content. Five milliliters of culture were centrifuged and
the pellet was extracted with methanol and the absorbance measured
at 666nm and 653nm [17]. The samples were taken every five days.Screening for lipid content:
Screening the algal strains for lipid content was based on the Nile
red method [18,19]. Algae cultures were grown in 250 ml Erlenmeyer
flasks in BG11 medium on a shaker at 25 ºC under constant light
(30μΕm-2 s-1). Samples were taken on the 13th and 45th day of
cultivation. Sample of every culture was normalized by dilution to
an optical density (OD600) of 0.2. One milliliter of a sample (three
replicates) was transferred into eppendorf tube, homogenized by
vortexing, and then DMSO was added (final concentration 20%) to
improve penetration of the dye. To this cell suspension, the Nile red
solution (6.5 mg of Nile red in 26 ml acetone) [20] was added to final
concentration of 1% (v/v). This mixture was incubated in the dark for
10 min and transferred into 96-well plates. The plates were read by
using the excitation wavelength of 530 nm and emission wavelength
at 575 nm [21] on a plate reader Synergy 2 (BioTek, Winooski,
VT). The relative fluorescence intensity of Nile red in the stained cells was obtained after subtraction of both the autofluorescence of
algal cells (cells without Nile red) and the fluorescence of Nile red
alone in the medium [20]. These values were normalized by dividing
them by the absorbance values measured at a wavelength of 600nm.
In the case of the primary screening (Table 1) the intensity of Nile
red fluorescence was further translated into lipid content per 100
μl of the culture by using standard curve generated with Triolein as
a lipid standard (Fischer Scientific, USA).In other experiments the
Nile red fluorescence was translated into lipid amount calculated as a
percentage of the dry weight.Triolein standard curve was used in the determination of lipid
content. One milliliter of triolein which weighed 0.9194g was
dissolved in 9ml of chloroform and diluted to 10-4. The concentration
of the lipid was brought to 90 μg and further diluted in chloroform
in order to obtain a concentration in the range of 5 to 50 μg ml-1
with the total working standard volume of 3ml. To 3ml of solution,
300 ml of chloroform and 150 ml of methanol was added. Also, 7
μl of Nile red was added and boiled for 1- 1.5 min and allowed to
cool down to room temperature [22]. Fluorescence was read using
the plate reader at 530 nm and 575 nm respectively. The blank in this
case was chloroform, methanol and Nile red. The calibration curve
was constructed with the fluorescence intensity against the lipid
concentration.
The effect of nitrogen and phosphorus depletion:
Those strains showing the highest lipid content were cultured
under varying nitrogen and phosphorous concentrations to
provide information on how different strains respond to nitrogen
and phosphorus limitation. Three-week old cultures grown in
BG11 medium were centrifuged and the biomass washed with the
nitrogen-free or phosphorus-free medium. This biomass was used to
inoculate media containing 0%, 50% and 100% of a standard nitrogen
and phosphorus content in BG11 medium. The lipid content was
determined using the Nile red method for samples taken after 5 and
10 days of growth. Nile red fluorescence was translated into percent
lipid per unit cell dry weight.Statistical analysis:
All statistical analysis were conducted using SPSS 15.0 software.
In this study, it was hypothesized that the lipid content determined
by both gravimetric technique and Nile red method were the same.
In order to test our hypothesis, Independent sample T test was used by comparing the means. It was also hypothesized that nitrogen and phosphorus depletion would increase lipid levels in selected strains.
The significance of differences were tested using one way ANOVA.
Comparisons that show p-values< 0.05 were considered significant.
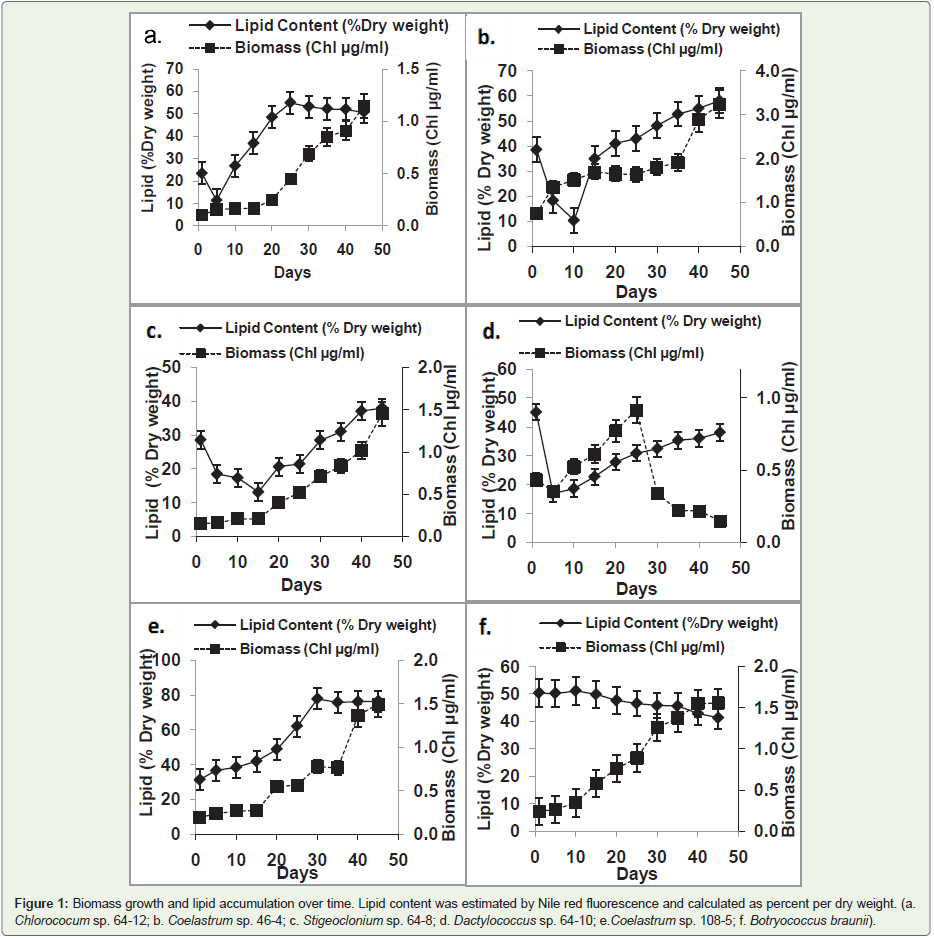
Figure 1: Biomass growth and lipid accumulation over time. Lipid content was estimated by Nile red fluorescence and calculated as percent per dry weight. (a.
Chlorococum sp. 64-12; b. Coelastrum sp. 46-4; c. Stigeoclonium sp. 64-8; d. Dactylococcus sp. 64-10; e. Coelastrum sp. 108-5; f. Botryococcus braunii).
Results
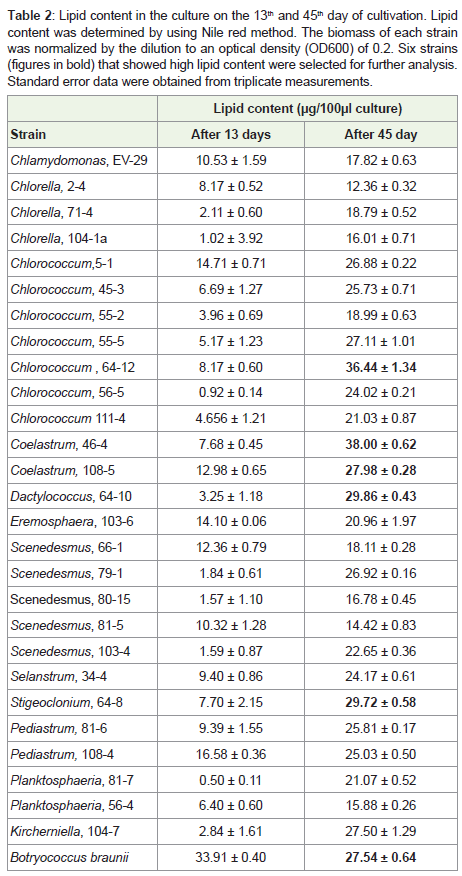
Screening the isolates by Nile red method:
The initial screening was performed with 27 algal strains. Lipid
content was estimated during the exponential phase of growth (day
13) and at the end of cultivation (day 45). During early exponential phase there was no substantial accumulation of lipidsin any of tested
strains except in reference strain Botryococcus braunii (Table 2). Lipid
content went up in stationary phase for all the strains. Strains that had
lipid content higher than 27μg per 100μl of culture,were considered
promising strains and used in further studies including Coelastrum sp.
46-4; Chlorococcum sp. 64-12, Dactylococcus sp. 64-10; Stigeoclonium sp. 64-8; and Coelastrum sp. 108-5, (Table 2). In a separate
experiment using the selected strains, lipid content was determined as
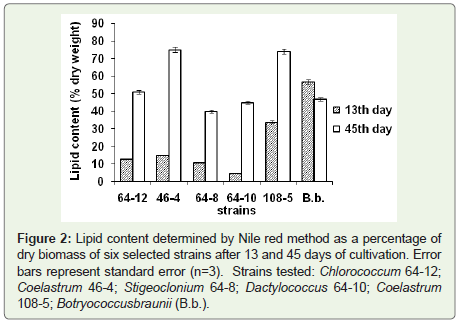
a percentage of cell dry weight (Figure 2). Coelastrum sp. strain 46-4
had the highest lipid content. After 45 days of cultivation, this strain contained 75% lipids, Coelastrum sp. strain 108-5 and Chlorococcum sp. strain 64-12, contained 73% and 51% of lipids respectively.Lipid accumulation depending on the culture age:
In order to determine effect of culture age on lipid accumulation,
lipid levelwasdetermined in the biomass of selected six algal strains
every five days over a period of 45 days. In most tested strains,
biomass continuously increased during the entire cultivation period
(Figure 2). The exception was Dactylococcus sp. strain 64-10 (Figure 2d) that reached the maximal biomass yield at day 25 followed by
a sharp biomass decline. The pattern of lipid accumulation differed
from one strain to another. In four stains (Chlorococcum sp. strain
64-12; Stigeoclonium sp. strain 64-8; Coelastrum sp. strain 46-4; Dactylococcus sp. strain 64-10) there was an initial evident decline in
lipid content. After that initial phase, several different patterns of lipid
accumulation were observed. There was (1) a continuous increase
of lipid content during the entire cultivation period (Coelastrum sp.
strain 46-4; Stigeoclonium sp. strain 64-8; Dactylococcus sp. strain 64-
10) (Figure 2b, 2c and 2d); (2) the lipid content reached a plateau after
25 days of cultivation (Chlorococcum sp. strain 64-12; Coelastrum sp.
strain 108-5) (Figure 2a andFigure 2e); (3) and in the case of Botryococcus
braunii, there was no initial change of lipid content with the slow
decline towards the end of cultivation period (Figure 2f).
Table 2: Lipid content in the culture on the 13th and 45th day of cultivation. Lipid content was determined by using Nile red method. The biomass of each strain was normalized by the dilution to an optical density (OD600) of 0.2. Six strains (figures in bold) that showed high lipid content were selected for further analysis. Standard error data were obtained from triplicate measurements.
A linear regression test showed that only in four strains
(Chlorococcum sp. strain 64-12, Coelastrum sp. strain 108-5,
Stigeoclonium sp. strain 64-8 and Coelastrum sp. strain46-4) there
was a linear relationship between algal biomass yield and lipid
accumulation with the following correlation coefficients (R2) of 0.834,
0.703, 0.632 and 0.612 respectively.
Effect of nitrogen concentration on lipid accumulation:
In order to assess effect of nitrogen depletion on lipidlevel, biomass
of a 30-day-old culture was washed with nitrogen-free medium and
transferred into nitrogen-free medium and medium containing
50% and 100% of nitrogen concentration normally found in BG11
medium. The lipid level of the inoculum (day 0) was determined and
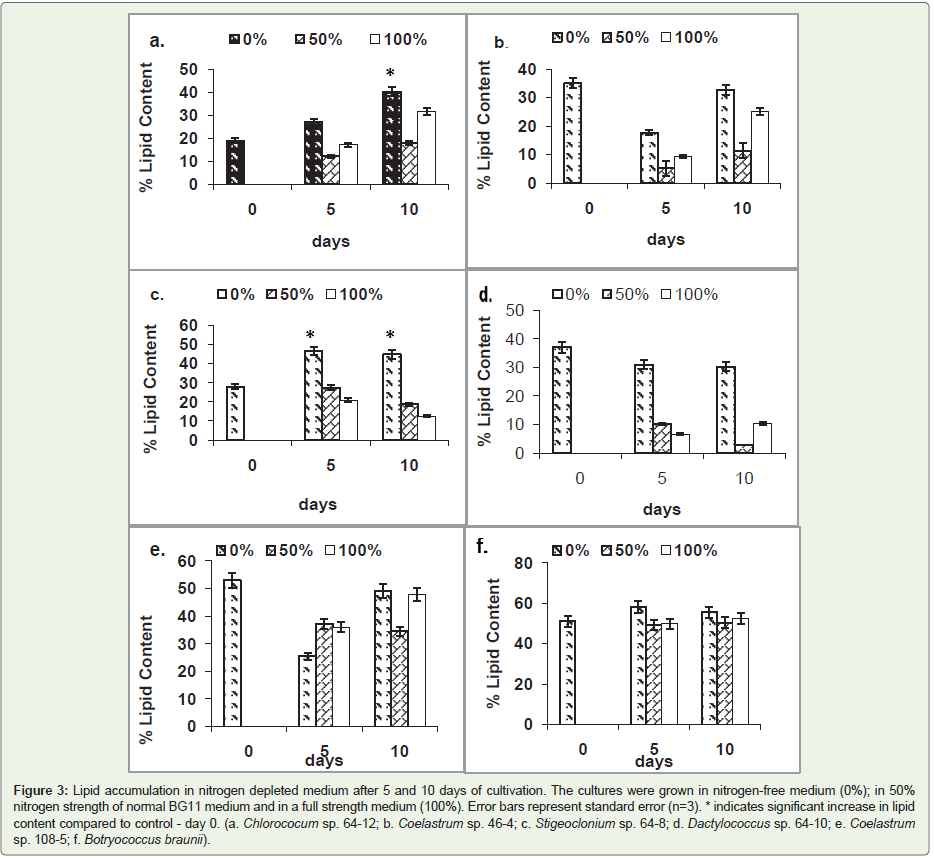
used as control (Figure 3). Of six strains tested, a significant increase
(P > 0.05)in lipid levelin nitrogen-free medium was observed in only
two strains. The lipid level increased in Chlorococcum sp. strain 64-12
(Figure 3a); and Stigeoclonium sp. strain 64-8 (Figure 3c) 105% and
57% respectively. Nitrogen depletion did not significantly affect lipid
levelin other tested strains (Coelastrum sp. strain 108-5; Dactylococcus
sp. strain 64-10; Coelastrum sp. strain46-4; Botyrococcus braunii).Effect of phosphorous concentrationon lipid accumulation:
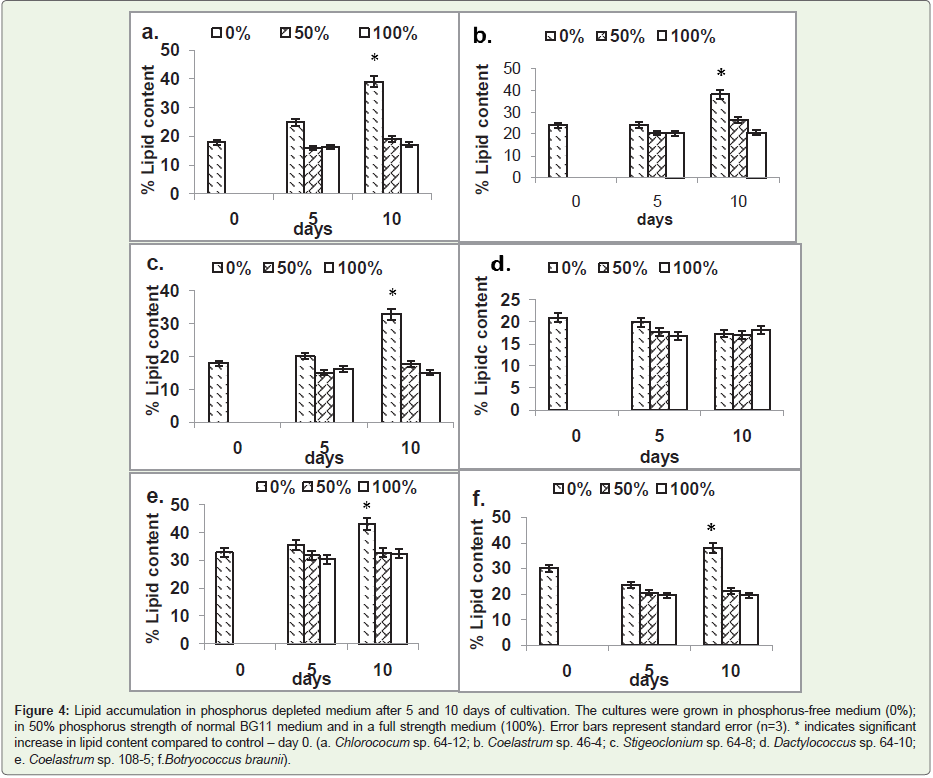
Of the six strains tested, five showed significant lipid accumulation
in phosphorus-free medium after 10 days of cultivation. The highest
increase of lipid level of 116% was observed in Chlorococcum sp. 64-
12, followed by Stigeoclonium sp. 64-8(83%); Coelastrum sp. 46-4
(58%); Coelastrum sp. 108-5 (30%); and; Botryococcus braunii (26%) (Figure 4d). In the strains affected by phosphorus depletion, the
significant accumulation of lipids occurred after 10 but not after 5
days of cultivation. Reduction of phosphorus concentration in the
medium by 50% did not affect lipid levels significantly.
(Figure 4). The only strain that showed decrease of lipid content in
phosphorus-depleted medium was Dactylococcus sp. strain 64-10
Figure 2: Lipid content determined by Nile red method as a percentage of
dry biomass of six selected strains after 13 and 45 days of cultivation. Error
bars represent standard error (n=3). Strains tested: Chlorococcum 64-12;
Coelastrum 46-4; Stigeoclonium 64-8; Dactylococcus 64-10; Coelastrum
108-5; Botryococcusbraunii (B.b.).
Figure 3: Lipid accumulation in nitrogen depleted medium after 5 and 10 days of cultivation. The cultures were grown in nitrogen-free medium (0%); in 50%
nitrogen strength of normal BG11 medium and in a full strength medium (100%). Error bars represent standard error (n=3). * indicates significant increase in lipid
content compared to control - day 0. (a. Chlorococum sp. 64-12; b. Coelastrum sp. 46-4; c. Stigeoclonium sp. 64-8; d. Dactylococcus sp. 64-10; e. Coelastrum sp. 108-5; f. Botryococcus braunii).
Discussion
An earlier study conducted by the Aquatic Species Program [23]
emphasized the importance of collecting native algal strains and
analyzing their potential for lipid accretion. Using strains that are
adapted to a climate conditions in which they are expected to be masscultured
is of crucial significance. In this study, 27 Florida-native
green algal (Chlorophyta) strains and their potential to accumulate
lipids for the purpose of biodiesel production is investigated. In this initial screening using Nile redmethod, lipid content was determined
in the cultures after 13 and 45 days. The first-time sampling (day 13)
was selected based on the study of Kalacheva [24] who reported that
neutral lipids start accumulating after 13 days of cultivation. Five
strains thatproduced 27 mg of neutral lipids or more per 100 ml of
culture after 45 days of cultivation were used for further analysis
(Table 2). In the strains selected for further study, the pattern of
both growth and lipid accumulation differed from one strain to
another. During 45 days of cultivation, two strains (Coelastrum sp.
strain 46-4; Botryococcus braunii reached stationary phase, three
strains (Chlorococcum sp. strain 64-12; Coelastrum sp. strain 108-
5; Stigeoclonium sp. strain 64-8) continued growing till the end of
experiment and Dactylococcus sp. strain 64-10 showed sharp growth
decline after 30 days.
In most strains tested, the initial lipid content decreased after
inoculation (Figure 2) a consequence of using inoculum in the late
stationary phase. This phenomenon of decreasing lipid content upon
transferring the cells into new medium has been described before
and reflects metabolic activity where the lipids are decomposed to
supply energy for cell division and growth [25]. The lipid content in
all strains, except control strain Botryococcus braunii, continued to
increase until the end of experiment (45 days). The increase in lipid
level in aged cultures is in accordance with the report of Chen [18]
who showed that the lipid content in the cells of Chlorella vulgaris
increased from 30% to 56% from day 1 to day 12. It is known that
the old cultures undergo stressful conditions and the majority of the
lipids in cells in stationary phase are neutral lipids in the form of
triacylglycerides (TAGs). This is apparently due to the shift in lipid
metabolism from synthesis of membrane to storage lipids either
through de novo biosynthesis or through conversion of membrane
polar lipids into TAGs [2].
Figure 4: Lipid accumulation in phosphorus depleted medium after 5 and 10 days of cultivation. The cultures were grown in phosphorus-free medium (0%); in 50% phosphorus strength of normal BG11 medium and in a full strength medium (100%). Error bars represent standard error (n=3). * indicates significant increase in lipid content compared to control – day 0. (a. Chlorococum sp. 64-12; b. Coelastrum sp. 46-4; c. Stigeoclonium sp. 64-8; d. Dactylococcus sp. 64-10; e. Coelastrum sp. 108-5; f.Botryococcus braunii).
Botryococcus braunii showed no increase in lipid level in aging
cultures, instead, a decrease of lipid content was observed (Figure 2).
Similarly, Alonso [21] reported that culture age had no influence in
accumulation of TAGs in the diatom Phaeodactylum tricornutum.
Possible explanation for reduced lipid synthesis in a stationary
phase of Botyrococcus braunii is that there is an increased rate of
polysaccharide accumulation [24]. Additionally, Botryococcus braunii
has a slower growth rate than other tested strains. According to Wolf
[26], the unusual morphology of this alga could possibly be the factor
for imposing constraints on growth.
The phenomenon of increased lipid level in older cultures is well
documented [27] and could have important implications on the
economics of algal biofuels [28]. Selecting strains that have a high
growth rate and simultaneously capable of accumulating high lipid
levels in a short period of time would be desirable features that could
significantly reduce the production costs of biodiesel.
Once a promising strain is selected, ability of the cell to
accumulate lipids can be further improved by manipulating the
metabolic pathways through changes in environmental conditions
[2]. It is known that under sufficient nutritive conditions, proteins
are synthesized, while during nutrient deprivation, the cell division
is suppressed and a greater amount of carbon remains available for
lipid synthesis [29].
Increased accumulation of lipids, particularly TAGs, has been
observed in response to nitrogen deficiency in various strains of
microalgae [30-32]. With nitrogen and phosphorus deprivation,
Scenedesmus sp. showed a substantial increase in lipid as high as
30% and 53% respectively [33]. This indicates different algal strains
respond differently to nitrogen deficiency. For example, diatoms do
not seem to respond to nitrogen depletion by increasing their lipid
content [34], while green algae (Chlorophyceae) show different
responses to nitrogen depletionranging from several fold increases
(e.g., Chlorella pyrenoidosa) to no change (Dunaliella species) [35].
Phosphorus deprivation has been reported as another factor that
contributes to increased lipid accumulation [36,37]. In this study, five
out of six strains tested responded to phosphorous depletion with
an increased accumulation of lipids after 10 days of culturing. Even
though it is widely accepted that a nitrogen-limitation is the most
important factor that induces lipid accumulation [38-40], in this
case, phosphorus deprivation affected most of the strains tested. In
phosphorus-depleted medium, phospholipids of cell membranes are
broken down into neutral lipids [41]. This implies that when using
nutrient deficiency as a method for increasing the lipid synthesis
and storage, those conditions should be determined for each strain
individually.
The goal of manipulating algal cell metabolism is to increase lipid
content without loss of biomass productivity; however, this appears
to be a challenging task. Under nutrient shortage, lipid levels will
increase but cellular growth will decline, resulting in a decreased lipid
production. Therefore, in order to make microalgae-based biofuel
technology feasible, it is essential to achieve a balance between lipid
production and biomass yield.
In conclusion, of twenty-seven native Florida algal strains
screened, three strains contained more than 50% of lipid of dry
biomass (Coelastrum 108-5, Chlorococcum 64-12 and Stigeoclonium 64-8) and therefore identified as potential organisms to be used in
algae-based biodiesel technology. Deprivation of nutrients such as
nitrogen and phosphorus affected various strains differently; most
strains showed better response to phosphorus rather than to nitrogen
deprivation. According to this preliminary screening, biodiversity
of naturally occurring algae in Florida aquatic environments can
provide a good feedstock in algae-based biodiesel technology.
Acknowledgement
We would like to thank David Berthold for editing our English.
References
Citation
Narendar P, Jayachandran K, Gantar M. Screening of Florida Native Green Microalgae as a Potential Source of Biodiesel. J Environ Soc Sci. 2015;2(1): 111.