Research Article
Heterocystous Cyanoprokaryotes: Anabaena Bory ex Bornet et Flahault and Trichormus (Ralfs ex Bornet et Flahault) Komárek et Anagnostidis (Nostocaceae, Nostocales) from Tripura, India
Sarma K1,4, Kumar N4, Pandey A3, Halder NC5, Das D1 and Kant R2*
1Ramkrishna Mahavidyalaya, Kailashahar, Unakoti (Tripura) India
2Department of Botany, Chaudhary Charan Singh University, Meerut, India
3Department of Botany, C.M.P. Degree College, University of Allahabad, Prayagraj, India
4Amity Institutes of Biotechnology, Amity University of Haryana, Maneser, Gurugram, India
5Department of Botany, Uluberia College, Howrah, India
*Corresponding author: Kant R, Department of Botany, Chaudhary Charan Singh University, Meerut 711315, India;
E-mail: ramakant.algae@gmail.com; rkojha_1@rediffmail.com
Copyright: © Sarma K, et al. 2022. This is an open access article distributed under the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 04/07/2022; Accepted: 08/08/2022; Published: 10/08/2022
Abstract
Heterocystous blue green algae are most fascinating group of microorganisms due to their unique characteristic bestowed with diazotrophic nitrogen
fixation. The blue green algae genera like Anabaena and Trichormus are common forms and often found growing in aquatic habitats. Present paper deals
with the diversity of two blue green algal genera Anabaena and Trichormus from Tripura. In the present paper, we report total 18 species, out of which 12
species belong to the genus Anabaena and 6 species belong to the genus Trichormus, family Nostocaceae, order Nostocales, Cyanoprokaryota. Out of 18
species, 11 species of Anabaena and 6 species of Trichormus are new to the flora of Tripura, India.
Keywords
Biodiversity; Biotopes; Cyanoprokaryotes; Heterocysts; Rice field; Tripura
Introduction
Cyanoprokaryotes (Blue green algae/Cyanobacteria) are the
first photosynthetic simple microorganisms and some of them
particularly heterocystous forms are bestowed with unique potential
of diazotrophic nitrogen fixation along with carbon fixation [1].
Because of these unique combinations of two entirely opposed
physiological processes i.e. oxygen evolution as a by-product of
carbon fixation via photosynthesis and diazotrophic nitrogen fixation
they contribute significantly in the ecosystem as well as nitrogen
economy [2]. They help in increasing the soil fertility and carbon sequestration. They gained a lot of attention in recent years because of
their potential applications in biology, biotechnology and agriculture.
The biomass of heterocystous blue-green algae is considered as one
of the valuable natural source of bio-fertilizer to increase the fertility
of the soil and improve physico-chemical characteristics of soils such
as water-holding capacity and mineral nutrient status of the soil [3].
The cyanoprokaryotes are classified conventionally on the basis
of morphological parameters [4,5] or following polyphasic approach
[6]. Later, Komárek et al. [7] revised the system of classification of
cyanoprokaryota based on molecular characterization, cellular ultra structure and thylakoid arrangement. Cyanobacteria represent a
major component of the photosynthetic microorganism community
of most of the aquatic and terrestrial ecosystems [8], but may grow
in a wide range of habitats including rice fields. As the North Eastern
part of India is considered as one of the mega hotspots for its diversity
richness including cyanobacteria. Cyanobacteria are found in diverse
habitats of Tripura, a north eastern state of the country India.
The genus Anabaena Bory ex Bornet et Flahault and Trichormus
(Ralfs ex Bornet et Flahault) Komárek et Anagnostidis are environmentally
and economically two important blue green algae and
identified mainly on the basis of morphological characteristics,
such as shape and size of trichomes and vegetative cells, size and
location of heterocysts and akinetes [9]. The genus Anabaena was
classified by Geitler [4] and Starmach [10] traditionally on the basic
concept of containing a wide spectrum of planktic and benthic
types. Desikachary [5] on the basis of morphological and ecological
description, identified and accepted the genus Anabaena by the
presence of uniform trichomes, absence of sheath or presence of more
or less diffluent sheath forming free or floccose or soft mucilaginous
thallus with heterocysts, generally intercalary and presence of a single
or series of spores near the heterocyst or between the heterocysts. On
the other hand, the genus Trichormus was traditionally described
under the name Anabaena, but Komárek and Anagnostidis [11]
described Trichormus as a separate genus using a polyphasic approach
but the strategy of akinete formation in both the species is completely
different [12]. Later 16s RNA sequencing confirmed the similarity of
Trichormus with Nostoc and Dolicospermum [7,13].
The main aim of the present investigation was to study the diversity
and distribution pattern of the genus Anabaena and Trichormus from
different habitats of Tripura, India. From the present study, we are
reporting total 18 strains of which 12 species belonging to the genus
Anabaena viz. A. constricta; A. duployae; A. ghosei, A. hieronymii;
A. minuta; A. oblonga; A. orientalis; A. papillosa; A. schauderi; A.
sedovii; A. spinosa, A. torulosa and 6 species belonging to the genus
Trichormus viz. T. azollae; T. ellipsosporus; T. gelatinicola; T. minor;
T. naviculoides; T. subtropicus. Out of total 18 strains, 17 strains (11
strains of Anabaena and 6 strains of Trichormus) are new to the flora
of Tripura.
Material & Methods
The sites of present study were different biotopes of Tripura
state of India. The Tripura lies between 22°56’-24°32’ N latitude and
91°09- 92°20’E longitude (Figure 1A-B). Total 1150 algal samples
were collected randomly from different habitats of Tripura during
the year 2017-2021. All the collected samples were mixed thoroughly
by homogenizer (Remi-RQT-127AD) and transferred into sterilized
petridishes (Borosil) filled nitrogen deficient liquid and solid BG-
11 culture medium [14], and total 149 strains of Anabaena and
Trichormus were raised as unialgal cultures based on method
described by Kant et al., [15]. Out of total 149 strains, 110 were of
Anabaena and 39 strains of Trichormus. The morphological details
of Anabaena and Trichormus strains were observed with the help
of Trinocular Research Microscope (Olympus, CH20i microscope)
fitted with digital camera (Magnus, Magcam DC10) and their
morphological details were recorded. All the isolated strains of
Anabaena and Trichormus were identified up to the species level with the help of available literatures and monographs [4-6]. Morphological
details of eighteen strains, one strain from each species of Anabaena
and Trichormus are being described in the present paper based on
Nostocales [6].
Results
Survey, collection of samples from rice fields and uncultivated
moist soils and microscopic analysis of algal growth of natural
material and culture, and their analysis of revealed the occurrence
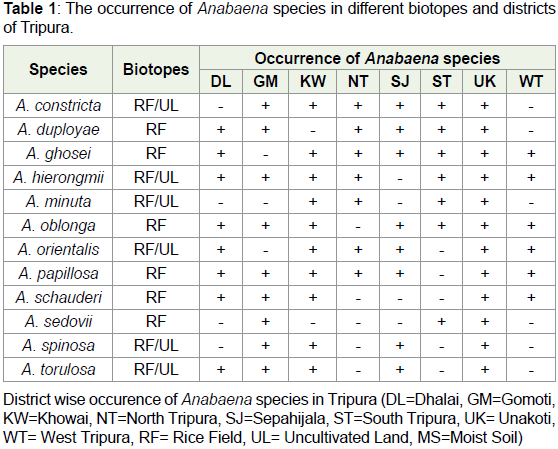
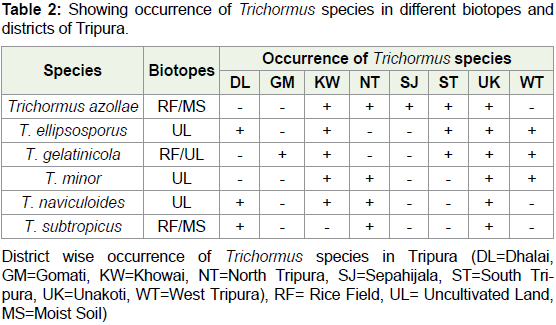
of total 149 strains of two genera Anabaena and Trichormus. Out of
which six strains of Anabaena usually grow in rice field and six strains
grow in both types of biotopes rice field as well as uncultivated moist
soils. However, as for as the occurrence of Trichormus is concerned
three species viz. T. ellipsosporus, T. minor and T. naviculoides grow in
uncultivated land and rest three grow in both rice field as well as moist
soil of uncultivated land. Results also revealed maximum occurrence
of Anabaena species in Unakoti district and minimum six species
in West Tripura district (Graph 1). However, as for occurrence of
Trichormus species are concerned maximum were found in Unakoti
and minimum in Gomati and Sepahijala district of Tripura. Detailed
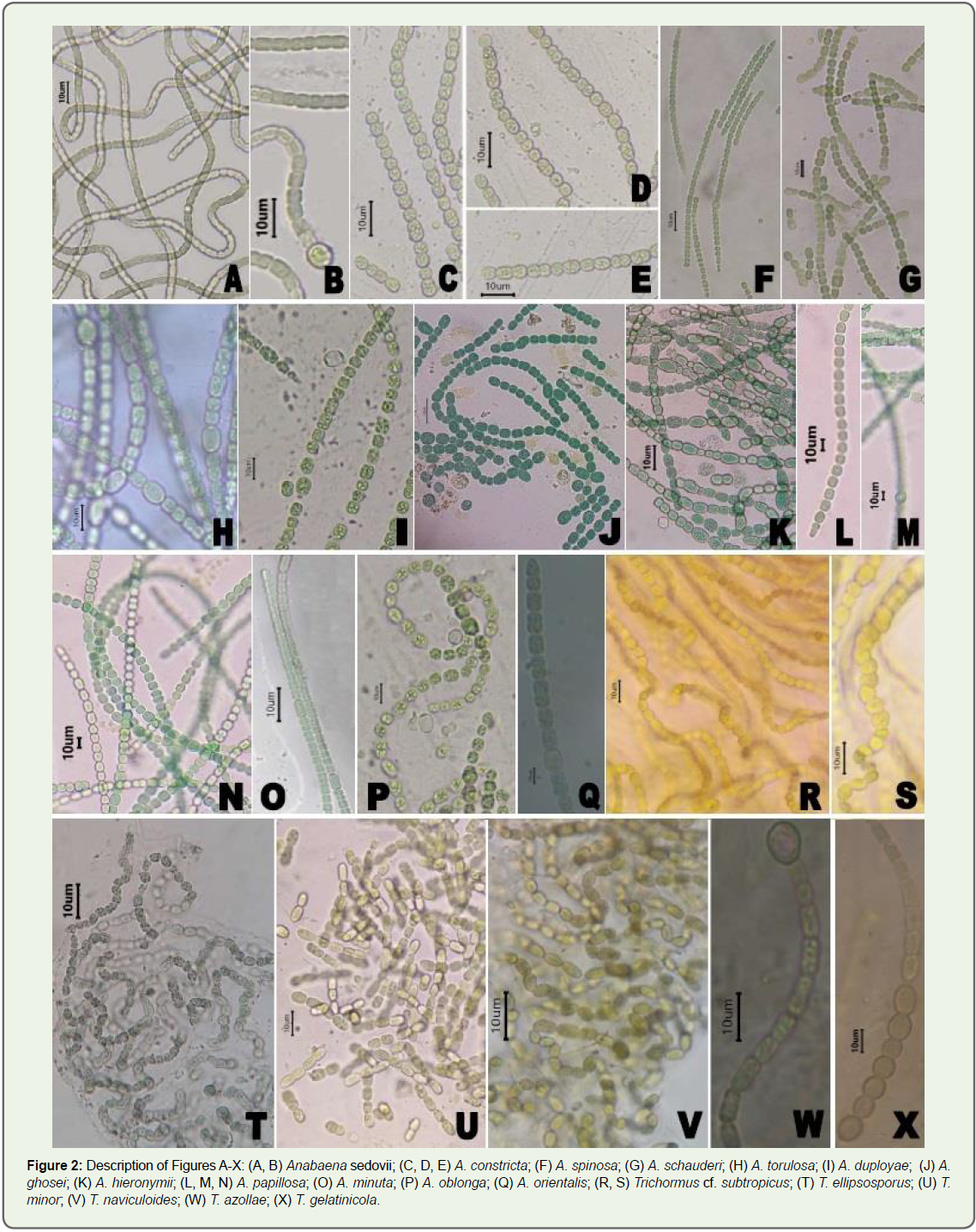
results are given in Table 1 and 2 and Figure 2A-2X.
Figure 2: Description of Figures A-X: (A, B) Anabaena sedovii; (C, D, E) A. constricta; (F) A. spinosa; (G) A. schauderi; (H) A. torulosa; (I) A. duployae; (J) A.
ghosei; (K) A. hieronymii; (L, M, N) A. papillosa; (O) A. minuta; (P) A. oblonga; (Q) A. orientalis; (R, S) Trichormus cf. subtropicus; (T) T. ellipsosporus; (U) T.
minor; (V) T. naviculoides; (W) T. azollae; (X) T. gelatinicola.
Morphological Observation:
Description of Anabaena and Trichormus species:
Anabaena constricta (Szafer) Geitler (Figure 2C-D & Figure 2E)
The colonies are usually microscopic, blackish-green or dark
blue-green, mucilaginous mats. Trichomes cylindrical, 5-7 μm wide,
composed from short, cylindrical to barrel–shaped cells. Heterocysts
are intercalary, solitary, spherical, 3.9-4.8 μm in diameter, Absence
of akinetes.Anabaena duployae Welsh (Figure 2I)
The colonies are usually microscopic, blue-green, free floating,
mixed with other algae. The filaments presents as solitary or in small clusters. Trichomes straight or curved constricted at the cross-walls,
and sometimes enclosed in thin, faint, colourless, diffluent mucilaginous
sheaths. Cells are sub-oval, shorter than wide, with unclear presence
of gas vesicles, 4-5 μm wide 6-7 μm long. Terminal cells do not differ
from other vegetative cells. Heterocysts are sub-globular, shorter
than wide, 8 μm wide and 8-9 μm long. Akinetes are intercalary, oval,
single or in pairs, distant from heterocysts, 14-17 μm long 10-12 μm
wide, with thick, smooth exospore.
Anabaena ghosei Welsh (Figure 2J)
[syn.: Anabaena variabilis sensu Ghose]
The microscopic colonies are forming gelatinous, dark blue-green
mats. Filaments short, 100-320 μm long. Trichomes without sheaths,
constricted at the cross-walls. The cells are barrel-shaped, 4-4.5 μm
long and 4-4.3 μm wide. Akinetes are oval, solitary, distant from
heterocysts, 5.5-7.5 μm long and 4-5 μm wide.
Anabaena hieronymii Lemmermann (Figure 2K)
[Syn.: Anabaena hieronymusii Lemmermann]
The microscopic colonies are mucilaginous, blue green forming
amorphous mats. Filaments straight or slightly flexuous constricted at
cross-walls, not attenuated towards ends. Cells are longer than wide,
ellipsoidal, 5-6 μm long and 3-4 μm wide, with rounded terminal
cells. Heterocysts are intercalary, solitary elongated, ellipsoidal, 9-10
μm long and 2.5-4.5 μm wide. Akinetes are cylindrical with rounded
ends, distant from heterocysts usually in rows of 2-4, akinetes 20-36
μm long and 5-8 μm wide.
Anabaena minuta Welsh (Figure 2O)
The colonies are microscopic, with solitary filaments, constricted
at the cross-walls, very slightly attenuated towards ends. Cells are
cylindrical to oval, 5 μm long and 2.5 μm wide. Heterocysts are
intercalary or apical, 5-7 μm long and 3-4 μm wide. Akinetes are not
observed.
Anabaena oblonga Wildemann (Figure 2P)
The colonies are microscopic, pale yellow-green or blue-green,
with filaments solitary or in mats. Trichomes irregularly flexuous and
coiled, without mucilaginous envelopes, slightly narrowed towards
ends, constricted at cross-walls. Cells barrel-shaped, isodiametric
or shorter or longer than wide, with pale yellow-green or bluegreen
contents, 4-5 μm wide, end cells rounded or conical rounded.
Heterocysts are cylindrical or oval, isodiametric or elongated, solitary,
6-11.2 μm long and 5-7.3 μm wide. Akinetes arise from heterocysts,
solitary or often in pairs up to 4 in a row, widely cylindrical, with
flattened ends, 9.6-25.6 μm long and 6-8.2 μm wide, with yellowish or
brownish endospore and colourless exospore.
Anabaena orientalis Dixit (Figure 2Q)
The colonies are microscopic, gelatinous forming blue-green
mat like clusters. Filaments single or in small, thin mat like clusters.
Trichomes straight, slightly coiled, constricted at the cross-walls,
attenuated towards ends. Cells pale blue green, cylindrical or slightly
barrel-shaped, almost isodiametric or up to two times longer than
wide, with finely granular content, 3.7- 4.8 μm wide and 2.5-8 μm long.
Apical cells conical-rounded. Heterocysts are intercalary, solitary
on both sides of heterocysts, solitary or (rarely) in pairs, ellipsoidal
or oval, 13-24 μm long and 7.4-14.3 μm wide, with smooth surface,
brownish endospore and colourless or slightly brownish exospore.
Anabaena papillosa Hirano (Figure 2L-N)
The colonies are microscopic, blue-green, forming mats.
Trichomes straight or slightly flexuous, constricted at the cross-walls,
slightly narrowed towards ends. Cells are spherical to oval, barrelshaped,
blue-green, with “gas vesicles”, isodiametric or elongated,
8-10 μm long and 7.5-8.8 μm wide. Heterocysts are similar to
vegetative cells, mostly oval, 11-12 μm long and 10.5-11 μm wide.
Akinetes are cylindrical to ellipsoidal-oblong, flattened at the poles,
mostly solitary, distant from heterocysts, 35-55 μm long and 13-16
μm wide.
Anabaena schauderi Welsh (Figure 2G)
The colonies are usually microscopic, mucilaginous, blue-green
mats. Filaments solitary or several in mucilaginous tubes, distinctly
constricted at the cross-walls, not attenuated towards ends. The cells
are barrel-shaped, 4.6 μm long and 3.2-3.9 μm in wide. Heterocysts
are ellipsoidal-oval, larger than vegetative cells, 6.5 μm long and 4.6
μm wide. Akinetes are oval distant from heterocysts, 10 μm long and
3.5- 4 μm wide.
Anabaena sedovii Kosinskaja (Figure 2A-B)
The microscopic colonies are irregular, flat, gelatinous, dirty
olive-green mats. Filaments solitary, straight or flexuous, short, often disintegrating, deeply constricted at the cross walls. Cells are
barrel-shaped to spherical, bright blue green, 3-4.8 μm in diameter
with rounded end cells. Heterocysts are solitary, intercalary, spherical
usually of the same width, rarely a little wider in diameter of vegetative
cells. Akinetes are solitary or in a row, cylindrical with rounded ends,
with smooth, thin, colourless exospores, distant from the heterocysts
or sometimes joint to both sides of heterocysts, 7.2-16.8 μm long and
3.6-5.4 μm wide.
Anabaena spinosa Laloraya et Mitra (Figure 2F)
The colonies are microscopic, free-floating amongst other algae.
Trichomes are straight or curved, moniliform, constricted at crosswalls,
without mucilaginous sheaths. Cells are mostly spherical and
isodiametric up to barrel-shaped, 3 μm wide; end cells truncate-conical,
up to 5 μm long. Heterocysts are solitary, intercalary, spherical up to
barrel-shaped, 4-5 μm long and 5 μm wide. Akinetes are spherical or
slightly elongated, at one or both sides of a heterocysts, 12-14 μm in
diameter, with thick, yellowish exospore with short spines.
Anabaena torulosa (Carmichael) Lagerheim ex Bornet et
Flahault (Figure 2H)
The colonies are microscopic, fine mucilaginous, confluent, blue
green mats. Trichomes flexuous, cylindrical, joined and entangled in
the mats, to the ends sometimes slightly narrowed. Cells are barrelshaped,
isodiametric or slightly shorter or longer than wide, blue
green, 4.2-5 μm wide; with conical terminal cells. Heterocysts are
ellipsoidal or almost spherical, 6-10 μm long and 6 μm wide. Akinetes
are joined to both sides to heterocysts, solitary or in a rows, widened,
cylindrical (sometimes slightly concave in the middle of sites), flatrounded
at the ends, 14-24 μm long 7-12 μm wide, with smooth,
brownish exospore.
Trichormus azollae (Strasburger) Komárek et Anagnostidis
(Figure 2W)
[Syn.: Anabaena azollae Strasburger]
The colonies are usually microscopic, light blue-green or yellowgreen
mats, sometimes forming mucilaginous envelopes. Trichomes
are relatively short, straight or irregularly bent, constricted at the
cross-walls, not attenuated at the ends or very indistinctly attenuated.
Cells are variable in form, sub-globose or elongated or broadly
ellipsoidal, barrel-shaped to cylindrical. Cells 2.5-9.5 μm long and 1.8-
5 μm wide, blue green, sometimes with prominent granules. Apical
cells rounded, but often also conical. Heterocysts solitary, very rarely
in pairs, intercalary or less commonly in terminal positions, broadly
ellipsoidal, conical to cylindrical, distinctly larger than vegetative
cells, 6-11.5 μm long and 5-9.5 μm wide. Akinetes are varying from
elongate-ellipsoid to broadly cylindrical, up to 7 μm long and 4 μm
wide.
Trichormus cf. subtropicus Silva and Silva & Pienaar (Figure 2R-S)
The colonies are usually microscopic, pale yellow-green or
olive-green, forming gelatinous mats. Filaments single or entangled,
straight. Sheaths are mucilaginous, colourless, inconspicuous.
Trichomes are constricted at the cross-walls. Cells are olive-green, quadratic, longer than wide, 3.4-4.3 μm long and 3.1-5 μm wide.
Heterocysts are intercalary, subspherical, quadratic to cylindrical,
5.3-7.8 μm long and 3.7-5.9 μm wide. Akinetes are in rows, subspherical
or oblong, 5-9.3 μm long and 5.3-7.8 μm wide.
Trichormus ellipsosporus (Fritsch) Komárek et Anagnostidis
(Figure 2T)
The colonies are microscopic, pale blue-green, with irregularly
entangled filaments. Trichomes enveloped by thick, unclear,
gelatinous envelope, constricted at the cross-walls. Cells long
barrel shaped up to cylindrical, 3.6-7.5 μm long and 3-5 μm wide.
Apical cells cylindrical and rounded. Heterocyst slightly wider than
vegetative cells, spherical, oval to cylindrical, 5-9-11.5 μm long and
4.5-8.9 μm wide, also occurring in terminal position, spherical or
elongated. Akinetes are in rows distant from heterocyst, ellipsoidal,
slightly wider than vegetative cells 7.5-15.6 μm long and 4.6-8 μm
wide.
Trichormus gelatinicola (Ghose) Komarek et Anagnostidis
(Figure 2X)
[syn.: Anabaena gelatinicola Ghose]
The colonies are microscopic, mucilaginous, light blue-green or
olive green thick mats. Trichomes are mostly solitary, spirally coiled
in circular formations, less frequently straight or slightly flexuous,
constricted at the cross-walls. Cells are sub spherical, 6-7.5 μm wide;
and cells conical narrowed and pointed. Heterocysts are spherical, 7-8
μm wide. Akinetes are in rows, distant from heterocysts, spherical, ±
14 μm in diameter.
Trichormus minor (Laloraya et Mitra) Komarek et Anagnostidis
(Figure 2U)
[syn.: Anabaena catenula var. minor Laloraya et Mitra]
The colonies are microscopic, gelatinous, expanded, blue green
or yellow brown. Trichomes free floating, curved, with diffluent,
mucilaginous envelopes. Cells are yellow-brown, cylindricalrounded,
3.5-5 μm long and 3.5-4 μm wide, with rounded end cells.
Heterocysts are terminal and intercalary, akinetes many in chains,
mostly cylindrical, sometimes oblong, 10-18.5 μm long and 5-6.5 μm
wide, with smooth exospore.
Trichormus naviculoides (Fritsch) Komarek et Anagnostidis
(Figure 2V)
[syn.: Anabaena naviculoides Fritsch]
The colonies are microscopic, flat, mucilaginous, blue green
forming thin mats. Trichomes long, cylindrical, flexuous or coiled,
constricted at the cross-walls. Cells are barrel-shaped, iso-diametrical
or shorter or longer than wide, 3.5-5 μm wide. Apical cells are conical
and obtusely acuminate. Heterocyst is single, intercalary, barrelshaped,
iso-diametric or longer than wide, 5-6 μm wide. Akinetes are
serially in rows, sometimes slightly irregularly situated, to irregularly
aggregated, ellipsoidal, narrowed and obtuse towards ends, 8.5-12.5
μm long and 5.5-7 μm wide, with thin, hyaline cell-wall and wide
hyaline, gelatinous envelope.
Discussion
Cyanobacteria (Blue-green Algae) have been most interesting
group of microorganisms since long decade because of their
contribution as primary colonizer in the ecosystem and nitrogen
fixation [16-20]. The taxonomy of the filamentous heterocystous
Blue-green algal genera including Anabaena and Trichormus has
been very much disputed due to their many morphotypes and
genotypes. Trichomes of both the genera Anabaena and Trichormus
are differentiated into vegetative cells, heterocysts, akinetes [21]. As
the taxonomic entry totally depend on trichome and characteristics
of the vegetative cell, heterocyst and akinetes. The genus Anabaena
and Trichormus are controversial due to the occasional absence of
these characters and due to phenotypic changes under different
environmental conditions. Currently, the genera Anabaena and
Trichormus belong to order Nostocales, family Nostocaceae and
subsection IV.I by bacteriological classification [22]. At the world
level total 339 species of Anabaena [23] and 44 species of Trichormus
[24] are listed in the database, out of which 223 species of Anabaena
and 36 species of Trichormus have been accepted taxonomically
in Algae Base. Komárek [6] reported 88 species of Anabaena and
34 species of Trichormus. Desikachary [5] reported total 37 taxa of
Anabaena including 25 species 11 varieties and one forma.
In India, the blue-green algae have been explored by numerous
phycologists from different states and some of important contribution
include Mitra [25], Desikachary [5], Bharadwaja [26], Pandey and
Mitra [27], Tiwari [28], Sinha and Mukharjee [29], Tiwari and
Pandey [30], Prasad and Mehrotra [31], Anand [32], Santra [33],
Tiwari et al. [19], Roy et al. [34], Snehee and Verma [35], Singh et al.
[36], Maurya and Paliwal [37] and Singhet al. [38], but most of the
North Eastern region of India still remains less explored [39, 43-45].
Although few phycologists explored the blue-green algae of North
Eastern states of India. The rice fields of Tripura have been explored
by a few researchers in search for the cyanobacterial diversity [42],
but information on growth and occurrence of heterocystous forms
including Anabaena and Trichormus are very scanty from Tripura.
Tiwari et al. [19] made an exhaustive survey based on literature
of Indian phycologists and reported total one hundred taxa including
sixty five species and thirty five variety and forma of Anabaena from
all the types of Indian habitats [42], out of which total fifty eight
species including thirty eight species and twenty variety and forma
were from rice field soils of the country and they revealed that out
thirty eight species of rice field soils only five species including
Anabaena ambigua, A. sphaerica, A. fertilissima, A. oryzae and A.
variabilis are common species of Anabaena of rice fields but ignored
the rice field soils of Tripura in their study. Bhattacharya and Gupta
[46] reported nine species of Anabaena on algal collection in Central
National Herbarium (CAL). Singh et al., [39], reported total eight
species of Anabaena from rice field’s soils of Tripura. Occurrence of
maximum (12) species of Anabaena and (06) species of Trichormus
in Unakoti district and minimum (06) species of Anabaena in West
Tripura and one species of Trichormus in Gomoti and Sepahijala
district may be due to more and less collection of samples from the
respective districts of the Tripura. However, as for occurrence of
Trichormus species are concerned maximum were found in Unakoti
and minimum in Gomati district of Tripura.
In our present study, we are reporting 18 species belonging to
two heterocystous blue green algae,Anabaena and Trichormus, out
of which except A. torulosa, eleven species of the genus Anabaena
viz. A. constricta; A. duployae; A. ghosei, A. hieronymii; A. minuta; A.
oblonga; A. orientalis; A. papillosa; A. schauderi; A. sedovii; A. spinosa,
A. torulosa and 6 species of the genus Trichormus viz. T. azollae; T.
ellipsosporus; T. gelatinicola; T. minor; T. naviculoides; T. subtropicus
are new to the flora of Tripura.
Conclusion
On the basis of field’s survey and collection of blue-green algal
growth samples, and culturing and their morphological observations,
it is concluded that, the rice fields of Tripura, India harbor a good
number of heterocystous cyanobacteria but most of them belong
to Anabaena and Trichormus species. Further, it is also concluded
that occurrence of the species Anabaena and Trichormus in the rice
fields are comparatively more in numbers, which may be used as bioinoculants
of biofertilizer in the rice fields of Tripura and but needs
more thorough study before using them as bio-inoculants.
Acknowledgement
Authors (RK, KS & DD) are thankful to the Principal, Ramkrishna
Mahavidyalaya, Kailashahar, Tripura for providing necessary
facilities. Authors are also thankful to the Head, Botany Department,
Chaudhary Charan Singh University, Meerut for providing necessary
facilities. We are also thankful to Dr. G.L. Tiwari, Ex. Professor and
Head, Botany Department, Allahabad University, Prayagraj, India
for identification of Anabaena and Trichormus. Authors (RK, KS &
DD) thankfully acknowledge financial support by the Ministry of
Environment, Forest and Climate Change, Govt. of India, New Delhi.
References
5. Castenholz RW (1989) Oxygenic photosynthetic bacteria. Bergey's manual sys bacteriol 3: 1710-1806.
24. Prasad BN, Mehrotra RK (1980) Blue-green Algae of paddy fields of Uttar Pradesh. Phykos 19: 121-128.
Citation
Sarma K, Kumar N, Pandey A, Halder NC, Das D, et al. Heterocystous Cyanoprokaryotes: Anabaena Bory ex Bornet et Flahault and Trichormus
(Ralfs ex Bornet et Flahault) Komárek et Anagnostidis (Nostocaceae, Nostocales) from Tripura, India. J Plant Sci Res. 2022;9(2): 225