Research Article
Phyto-Chemical Study of Bark Extract of Nyctanthus Arbortristits LINN. Belonging to Family Oleaceae
Laware SG1*, Shirole NL2 Gawali VK3
1Department of Pharmacognosy, PRES’s College of Pharmacy (For Women), Chincholi, India
2Department of Pharmacognosy, DCS’s A.R.A. College of Pharmacy, Dhule, India
3Sidhakala Ayurved College, Sangamner, India
*Corresponding author: Laware SG, Department of Pharmacognosy, PRES’s College of Pharmacy (For Women),
Chincholi, India; E-mail: laware.sandip@gmail.com
Copyright:© Laware SG, et al. 2022. This is an open access article distributed under the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 17/08/2022; Accepted: 30/09/2022; Published: 05/10/2022
Abstract
Nyctanthus arbortristis, a commonly known as Parijatak, has traditionally been used in treatment of rheumatic joint pain, malaria and used as an
expectorant. The stem bark is recommended for curing periodic fever. This study aims to the characterization of the bioactive constituents from extract of
Nyctanthus arbortristis in Petroleum ether, methanol using UV-VIS, FTIR and NMR. Compounds isolated from Nyctanthus arbortristis were identified are
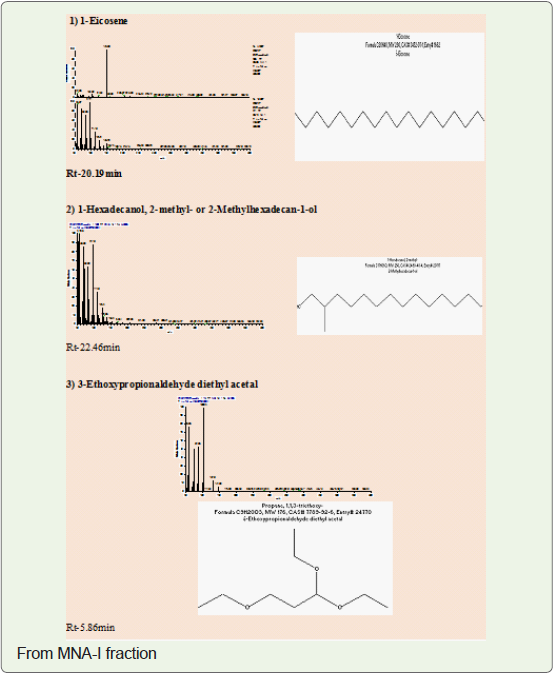
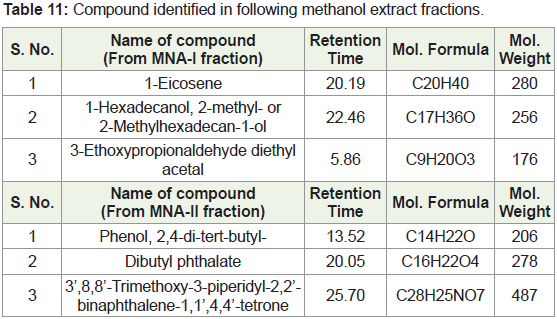
(MNA-I – fraction of ethyl acetate: methanol -1:1) as a 3-Ethoxypropionaldehyde diethyl acetal, 1-Eicosene, 1-Hexadecanol, 2-methyl- and (MNA-II - fraction
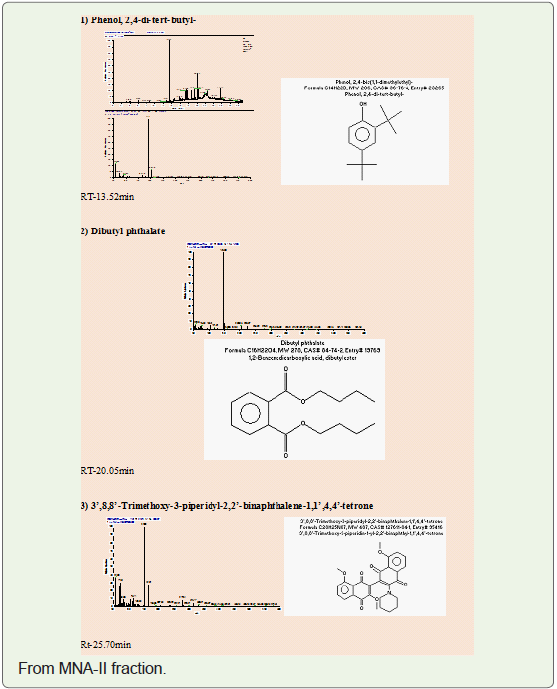
of ethyl acetate: methanol -3:7) as a 3’,8,8’-Trimethoxy-3-piperidin-1-yl-2,2’-binaphthyl-1,1’,4,4’-tetrone.
Keywords
Nyctanthus arbortristis, Bark, 1-Eicosene, 1-Hexadecanol
Abbrevations
PENA: Petroleum ether extract of Nyctanthus arbortristis; MNA:
Methanolic extract of Nyctanthus arbortristis; MNA-I: Methanolic
extract of Nyctanthus arbortristis - Ethyl acetate: Methanol (50:50);
MNA-II: Methanolic extract of Nyctanthus arbortristis - Ethyl acetate:
Methanol (30:70)
Introduction
India is one of the world’s 12 biodiversity centers with the
presence of over 45000 different plant species. India’s diversity is
unmatched due to the presence of 16 different Agri-climatic zones,
10 vegetation zones, 25 biotic provinces and 426 biomes (habitats
of specific species). However, only 7000-7500 species are used for
their medicinal values by traditional communities. In India, drugs
of herbal origin have been used in traditional systems of medicines
such as Unani and Ayurveda since ancient times. In the foregoing
works of wisdom by renowned Ayurvedic experts Charak have been
proved repeatedly by human experience all over the world. Man since
time immortal has been using herbs or plant products as medicine for
developing immunity or resistance against cold, joint pain, fever etc.
In 1994 Gupta & et.al reported as a vast majority of our population,
particularly those living in villages depend largely on traditional
remedies. In 2004, Kokate & et.al reported, the nature has provided
a complete storehouse of remedies to cure all ailments of mankind.
Since the down of civilization, in addition to food crops, man
cultivated herbs for his medicinal needs. The knowledge of drugs has
accumulated over thousands of years as a result of man’s inquisitive
nature [1-4].
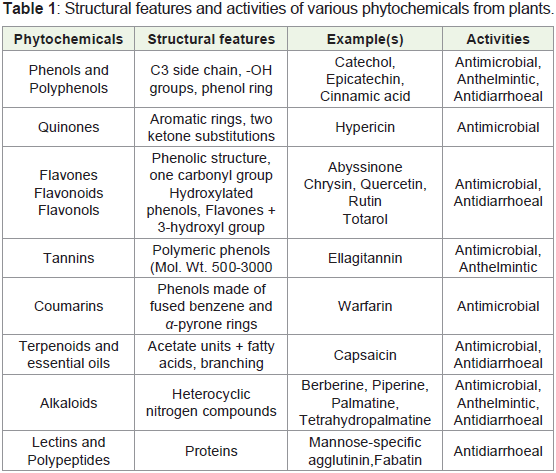
Phytochemistry is the study of phytochemicals produced in
plants, describing the isolation, purification, identification, and structure of the large number of secondary metabolic compounds
found in plants. Effect of extracted plant phytochemicals depends on:
- The nature of the plant material
- Its origin
- Degree of processing
- Moisture content
- Particle size
Carbon dioxide gas deals with the photosynthesis process in plants
in the presence of light energy. Photosynthesis and pentose pathway
together pools the phosphate group present in the sugar molecules of
plants which leads to glycolysis process and which is accounted for
producing many of phytochemicals of plants, such as, shikimic acid,
proteins, aliphatic and aromatic acids, mevalonic acids, fatty acids,
flavanoids, terpinoids, steroids etc. There are lots of medicinal plants
which contain a number of phytochemicals and those phytochemicals
are used medicine purpose to treat various kinds of diseases. In the
following table a list is shown of phytochemicals having medicinal
values [5,6].
Importance of Phytoconstituents: Therapeutic potential of plant
and animal origin are being used form the ancient times by the simple
process without isolation of pure compounds that is in the form of
crude drugs or galanicals prepared from them. The pharmacological
action of crude drug is determined by the nature of its constituents.
Thus, the plant species may be considered as a biosynthetic laboratory
not only for the chemical compounds e.g., carbohydrates, proteins
and fats that are utilized as food by humans and animals, but also for
the multitude of the compounds including alkaloids, triterpenoids,
flavonoids, glycosides etc. which exert definite physiological effects.
Generally chromatographic techniques are the most useful tool
for such purpose. Thin layer chromatography (TLC), preparative
TLC, column chromatography, HPLC, gas chromatography and
HPTLC are the various techniques for the separation and isolation of the phytoconstituents. Spectral analysis of such isolated constituents
by using following techniques as UV spectroscopy, IR spectroscopy,
GC - MS can be applied, NMR spectroscopy.
Standardization of Phytoconstituents: Organoleptic
characteristics, specific chemical tests, macroscopic analysis,
pharmacognostic parameter, microscopic analysis & comparison
using crude drug reference standard are conducted. After the proper
identification, the purity of herb should be assessed by way of
detection of foreign organic matter (i.e. insects, rodent debris etc.),
detection of foreign inorganic matter (i.e. total ash, acid insoluble ash
etc.) [7].
Materils & Methods
A) Plant Material:
a) Collection and Drying: The Bark of Nyctanthes arbortristis
belonging to the family Oleaceae was taken for present study based on the
literature survey. The crude drug was collected from Khadakewake
village, Tahasil Rahata, Ahamednagar district (M.S, INDIA).b) Plant Authentication: The plant was authenticated by P.
G. Diwakar, Joint Director, Botanical survey Of India, Pune by
comparing morphological features. The herbarium of the plant
specimen was deposited at Botanical Survey of India, Pune.
B) Preparation of N. arbortristis extracts::
A dried material was extracted with different solvents, starting
from solvent of low polarity. Initially crude drug was extracted with
petroleum ether (60-800c), furthermore was extracted with methanol.Briefly, 500 gm powdered bark of Nyctanthes arbortristis was
packed in thimble containing cotton cloth and extracted with
petroleum ether (60-800c) in soxhlet apparatus. After complete
extraction, filtrate was filtered off and solvent was recovered using
distillator. The extract was concentrated to dry residue and kept in
desiccator over adsorbent like sodium sulphate. Further, marc was
extracted using methanol.
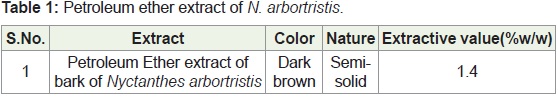
a) Preparation of Petroleum ether dried extract: The shade
dried, coarse powder of the stem bark of Nyctanthes arbortristis Linn.
(500gm) was packed well in a soxhlet apparatus and extracted with
petroleum ether (60-80°C) until the extraction was completed which
was confirmed by the color of the siphoned liquid. The extract was
filtered while hot and the resulting extract was distilled in vacuum
in order to remove the solvent completely and subsequently dried in
a desiccator. The extract was weighed and calculated the percentage
yield in terms of air-dried material.
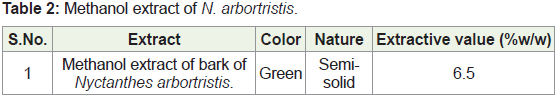
b) Preparation of Methanol dried extract: The marc was dried
in hot air oven below 50°C and packed well in Soxhlet apparatus
and extracted with methanol until the completion of the extraction.
The extract was filtered while hot and the resultant extract was
distilled in vacuum under reduced pressure in order to remove the
solvent completely and dried in a desiccator. Weighed the extract
and calculated its percentage in terms of air-dried powdered crude
material.
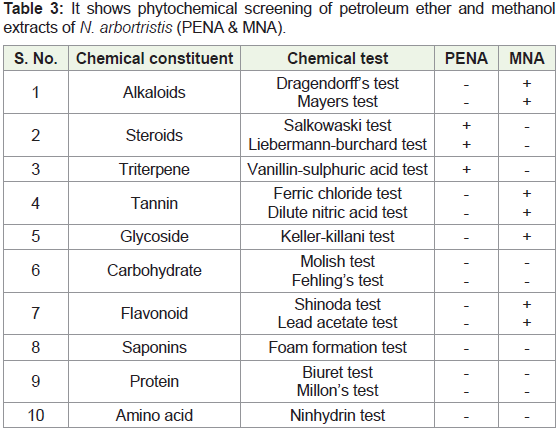
C) Preliminary Phytochemical Investigation::
The extract obtained after extraction was characterized by preliminary phytochemical test for rough ideas of main constituents
present in extracts. Alkaloid, Cardiac glycosides, Tannin, Terpenoid,
Phlobatannins, Fixed oils and fats and Flavonoid in the petroleum
and methanol extracts of the bark of N. arbortristis were identified.D) Separation & isolation of phytoconstituents by Chromatography:
Thin layer chromatography (TLC) and Column chromatography
technique is used for separation, isolation and identification of
constituents. First of all, TLC of different extracts was performed with
suitable solvent systems, different solvents were used to separate out
different chemical constituents. The TLC for PENA and MNA was
performed. Then, for isolation of the phytoconstituents, the methanol
extract was fractionated by the column chromatography.Thin layer chromatography: Thin layer chromatography (TLC)
technique is used as a base for separation, isolation and identification
of constituents. First of all, TLC of different extracts was performed
in different solvent systems, for selection of optimum mobile phase.
Different solvents were used to separate out different chemical
constituents. The TLC for PENA and MNA was performed [8].
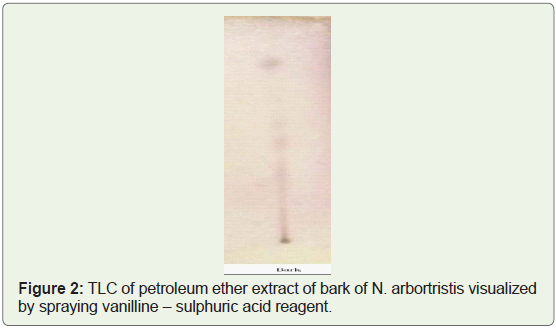
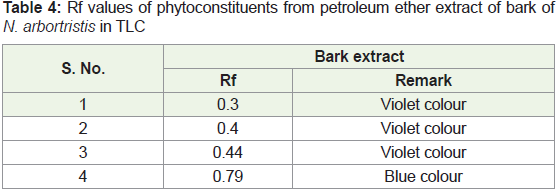
i. Thin layer chromatography of petroleum ether extract of
bark of N. arbortristis
➢ Stationary phase: Precoated silica plates (E. Merk)
➢ Mobile phase: Benzene: Ethyl acetate (9:1)
➢ Visualization: 1. Spray Vanilline – Sulphuric acid reagent
and heat at 1050C
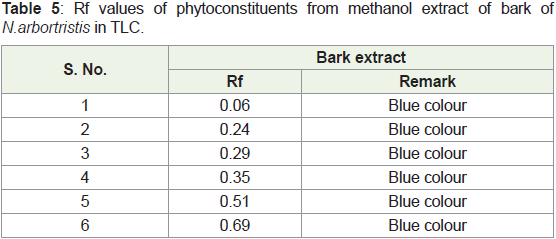
ii. Thin layer chromatography of methanol extract of bark of
N.arbortristis
➢ Stationary phase: Precoated silica plates (E. Merk)
➢ Mobile phase: Ethyl acetate: Formic acid: Glacial acetic acid:
Water (100:11:11:26)
➢ Visualization: Spray Anisaldehyde - Sulphuric acid reagent
and heat at 1050C
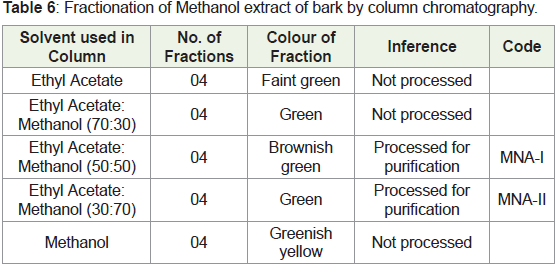
Column Chromatography: To isolate the phytoconstituents
responsible for the activity, the methanol extract was fractionated by
the column chromatography.
➢ Height of column: 30 cm
➢ Diameter of column: 3 cm
➢ Stationary phase: Silica for column chromatography
➢ Mobile phase: gradient elution method
1. Ethyl acetate
2. Methanol
Detection of spot: By spraying vanillin sulphuric acid reagent
and heating at 110oC for 10 min.
No. of fractions collected: 20
Volume of each fraction: 25ml
E) Phyto-chemical screening by Spectroscopic methods:
UV-visible spectrophotometric analysis was conducted on the
N. arbortristis extract using a UV-visible spectrophotometer with
a slit width of 2nm, using a 10-mm cell at room temperature. The
extract was examined under visible and UV light in the wavelength
ranging from 300-600nm for proximate analysis. For UV-VIS
spectrophotometer analysis, the extract was centrifuged at 3000 rpm
for 10 min and filtered through Whatman No. 1 filter paper. The
sample is diluted to 1:10 with the same solvent. Fourier transform
infrared (FTIR) was used to identify the characteristic functional
groups in the extract. It provides the information about the structure
of a molecule could frequently be obtained from its absorption
spectrum. A small quantity of the Mentha spicata extract was mixed
in dry potassium bromide (KBr). The mixture was thoroughly mixed
in a mortar and pressed at a pressure of 6 bars within 2 min to form
a KBr thin disc. Then the disc was placed in a sample cup of a diffuse
reflectance accessory. The IR spectrum was obtained using Bruker,
Germany Vertex 70 infrared spectrometer. The sample was scanned
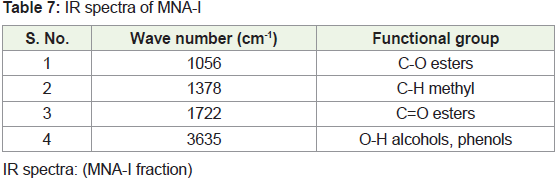
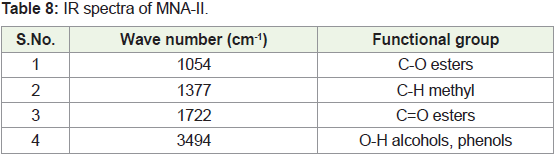
from 4000 to 400 cm-1. The peak values of the UV-VIS and FTIR were
recorded. NMR spectra were obtained from a Bruker spectrometer
using tetramethyl silane (TMS) as an internal standard in DMSO.
Chemical shifts were in ppm concerning TMS. Coupling constants
were in Hz [9-15].Results
Profile of Nyctanthes Arborstristis
Common Name: Parijatak (sad tree)
Botanical Name: Nyctanthes arbortristis
Vernacular Names: English - Night-flowering Jasmine, Coral
Jasmine
Hindi - Harashringara, Harsingar
Bangali - Sephalika
Nepali - Parijata, Paghala
Sanskrit – Parijatha
Malayalam – Parijatakam
Gujarathi – Jayaparvati
Kannada - Parijatha
Telugu – Pagadamalle
Oriya – Gangasiuli.
Plant authentication is done with Botanical survey of India,
regional office Pune.
A) Preparation of N. arbortristis extracts: The extraction values
of N. arbortristis is as follow
B) Preliminary Phytochemical Investigation:
C) Isolation & separation of phytoconstituents by
Chromatography:
a) Thin Layer Chromatography:
➢ Thin layer chromatography of petroleum ether extract of bark
of N. arbortristis
➢ Thin layer chromatography of methanol extract of bark of
N. arbortristis
b) Column Chromatography:
Table 3: It shows phytochemical screening of petroleum ether and methanol
extracts of N. arbortristis (PENA & MNA).
Figure 2: TLC of petroleum ether extract of bark of N. arbortristis visualized
by spraying vanilline – sulphuric acid reagent.
Figure 3: TLC of methanol extract of bark of N. arbortristis visualized
byspraying anisaldehyde-sulphuric acid reagent.
Table 4: Rf values of phytoconstituents from petroleum ether extract of bark of
N. arbortristis in TLC
Discussion
In the present study, we found that phytoconstituents in different
fractions separated through column chromatography. Ethyl acetate:
methanol (1:1) & ethyl acetate: methanol (3:7) fractions were
processed [16].
Conclusion
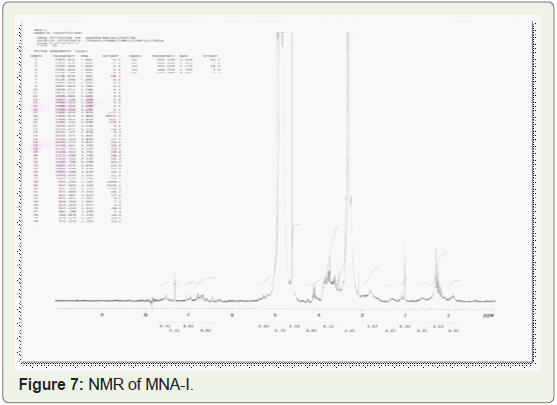
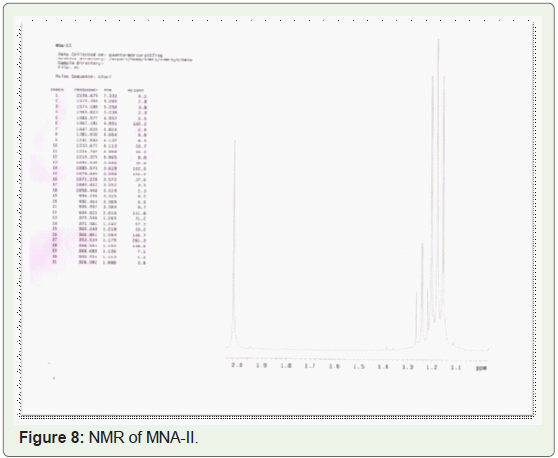
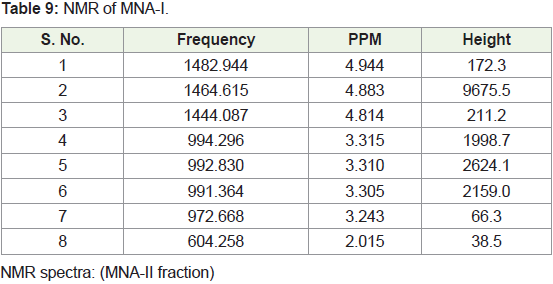
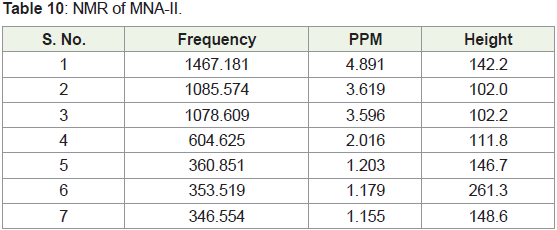
In present study, compounds isolated from this species were
identified by analytical tools like, I.R, GC-MS and NMR are (MNA-I –
fraction of ethyl acetate: methanol -1:1) as a 3-Ethoxypropionaldehyde
diethyl acetal, 1-Eicosene, 1-Hexadecanol, 2-methyl- and (MNA-II
- fraction of ethyl acetate: methanol -3:7) as a 3’,8,8’-Trimethoxy-3-
piperidin-1-yl-2,2’-binaphthyl-1,1’,4,4’-tetrone.
References
Citation
Laware SG, Shirole NL, Gawali VK. Phyto-Chemical Study of Bark Extract of Nyctanthus Arbortristits LINN. Belonging to Family Oleaceae. J
Plant Sci Res. 2022;9(2): 229