Research Article
Antimicrobial Activities of Tamarind (Tamarindus indica) Extracts
Moncrieffe S*, Williams LAD, Reece S, Thompson S, Knight G, Robatham S and Beaufort C
College of Health Sciences, University of Technology, Jamaica, West Indies
Corresponding author: Moncrieffe S, College of Health Sciences, University of Technology, Jamaica, Kingston 6, Jamaica, West Indies, Tel no: 8763835643; Fax no: 8769271699; E-mail: smoncrieffe@gmail.com
Citation: Moncrieffe S, Williams LAD, Reece S, Thompson S, Knight G, et al. Antimicrobial Activities of Tamarind (Tamarindus indica) Extracts. J Plant Sci Res. 2019;6(1): 184.
Copyright: © Moncrieffe S, et al. 2019. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Plant Science & Research | ISSN: 2349-2805 | Volume: 6, Issue: 1
Submission: 06/03/2019; Accepted: 29/03/2019; Published: 04/04/2019
Abstract
It is well documented that the Tamarindus indica plant has antimicrobial properties. However these properties are not unilateral in terms of the spectrum of activity observed when comparing across geographical locations due to variations in the plant’s phytochemical content. In this article, a laboratory based experiment was conducted on four bacteria and a fungus in order to analyze the plants inhibitory characteristics at the University of Technology, Jamaica campus. These microorganisms included: Staphylococcus aureus, Streptococcus pyogenes, Pseudomonas aeruginosa, Escherichia coli and the fungus Candida albicans. A crude ethanolic extract was derived from the dried stem and leaves of the T. indica plant and it was hypothesized that this crude extract would yield larger zones of inhibition when compared to the fresh leaf juice of the plant. Using a modified agar well diffusion method, the zones of inhibition produced by the crude ethanolic extract and the fresh leave juice at a 100% concentration were compared using an independent t test. Varying concentrations of the crude ethanolic extract were also used to determine a minimum inhibitory concentration. The results demonstrated that the mean zone of inhibition when comparing across all microorganisms for the 100% w/v crude ethanolic extract was 15.75mm, while for the 100% w/v fresh leaf juice a mean zone of 9.33mm was observed. Using a 95% confidence interval a P value of 0.013 was derived, showing that the crude ethanolic extract produced a significantly higher zone of inhibition than the fresh leaf juice of the plant.
Introduction
Tamarindus indica commonly known as Tamarind tree belongs to the family Caesalpiniaceae. It has been used locally in Jamaica for several purposes. These include but not limited to anti-itch remedy in persons experiencing chicken pox, the culinary industry or just generally as a tea for a stomach ache. This symptom of itching is ameliorated by boiling the leaves of the tamarind plant and then the liquid is used as a bath. The resulting effect of such a procedure has been reported to cause symptomatic relief of itching. There have been previous investigations into the antimicrobial properties of tamarind from various regions globally, but none has been reported for Jamaica. These investigations have found both antibacterial and antifungal activity of extracts from the pulp of the plant [1-3]. Various regions have conducted this research of their locally based plant. These various researches where conducted under the premise that a different percentage of phytochemicals present due to nutrimental exposure of the plant during its cultivation. These phytochemicals were shown to be the basis for the antimicrobial effects of this plant.
A culmination of parameters such as Jamaica’s soil type and local accounts of the ameliorative effect of tamarind can therefore be used to initiate an experimental research to explore both the antibacterial and antifungal effects of this plant. With the objective of gathering more knowledge of the potential medicinal benefits of tamarind this could possibly provide evidence of Jamaican grown Tamarindus indica’s medicinal benefits while theoretically stimulating additional research for further analysis of the plant’s antiviral properties which would prove to be novel in its capacity.
Material and Methods
Preparation of T. indica extracts
The stems and leaves were washed with tap water, subsequently with distilled water and then allowed to air dry for five days. At the end of five days the dried plant was milled to produce tamarind powder. Tamarind powder of weight 201 gram was placed in a large conical flask to which 1.5L of 200 Proof Ethyl Alcohol was added. The conical flask was covered and left to stand for five days. At the end of five days the mixture was filtered using filter paper.
The solvent and extract were separated using a Rotary evaporator. Approximately 200ml was placed in the evaporating flask, which was immersed in the water bath of temperature 41 °C spinning at medium revolution for 45 minutes. The extract was poured into a 60ml measuring beaker. This process was repeated until all the filtrate was concentrated. The final volume of the extract was allowed to stand in open air for another five days to remove any additional 200 Proof Ethyl Alcohol. The final volume of the crude ethanol extract produced was packaged in a 100ml amber bottle.
Fresh T. indica leaves were washed with five percent chlorine bleach on the day of the laboratory based experiment. The leaves were subsequently rinsed with distilled water and using a mortar and pestle were crushed to extract the fresh leaf juice. Sterile gauze was used to squeeze out the leaf extract into sterile four millilitre (4ml) glass bottles.
Preparation of Agar Plates
Approximately five hundred millilitres (500ml) of nutrient agar was prepared based on manufacturer specifications along with one hundred millilitres (100ml) of Sabouraud’s dextrose agar. The prepared agars were then autoclaved at fifteen pounds per square inch (15psi) at 120 °C with a timer set for 15 minutes. At the end of the time when the temperature of 90 °C was attained and the pressure fell to zero (0) psi, the agars were then removed. Approximately 20ml of nutrient and Sabouraud’s dextrose agar were poured aseptically into separate Petri dishes respectively and allowed to set.
Determining the zones of inhibitions
The antibacterial and antifungal properties of the extracts of T. indica were determined using the agar- well diffusion method. Subcultures of the bacterial and fungal strains were prepared and stored in an incubator at 37 °C for 24 to 48 hour period. The bacterial isolates were standardized to a 0.5 McFarland standard. This was done using test tubes of nutrient broth inoculated with each bacterium separately. The inoculated test tubes were allowed to grow over a 24 hour period, examined using a spectrometer and then adjusted to a McFarland standard of 0.5 using more nutrient broth when necessary.
The agar plates that were filled with 20ml of nutrient agar were firstly demarcated with markers into four separate partitions. The four partitions present included ethanol extract at 50%, fresh leaf extract 100%, amoxicillin and clavulanate 20/10mcg (AMC - 30) as the positive standard and distilled water as the negative standard. Separate nutrient agar plates were demarcated into three partitions with each partition having the ethanol extract at 100% concentration. All the plates prepared for each bacterium was done in triplicates in order to produce a sample for analysis. This process was repeated for the fungal strain of C. albicans.
The standardized colonies of bacterium were used to inoculate the agar plates using the spread plate method, which was also done for the fungal strain. Agar wells were then formed aseptically with a diameter of 8mm for all the demarcated zones except that of which was formed for the AMC - 30 disks. The diameter for the AMC - 30 disks were measured and found to be 6mm.
The nutrient and Sabouraud’s dextrose agar inoculated plates were then incubated at 37 °C. These plates were observed at both 24 and 48 hour intervals. The observed zones of inhibitions were measured and tabulated for statistical analysis.
Determining of minimum inhibitory concentration
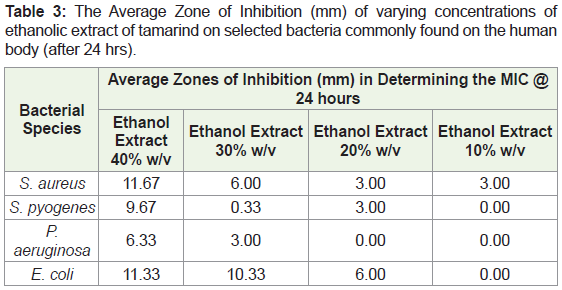
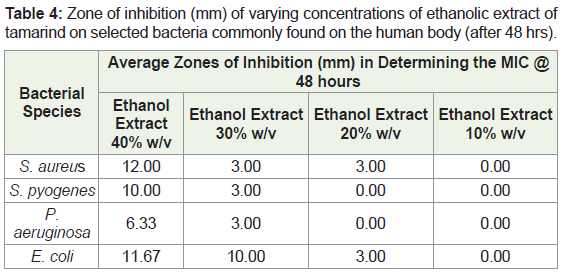
Extrapolating from the results produced for ethanol extract at a concentration of 50%, 40%, 30%, 20% and 10% were produced for analysis of the MIC value. The descending dilutions were produced using the crude extract and distilled water measured using a micropipette. The least concentrated dilution was used as the MIC for each antimicrobial strain under study.
Data analysis
The analysis for each extract was done in triplicates. Using SPSS, an independent T test was done to demonstrate any significant differences between the crude extract and the fresh juice mechanically extracted from the T. indica leaves.
Results
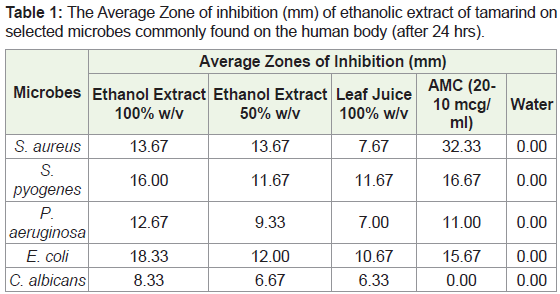
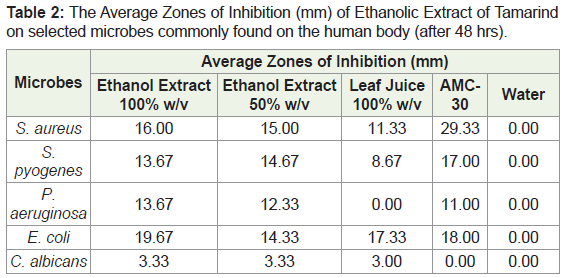
The antimicrobial activity of the tamarind extract was determined by using the agar well diffusion method. The zones of inhibition were measured at 24 hours and 48 hours after incubation seen in Table 1 and Table 2. The ethanol extract was further tested at lower concentrations to ascertain its Minimum Inhibitory Concentration (MIC). The MIC is the lowest concentration of the extract that will inhibit the visible growth of the bacteria after overnight incubation. Significant differences between means were determined by using an Independent t test mode of analysis. P values < 0.05 were regarded as significant. Software employed was SPSS 20. The results of the experiment and data analysis are represented in the following figures and tables.
After 24 hours (Table 1), the amoxicillin showed the greatest zone of inhibition for S. aureus with an average diameter of 32.33mm. Both concentrations of the ethanol extract produced average diameters of 13.67mm. The leaf juice had the smallest zone of inhibition - 7.67mm. At the end of 48 hours, there were increases in the zones of inhibition for the ethanol extracts and leaf juice. Table 1 show that the zone of inhibition of the AMC-30, however, reduced by an average 3 mm. It can be noted that both concentrations of ethanol extract and AMC-30 showed more activity for S. pyogenes than the 100% w/v leaf juice, which had an average zone of inhibition of just 8.67 mm after 48 hours. At the end of 48 hours, the 100% w/v ethanol extract had the largest diameter (zone of inhibition) for P. aeruginosa and E. coli. This suggests that the ethanol extract has more antibacterial activity against E. coli than the AMC-30 standard.
Incorporating the results demonstrated in Table 2, an individual analysis for zones of inhibition for each microorganism clearly shows a disparity in the results produced. When looking at S. aureus, the AMC-30 results showed the highest mean zone of inhibition (29.33mm) followed by the 100% ethanol extract (16.00mm) and then the fresh leaf juice (11.33mm). The disparity between the 100% ethanol extract and the 100% fresh leaf juice showed a mean zone of inhibition at 16.00mm and 11.33mm respectively (P = 0.33). When looking at S. pyogenes, the zone of inhibition between AMC-30 and 100% ethanol extract did not show an exponential difference when comparing the means. The AMC-30 had a mean zone of inhibition at 17.00mm and 100% ethanol extract showed a mean zone at 13.67mm (P = 0.36). This thereby demonstrates that in this instance there wasn’t a statistically significant difference between the mean zones of inhibition produced by AMC-30 and the 100% concentration of ethanol extract. The 100% ethanol extract had a mean zone of 13.67mm, showing to be higher than 100% leaf juice at 8.67 (P = 0.33). Interestingly, for P. aeruginosa there was a higher mean zone of inhibition when exposed to 100% ethanol extract when compared to AMC-30 (P = 0.67). The mean zone was shown to be 11.00mm for AMC-30 while it was 13.67mm for the 100% ethanol extract. This was similar to the results seen for E. coli in terms of a higher mean zone of inhibition seen the 100% ethanol extract when compared to the AMC-30 standard (P = 0.067).
In assessing the MIC, after 24 hours the 40% ethanol extract showed the greatest zone of inhibition for the four bacteria (Table 3). Observations after 48 hours revealed that ethanol extract at 10% w/v showed no zones of inhibition with the concentrations of 30% w/v and 20% w/v showing some activity. After 48 hours, the greatest diameter for the zone of inhibition for the bacteria was still seen in the 40% w/v ethanol extract. The ethanol extract at 30% w/v showed activity against E. coli with a diameter of 10.00mm. The 20% w/v and 10% w/v ethanol extract showed little to no activity against all bacteria after 48 hours (Table 4).
Data analysis results
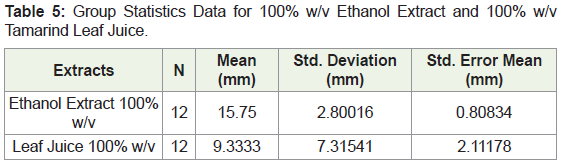
In order to fully analyse the spectrum of activity, all the zones of inhibition for each sample under investigation were collated irrespective of the microorganisms used and an independent T test was employed to examine each compound at its specified concentration. Table 5 presented below demonstrates a comparing between the mean zone of inhibition for all ethanol extract at 100% (w/v) and fresh leaf juice also at a 100% (w/v) concentration. There is a clear difference between the mean zones produced which the ethanol extract having a higher mean zone.
The homogeneity of variances was tested using Levene’s F test where a p value of 0.001 was obtained. The 100% ethanol extract (N = 12) was associated with mean zone of inhibition of 15.7500 (SD = 2.80016) and the 100% w/v leaf juice (N = 12) was associated with a Mean of 9.3333 (SD = 7.31541). To determine if a significant difference between the means exists between the 100% (w/v) ethanol extract and 100% w/v leaf juice, the p value for the sample t test was interpreted which was found to be 0.01.
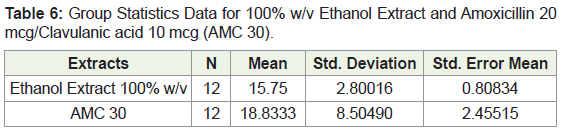
In examining the data found on the comparison between the 100% ethanol extract and AMC-30 it was seen where that the Levene’s test revealed a p value of 0.66 which shows that variability between the means of the 100% (w/v) ethanolic extract and AMC-30 are not the same (Table 6). The p-value obtained for the independent sample t Test which compared the means of the 100% (w/v) ethanolic extract and the AMC-30 was 0.254 indicating that there is no significant difference between the means.
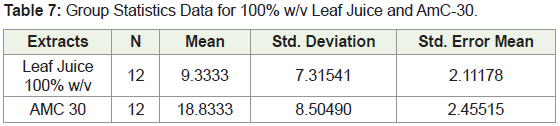
In comparing the mean zones of inhibition for the 100% leaf juice and the AmC-30 data depicted in Table 7 below, a p value of 0.785 was obtained for the Levene’s test for equality of variances. This showed that variability between the means are the same. The independent samples t-test was performed and a p value of 0.008 was obtained indicating that there is a significant difference between the two means.
Discussion
To assess the antimicrobial activity of the aqueous-ethanolic tamarind extract, a total of four bacterial species (two Gram-positive and two gram-negative) and one fungal species were used.
The diameters of the zone of inhibition were interpreted as follows: <9 mm, inactive; 9-12mm, partially active; 13-18mm, active; >18mm, very active [3]. After 24 hours the ethanolic extract of concentration 100% exhibited activity against both Gram-positive and Gram-negative bacteria which was similarly observed in another study [4]. The ethanolic extract showed activity against E. coli with a diameter of 19.67 mm and was active against P. aeruginosa with a diameter of 13.67 mm. However in one study [3], the opposite was observed where the best activity was produced against P. aeruginosa with a diameter of 16mm and the least activity against E. coli with a diameter of 13mm. The activity against Gram-positive bacteria was higher in S. pyogenes with a diameter of 16mm after 24 hours. However, after 48 hours this decreased to 13.67mm. The diameter of the zone of inhibition for S. aureus after 48 hours was 16mm. The 100% ethanol extract was inactive against the Candida sp. with a diameter <9mm; this finding is contrary to results obtained in another study, which showed that ethanol extracts produced high antifungal activity against C. albicans [5].
The 50% extract was also effective against the selected Grampositive and Gram-negative bacteria. The diameters of the zone of inhibitions were greater for the Gram-positive bacteria than the Gram-negative. This difference in susceptibility may be due to the difference in cell wall structure between Gram-negative and Grampositive bacteria. Gram-positive bacteria have peptidoglycan and no lipopolysaccharide in their cell structure; peptidoglycan shows less resistance regarding the permeability of the cell wall to various agents. On the other hand, Gram-negative bacteria have a lipopolysaccharide layer present on the layer outside the peptidoglycan. This lipopolysaccharide layer increases resistance and prevents various agents from crossing the cell wall easily. The 50% (w/v) extract was also inactive against C. albicans with a diameter <9mm.
The 100% leaf juice showed its highest activity against E. coli producing a diameter of 17.33mm. This concentration was inactive against P. aeruginosa as it produced a diameter of <9mm. For the Gram-positive bacteria, the leaf juice was more effective against S. pyogenes after 24 hours producing a diameter of 11.67mm. However, at 48 hours this decreased to 8.67mm with the higher activity now seen against S. aureus with a diameter of zone of inhibition of 11.33mm. The 100% Leaf juice was found to be inactive against C. albicans producing a diameter <9mm.
The 100% ethanolic extract and the 100% leaf juice showed a similar trend in their activity. Both showed the highest activity against E. coli and the second highest activity against S. aureus. The diameter of the zone of inhibition for S. pyogenes decreased after 24hours for both the 100% ethanolic extract (16mm to 13.67mm) and the 100% Leaf Juice (11.67mm to 8.67mm). While the 100% ethanolic extract was active against P. aeruginosa, the 100% leaf juice was inactive against this bacterial species. The diameter for the zones of inhibition was greater for the 100% (w/v) ethanolic extract than the 100% (w/v) leaf juice. Reports state that ethanolic extracts showed higher antimicrobial activity than water extracts [6]. Both were found to be inactive against C. albicans.
In determining the Minimum Inhibitory Concentration (MIC), the concentration that had the highest spectrum of activity was the 40% w/v ethanolic extract. This concentration produced a diameter with a range of 10-12mm in three of the bacterial species with P. aeruginosa being the exception with a diameter <9mm (6.33mm). The 30% w/v of the ethanolic extract was the only other concentration to produce a diameter of zone of inhibition >9mm. It was only effective against E. coli producing a diameter 10mm. The fungal species C. albicans was omitted from the MIC experiment as the 50% w/v ethanolic extract was not effective to produce a diameter >9mm. This suggested that the extract was inactive against the fungus and therefore would not show activity at lower concentrations.
From these studies, it was observed that tamarind is effective against both Gram-positive and gram-negative bacteria. The ethanolic extract showed the higher spectrum of activity compared to the leaf juice. For Gram-positive bacteria activity was higher in S. aureus followed by S. pyogenes and for gram-negative bacteria, activity was higher in E. coli followed by P. aeruginosa. These findings are in agreement with two studies [5,6]. The ethanol extract may have a higher solubility for the phytochemicals present [5]. This would increase their concentration and thus the antimicrobial effect of the extract.
The juice of the Tamarind leaves showed activity against three bacteria S. aureus, S. pyogenes and E. coli. The diameter produced for P. aeruginosa was <9m which then decreased to 0.00mm. The diameter produced for S. pyogenes also decreased, a trend also seen in the 100% w/v ethanolic extract. The decrease in diameter of zone of inhibition could be as a result of the cessation of the extracts inhibiting abilities which allowed the bacteria to grow. Also, P. aeruginosa is a bacterium known for possessing multiple mechanisms for resistance. The bacteria could have become resistant by lowering its membrane permeability or by other mechanisms to overcome the inhibitory effects of the extract. According to Escalona-Arranz JC, et al. [1], the leaves of the tamarind plant found in Cuba, showed limited antimicrobial activities due to the climatic, ground and growing conditions resulting in a poor yield of essential oils. Ahmad N stated that Jamaica’s soil is rich in limestone which could provide contrasting nutrimental supply [7]. However, the results presented were not contrary to the findings in Cuba.
The 100% w/v ethanolic extract showed higher activity against Gram-negative bacteria than AMC 30. Amoxicillin has a moderate spectrum of activity being effective against a wide range of Grampositive bacteria and a limited range of gram-negative bacteria.
In this experiment the extracts activity against the Pseudomonas sp. produced comparable results to AMC 30; it was also active against S. aureus, S. Pyogenes and E. coli. These bacteria can result in serious infections with some strains being more resistant to multiple synthetic agents than others. This can negatively impact the health care system and result in increased treatment failure. We recommend phytochemical testing to examine the constituents responsible for the antibacterial effects. These constituents could be formulated in a suitable dosage form which can be used as a safer, cost-effective firstline antibacterial treatment.
While the plants phytochemicals are responsible for its desirous antimicrobial properties, there are phytochemicals that maybe toxic to humans. We recommend that toxicological investigations and purifications methods be carried out to create a product that is both safe and effective.
Conclusion
The foregoing study suggests that Tamarindus indica cultured in Jamaica exhibits a moderate range of antimicrobial activity similar to Amoxicillin. Further research on the phytochemicals responsible for this activity can result in formulation of an antibacterial agent. The plant does not show antifungal activity against C. albicans. However, tests should be conducted on other fungal species. Based on the statistical analysis conducted it was found that the crude extract of the stems and leaves of the T. indica plants exhibit antimicrobial activity and produces a larger mean for the zones of inhibition when compared to the fresh leaf juice of the plant.
References
- Escalona-Arranz JC, Peres-Roses R, Urdaneta-Laffita I, Camacho-Pozo MI, Rodriguez- Amado J, et al. (2010) Antimicrobial activity of extracts from Tamarindus indica L. leaves. Pharmacogn Mag 6: 242-247.
- Abdel Gadir WS, Mohamed F, Bakheit AO (2007) Antibacterial activity of Tamarindus indica fruit and Piper nigrum seed. Res J Microbiol 2: 824-830.
- Gupta C, Prakash D, Gupta S (2014) Studies on the antimicrobial activity of Tamarind (Tamarindus indica) and its potential as food bio-preservative. Int Food Res J 21: 2437-2441.
- Nwodo UU, Obiiyeke GE, Chigor VN, Okoh AI (2011) Assessment of Tamarindus indica extracts for antibacterial activity. Int J Mol Sci 12: 6385-6596.
- Gumgumjee NM, Khedr A, Hajar AS (2012) Antimicrobial activities and chemical properties of Tamarindus indica L. leaves extract. Afr J Microbiol Res 6: 6172-6181.
- Daniyan SY, Muhammad HB (2008) Evaluation of Antimicrobial activities and phytochemical properties of extracts of Tamarindus indica against some diseases causing bacteria. Afr J Biotechnol 7: 2451-2453.
- Armand N (2009) Tropical soils with focus on West Indian Soils. The University of the West Indies, St. Augustine, Trinidad & Tobago.