Research Article
Physiological and Biochemical Changes during Seed Development in Hopea ponga (Dennst.) Mabberley: An Endemic Endangered Tree Species of Western Ghats
Mithun V, Pradeep NS and Krishnan PN*
Malabar Botanical Garden and Institute for Plant Sciences, India
*Corresponding author: Krishnan, KSCSTE - Malabar Botanical Garden and Institute for Plant Sciences, Guruvayoorappan College P.O. Kozhikode Kerala, India, PIN 673014
Copyright: © Mithun V, et al. 2020. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 14/05/2020; Accepted: 11/06/2020; Published: 15/06/2020
Abstract
Hopea ponga (Dennst.) Mabberley is an endemic and endangered tree belonging to family Dipterocarpaceae, is economically important as timber
and reported with several active compounds that have effective as antioxidant and antimicrobial activities. Due to its high economic and medicinal value,
the species is over exploited and has become rare and endangered and need high conservation protocols. The present study includes physiological (fresh
and dry weight, moisture content and germination) and biochemical changes (total soluble sugars, total soluble protein, lipids, total phenols) during seed
development in Hopea ponga. Mass flowering of H. ponga occurred during the month of February and extended upto April followed by mass seed set, that
took two months for its maturation. The majority of individuals have developed their seeds in the month of April to June and dispersal occurs with the help
of wind. Changes in fresh and dry weights, % MC showed a sigmoidal pattern during seed development and maturation. Total soluble sugar and protein
recorded an increase with the seed development while starch and phenols were followed the sigmoidal pattern and were significant as per calculated r2.
Introduction
Hopea ponga (Dennst). Mabberley is an endemic and endangered
tree belonging to family Dipterocarpaceae, found in the tropical
evergreen forests distributed all along the Western Ghats of
Tamil Nadu, Kerala, Maharashtra and Karnataka [1]. The tree is
economically important as timber, the bark is also a good tanning
material and astringent with slow speed of diffusion [1]. The wood
obtained from the H. ponga is used for building construction,
wooden article and furniture preparation [2]. The effect of desiccation
on germination and vigour of seeds of the Hopea parviflora and
Hopea ponga was carried out [4]. The influence of wing loading and
viability of seeds of Hopea ponga was investigated [5]. Phytochemical
evaluation, antioxidant and antibacterial activity of seed wings of
Hopea ponga has been evaluated [6]. The entire plant can be used to
cure piles [7]. The methanolic extracts of the leaf of H. ponga revealed,
excessive scavenging ability towards specific free radicals. Further,
the tree reported with several active compounds that have effective
as antioxidant and antimicrobial activities [8]. Due to its commercial
value, economic and medicinal importance, this tree has been illegally
cut down for various commercial uses in the past few decades. The
seeds of Dipterocarpaceae species are generally short lived and
are incapable of overcoming desiccation [9]. Knowledge on seed
development is essential in case of endemic and endangered species
for successful application of conservation strategies. There has been
no report on the seed development and related biochemical changes
in recalcitrant seeds of endemic and endangered Dipterocarpaceae
members of Western Ghats. Therefore the present study was undertaken to evaluate the seed development from fertilization
to maturity. Data includes physiological (fresh and dry weight,
moisture content and germination) and biochemical changes (total
soluble sugars, total soluble protein, lipids, total phenols) during seed
development in Hopea ponga.
Materials and Methods
Healthy and disease free Hopea ponga trees (15 numbers)
naturally grown in the campus of KSCSTE-Malabar Botanical Garden
and Institute for Plant Sciences (MBGIPS), Kozhikode Kerala India
was selected and observed regularly for the detailed studies on
phenology and seed development. Frequent visits have been made
to each tree to observe flower initiation, anthesis, seed development
etc. For ascertaining the pollen-ovule ratio, the number of pollen
grains has been calculated using a Haemocytometer. The number of
ovules in an ovary has been calculated by taking section of the ovary
at young stage. Total number of flowers in a tree was calculated by
counting the number of flowers per inflorescence and then number
of inflorescences per branch. By counting the number of branches per
tree, average number of flowers has been calculated. Stigma maturity
has been recognized by observing the stigma at the time of flowering.
Average number of buds/flowers produced in an inflorescence and
branches are assessed, along with the number of seeds produced in
each inflorescence also counted. This will give an idea about the seed
production and thereby reproductive efficiency of the plants.
For the experimental studies flowers were tagged on the day
of opening and the developing seeds were collected periodically
at an interval 4 days, from 4 DAA to 60 DAA for H. ponga. For the
pollination and related studies both ex situ and in situ observations
were made. For the laboratory studies the flowering twigs were taken
and kept in a moist environment till the observations. The collected
developing seeds were sealed in polythene bags to avoid moisture loss.
The diameter and length of the fruits were measured using Vernier
callipers and their weight by an electronic balance (ANAMED, Model
Z-400). External features of the seeds like texture, colour and hairiness
etc. were also noted and this helped to identify the approximate age of
the developing seeds. Percent Moisture Content (MC) of the collected
seeds were analyzed by Low Constant Air Owen method (103 °C for
17 hours) as stipulated by ISTA.
Sample materials after taking dry weights at different periods were
used as a source material for estimating metabolites like total soluble
sugar, phenols, total soluble proteins and starch. Three samples of
each stage were sampled for biochemical analysis. Tissue samples was
ground in known volume of 80% ethanol (v/v) in distilled water and
centrifuged at 4000 rpm for 10 minutes. The residue was washed thrice
and part of the combined supernatant used for the estimation of total
sugar, phenol and amino acids. The rest of the supernatant was kept in
a china dish and evaporated in a hot air owen at 60 °C and the residue
dissolved in distilled water, centrifuged and served as the source for
soluble sugar. The left over residue was ground in 30% perchloric acid
centrifuged, re- extracted and the combined supernatant is used for
starch estimation. Total soluble sugar was estimated using phenol
sulphuric acid method [10], total phenols by Swain and Hillis [11],
Protein content by Lowry et al. [12], starch by Mc Cready et al. [13],
amino acid by Sadasivam and Manickam [14].
Statistical analysis:
The data obtained on total soluble sugar and total protein were
analysed by linear regression analysis and the regression coefficient
were calculated and significance tested at 0.05% as per Sokal and
Rohlf [15]. Data for starch and phenols were anlysed by polynomial
regression at 3rd level and the trend was drawn and correlation
coefficient calculated and significance tested at 0.05 or 0.01% P as the
case may be.Results
Phenology and Seed development:
The observation on the phenological studies of H. ponga showed
that it is an evergreen tree growing in the lowland forest area to mid
elevation up to 900 msl. Healthy mature individuals of H. ponga
selected for phenological studies showed thatleaf shedding occurred
as a periodical event once after the maturation of old leaves.The tree
periodically renewed their leaves by mass leaf flushing in the month
of October to December. Mass flowering of H. ponga occurred during
the month of February and sometimes extended upto April. The
peak flowering of the tree was followed by mass seed set, that took
two to three months for its maturation. The majority of individuals
have developed their seeds in the month of April to June and the fruit
dispersal occurs with the help of wind. The mass seed set was followed
by mass seed germination through rainfall during June with the aid of
southwest monsoon.Hopea ponga produces axillary raceme/ panicle in every leaf axis
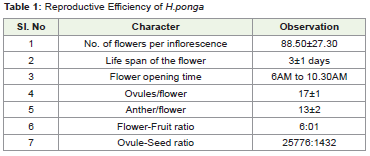
as well as in the nodes of side branches. The tree produced an average
of 12,536 ± 3106 flowers per tree during the peak flowering period.
The continuous observation on the matured floral buds confirmed
the diurnal flowering habit of the tree. Anthesis of flowers started
with the opening of floral parts in the morning hours between 6 AM
and 11 AM (Table 1). Flowers were pale pink to white in color with
a mild fragrance and the petals twisted at the tip with a pointed end.
The flowers were remained fresh for 3 days from the day of anthesis
and the petals were withered. This floral mechanism showed that
the flowers had a short lifespan of 3±1 days. Each flower consists of
13±2 epipetalous anthers.The unopened flowers observed for anther
dehiscence showed that the anthers were dehisced one day before
anthesis which in turn confirmed the protandrous nature of the
flowers. The stigma of the ovary was projected out to receive cross
pollen grains. The calculated flower fruit ratio and ovule seed ratio
were 6:1 and 25776:1432 respectively. Flowers are pale pink to white
in color with a mild fragrance and the petals twisted at the tip with
a pointed end. The flowers were remained fresh for 3±1 days from the day of anthesis and the petals were withered along with the
epipetalous anthers. The reproductive efficiency was calculated and
depicted in (Table 1).
On anthesis (identified with the mid fragrance) the flowers
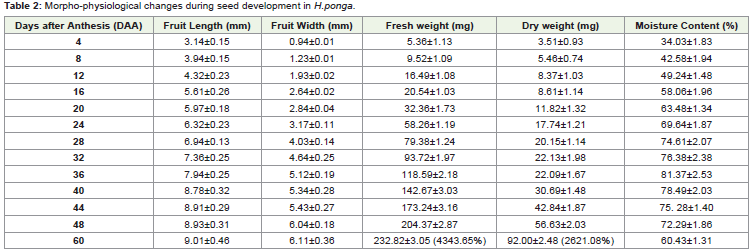
were tagged as 0 day (DAA) and the data on seed development was
carried out by measuring the length, width of the developing seeds
at an interval of 4 days starting from 4th day after anthesis (DAA) till
maturity at 60 days together with fresh and dry weight and % MC
(Table 2). Seed length and width increased from 3.14±0.15 mm and
0.94±0.01 mm at 4 DAA to 10.34±0.46mm and 7.11±0.36 at 60 DAA
respectively. Parallel with the increase in length and width there was a
linear and significant increase in Fresh Weight (FW) and Dry Weight
(DW) during seed development. FW has increased from 5.36±1.12
mg per seed at 4 DAA to 232.82 mg per seed at 60 DAA which is
about 4343.65%. Similarly the final DW increase was about 2621.08%
from the 4DAA. The initial %MC was 34.15% at 4DAA which slowly
increased and reached 81.37% at 36DAA and then declined slowly on
maturity, 60.43 ± 1.31 % at 60DAA (Table 2).
Biochemical changes:
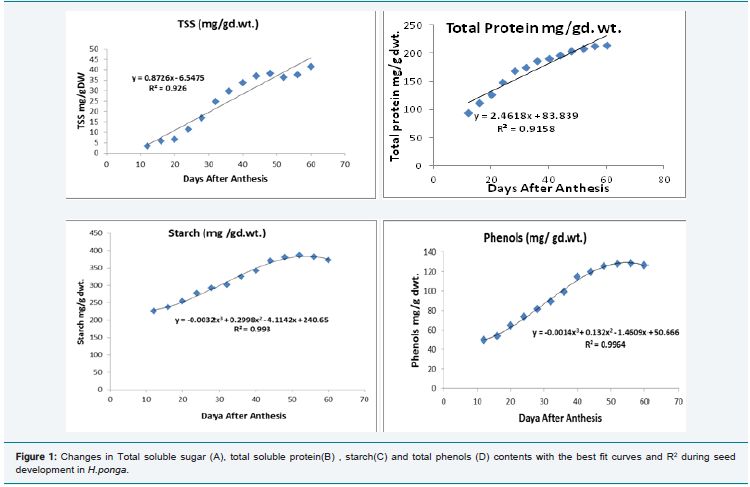
Because of the low amount of samples during the initial phases,
changes in Total Soluble Sugars (TSS), Total protein, starch and
phenol contents were evaluated from 12 DAA. Gradual increase in
TSS (Figure 1A), was recorded with the seed development in H.ponga.
TSS recorded 3.41±0.11 mg-1 g.dwt at 12 DAA to 41.33 ± 0.24 mg-1
gd.wt at 60 DAA, a 1212.02% increase and was significant at 0.05% P
level. Changes in total protein also sowed the same pattern as that of
TSS and the changes were significant at 0.05% P level (Figure 1B). O
the other hand, starch and phenol content recorded a linear increase
up to 40 DAA which furthers reduced and stable and later a slight
decrease was noted. The data were analysed using polynomial curve
as per the formula (y = -0.0032x3 + 0.2998x2 - 4.1142x + 240.65 R² =
0.993 for starch and y = -0.0014x3 + 0.132x2 - 1.4609x + 50.666, R² =
0.9964 for phenol ) and were significant in both cases at 0.05% P level
(Figure 1C and 1D).Discussion
Healthy mature individuals of H. ponga selected for phenological
studies from KSCSTE MBGIPS campus showed that leaf shedding occurred as a periodical event once after the maturation of old leaves.
However, leaf flushing was observed during the month of October to
December though the tree canopy appears to be evergreen throughout
the year. The majority of individuals that produced their floral buds
in the month of February were attained their peak flowering in the
month of March and April. After flowering it took two months for
the complete development and maturity of the seeds. The majority
of individuals have developed their seeds in the month of April to
June (Table 1). Accordingly that many dipterocarps species has the
phenomena of mass flowering and fruiting and the seeds mature
by the onset of South – East monsoon as the seeds are short lived
and sensitive to desiccation (1 & 16). Supporting to this the seeds of
H.ponga matured at 60 DAA with a high % moisture content of 60.43
± 1.31% indicate the recalcitrant nature and lose viability within short
period of 8 days.
Analysis of seed measurements started at 4 DAA in case of
seed length, width, fresh and dry weight and % moisture content as
the seeds were not large enough for analysis till 4DAA. Similarly,
biochemical and enzyme analysis started at 12 DAA before which
sufficient samples could not be obtained due low size and weight. Seed
length, width and fresh and dry weights followed the typical sigmoid
growth pattern of development and reaches maximum at maturity
(Table 2).
The deposition of storage substances is one of the key process of
zygotic embryogenesis providing compounds that will be used from
the early stages of the embryonic development until autotrophy,
after germination [17]. As sugars may be a major component as
biochemical precursors, an understanding on the changes in sugar
content during maturation is important. In the present study sugar
content tended to increase throughout the maturation and the
increase was significant at 1 % P level (Figure 1A). Sucrose synthesised
in green (photosynthetically active) tissues is transported through the
phloem to support growth and maturation of heterotrophic tissues
such as seeds [18]. Thus, seed filling depends above all on the rate
of photo assimilate supply and on metabolic regulation of transport
[19]. Starch normally considered a carbohydrate reserve in seeds
recorded accumulation during seed development in H. ponga till 50
DAA and later it stabilised and reduced at 60 DAA (Figure 1C). The
polynomial curve fitted was significant at 0.01%P level and showed a
sigmoid pattern. The possible role of starch reserve is to provide sugar source during germination. During seed development in H. ponga
total protein content increased linearly till 50DAA andlater the rate
was reduced but the changes were significant at 0.05% P level (Figure 1B). Phenol content recorded a typical sigmoidal pattern during
seed development in H.ponga. Weidnera et al. reported that both
phenolic acids and total phenolic compounds reach the highest levels
at the initial stage of development of cereal caryopses and decrease
considerably at the final stage of grain maturation [20]. Most higher
plants responded to various environmental stimuli by activating
secondary metabolic pathways such as the phenylpropanoid
metabolism. Phenylalanine ammonia-lyase (PAL, EC 4.3.1.5) is the
first and one of the key regulatory enzymes of this pathway [21].
Figure 1: Changes in Total soluble sugar (A), total soluble protein(B) , starch(C) and total phenols (D) contents with the best fit curves and R2 during seed
development in H.ponga.
The increase in DM (dry weight) during seed development is a
result of the synthesis and deposition of storage substances (Table 2).
In our work the total soluble sugars, proteins and starch constituted
4.13%, 21.51% and 37.32% respectively per gram dry weight of mature
seed. The accumulation of storage proteins in conifer somatic embryos
is influenced by ABA and water stress, by addition of an osmoticum
into the culture medium [22]. It has been suggested that the effects
of ABA and osmoticum are additive, where the synthesis of storage
protein is initiated by ABA and regulated at the post-transcriptional
level by the osmoticum [22].
Many of the physiological and biochemical effects brought about
in developing embryos by ABA can also be induced by low osmotic potentials [23]. The continuous increase in starch content and increase
in dry matter may result in the proportional reduction of osmotic
potential (though in the present work not evaluated). Probably, the
increase in starch content during the seed development was due to
triacylglycerols breakdown during seed development. Stone and
Gilford in P. taeda reported changes in starch accumulation [24],
during early seedling growth and role of triacylglycerols breakdown
and sub- sequent carbohydrate metabolism. It has been suggested that
endogenous ABA plays a role in the stimulation of specific storage
proteins, in the dehydration at the end of development and in the
prevention of precocious germination [25].
In conclusion, our results represent a snapshot of activities of
various morphological, physiological and biochemical changes during
the seed development of H. ponga an endangered and endemic tree
species from Western Ghats. This may useful for further studies on
accumulation patterns of different metabolites like individual sugars,
protein fractions and specific phenols and their role in the recalcitrant
nature of the seeds of H. ponga which may help in the conservation
and sustainable utilization of this tree species.
Acknowledgements
Authors acknowledge Kerala State Council for Science
Technology and Environment for the financial assistance as Emeritus
Fellowship to PNK and Director KSCSTE-MBGIPS for facilities and
encouragements.
References
Citation
Mithun V, Pradeep NS, Krishnan PN. Physiological and Biochemical Changes during Seed Development in Hopea ponga (Dennst.) Mabberley: An Endemic Endangered Tree Species of Western Ghats. In Lam. J Plant Sci Res. 2020;7(2): 193