Research article
Relationships among Anatomical Characteristics of Nine Japanese Angiosperm Trees without G-Fiber in Reaction Wood
Sultana RS*
Department of Botany, University of Rajshahi, Rajshahi 6205, Bangladesh
Corresponding author: Sultana RS, Department of Botany, University of Rajshahi, Rajshahi 6205, Bangladesh, E-mail: sultanaru@yahoo.com
Citation: Sultana RS. Relationships among Anatomical Characteristics of Nine Japanese Angiosperm Trees without G-Fiber in Reaction Wood. J PlantSci Res. 2018;5(1): 174.
Copyright © Sultana RS 2018. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Plant Science & Research | ISSN: 2349-2805 | Volume: 5, Issue: 1
Submission: 15/12/2017; Accepted: 02/01/2018; Published: 05/01/2018
Abstract
The relationships among the anatomical features in reaction wood of nine Japanese hardwood species without G-fiber were studied. Leaning branches of nine naturally growing species were examined: Euscaphis japonica, Viburnum dilatatum, Rhododendron wadanum, Enkianthus subsessilis, Euonymus alatus, Ilex macropoda, Ilex crenata, Clerodendrum trichotomum and Daphne odora. As complementary characteristics of the previous report in these nine species, the changes in vessel diameter, vessel length, fiber diameter, cell wall thickness of fiber, fiber length, and Microfibril Angle (MFA) of the S2 layer in wood fiber were examined in reaction wood in comparison with opposite wood. Depending on the species during reaction wood formation, the vessel and fiber characteristics in reaction wood were significantly altered. A significant negative correlation (0.635, p ≤ 0.05) was observed between eccentric growth and the rate of increase in the fiber wall thickness. On the other hand, the decrease rate (DR) of the lignin content was significantly correlated to the DR of the vessel number (0.869, p ≤ 0.01).
Keywords:
G-fiber; Hardwood species; Lignin content; Microfibril angle; Opposite wood; Reaction wood
Introduction
Reaction wood is a modified wood with special anatomical and chemical properties which is formed on leaning stems and branches to maintain the vertical orientation of the main stem or the equilibrium position of branches in order to receive sufficient light or in response to a strong dominant wind. This modified wood is considered to be formed by the cambium reaction to a gravitational stimulus [1-4]. In hardwood species, reaction wood is generally formed on the upper side of a leaning stem and branch and is called tension wood [1]. The existence of a gelatinous layer (G-layer) is an important characteristic of tension wood, which has a greater increase of cellulose content and reduction in the Microfibril Angle (MFA) and lignin content than those in normal wood. In some species, possibly those belonging to the primitive angiosperms, the G-layer is not always formed; however, the MFA and lignin content of the S2 layer are smaller in reaction wood [1,2,5]. In the nine species studied here, we previously reported that a secondary wall of reaction wood fiber had three layers (S1+S2+S3) and the G-layer was absent [6]. As a common feature of reaction wood anatomy, all of these species showed greater growth increments on the upper side of leaning branches and a reduced lignin content in the reaction wood. However, some other relevant characteristics of reaction wood in these nine species are not investigated.
Several reports have been published on the relationships of the characteristics of the tension wood of many angiosperms. Most of the relationships were established on the growth stress and the anatomical and chemical characteristics of reaction wood. Okuyama, et al. reported that the increased longitudinal tensile stress resulted in a decreasing MFA and lignin content and increase in α-cellulose content and cellulose crystallinity in reaction wood of Liriodendron tulipifera without a typical G-fiber [7]. The tensile released strain increased with a decreasing MFA and Klason-lignin content and with an increasing α-cellulose content and cellulose crystallinity in reaction wood of Liriodendron tulipifera with G-layer-like fiber [8]. Fang, et al. revealed the relationships between the growth indicator of trees and anatomical properties in the reaction wood characteristics of Populus 14551 with G-fiber [9]. They observed positive correlations between the growth indicator and a) fiber diameter, b) double cell wall thickness of fiber, and c) fiber length. Although the relationships among the physical properties as well as the chemical properties of reaction wood have been extensively investigated, reports dealing with the relationships among the anatomical characteristics of reaction wood are limited. Moreover, the relationships of the reaction wood characteristics with either the presence or absence of a G-layer have been investigated in many angiosperm trees, but no relationships have been identified in the reaction wood characteristics of angiosperm trees that lack G-fiber but have an S3 layer in the secondary wall of reaction wood fiber.
In a previous study, Sultana, et al. examined some anatomical features in the reaction wood of nine Japanese hardwood species that do not G-layer but have an S3 layer in the secondary wall of reaction wood fiber [6]. In the present study, some unexplored complementary reaction wood features of these species were characterized, and the relationships among the reaction wood characteristics were established.
Materials and Methods
The leaning branches of nine naturally growing Japanese hardwood species, namely, seven diffuse porous species, Euscaphis japonica, Viburnum dilatatum, Rhododendron wadanum, Enkianthus subsessilis, Euonymus alatus, Ilex macropoda, and Ilex crenata, one ring porous species, Clerodendrum trichotomum, and one figured porous species, Daphne odora, were collected from the Utsunomiya University Experimental Forest, Funyu, Tochigi, Japan. The processes of sample collection and transverse section preparation were described in our previous report [6].
For the determination of vessel diameter, fiber diameter, and fiber wall thickness, microphotographs were taken randomly from safranine-stained transverse sections of reaction and opposite woods, and measurements were obtained from printed microphotographs.
As the vessel diameters were being measured, the radial and tangential distances of the vessel lumen were obtained with a digital caliper (Mitutoyo, CD-15CP). The average value of the radial and tangential distances of a vessel lumen indicates each vessel diameter. In each species, the vessel diameter was recorded from an average value of 30 vessels. In case of Clerodendrum trichotomum (ring porous), the vessel diameter was measured randomly from latewood vessels except for the pore zone in reaction and opposite woods.
The fiber lumen diameter was also measured using the above procedure. The wall thickness of wood fiber was obtained from one half of the double cell wall distance. Data of fiber diameter and fiber wall thickness were recorded from 50 fibers.
For measuring the length of vessel element and wood fiber, small blocks (1 × 1 × 10 mm) were macerated with Schultz’s solution. Macerated samples were mounted on glass slides with a 75% glycerin solution and then projected with a profile projector (Nikon, V-12). The length of 50 vessel elements and 100 fibers was measured using a digital caliper, and the average values of vessel element and wood fiber length were recorded. In case of Clerodendrum trichotomum (ring porous), wood blocks were prepared from latewood of reaction and opposite woods, and average measurements were recorded.
For measuring the MFA of the S2 layer in wood fibers, radial sections (20 µm in thickness) were prepared from reaction and opposite woods using a sliding microtome (Yamato, ROM380, Japan). The MFAs of the S2 layer of wood fibers were measured with the iodine method using a system microscope (Nikon, ECLIPSE E600, Japan), and microphotographs were then taken with a digital camera [10]. Measurements of the MFA for each species were obtained from 30 different fiber walls in microphotographs with image analysis software (Image J, National Institute of Health).
Data of DR of lignin content reported previously are used here to examine the relationships among the anatomical characteristics of reaction wood [6].
Significance tests between the characteristics of reaction and opposite woods with respect to the vessel diameter and length, fiber diameter, wall thickness and length, and MFA were performed by the student’s test (t-test). The relationships were determined among the reaction wood characteristics of nine species with the Pearson correlation coefficient test.
Results and Discussion
Changes of vessel diameter in reaction wood
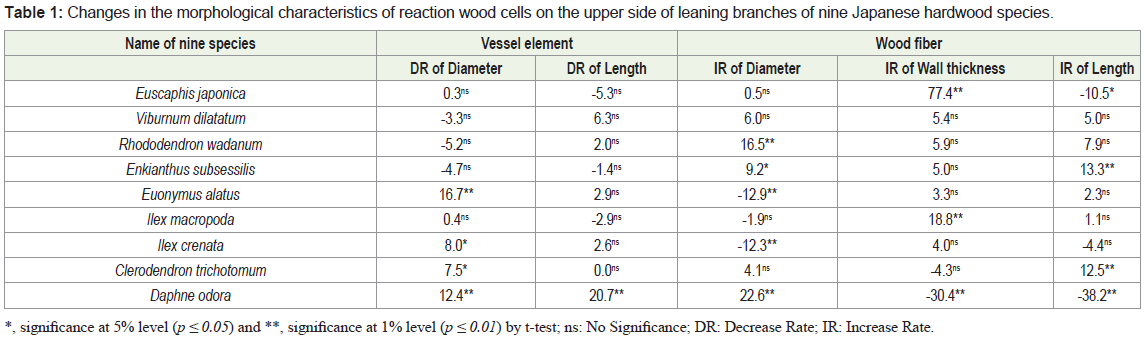
The vessel diameter decreased significantly more in reaction wood of Euonymus alatus (p ≤ 0.01), Ilex crenata (p ≤ 0.05), Clerodendron trichotomum (p ≤ 0.05), and Daphne odora (p ≤ 0.01) than it did in opposite wood among the nine species tested. The highest decrease rate (DR) (16.7%) was observed in Euonymus alatus. Almost no significant differences in the vessel diameter were observed between reaction and opposite woods in the other species studied (Table 1). In general, the diameter of a vessel decreases with the formation of reaction wood in many angiosperms [5,11]. Our previous report revealed that the vessel number decreased in all nine species; therefore, the pore distribution in the transverse section of reaction wood of Daphne odora (figured porous) was discontinuous and had oblique to dentritic vessel arrangement [6]. In the present experiment, the diameter of the vessel in Daphne odora also decreased significantly and was accompanied by reaction wood formation (Figure 1). These observations indicate that discontinuous vessel arrangement in reaction wood of Daphne odora might be caused by the reduction of the vessel diameter as well as the vessel number. A similar trend was reported by Hiraiwa, et al. in reaction wood of Osmanthus fragrans (figured porous) [11]. The decrease of the vessel diameter and number in reaction wood is a general trend of the cambial response to a gravi-stimulus. Various DRs of vessel diameter in reaction wood were observed in nine species, irrespectively of differences in the pore distribution pattern.
*, significance at 5% level (p ≤ 0.05) and **, significance at 1% level (p ≤ 0.01) by t-test; ns: No Significance; DR: Decrease Rate; IR: Increase Rate.
Changes of fiber diameter in reaction wood
With the formation of reaction wood, the fiber diameter significantly increased in Rhododendron wadanum, Daphne odora, and Enkianthus subsessilis and decreased significantly in Euonymus alatus and Ilex crenata. The highest Increase Rate (IR) and DR were observed in Daphne odora (22.6%) and Euonymus alatus (12.9%), respectively (Table 1). In the four other species, Euscaphis japonica, Viburnum dilatatum, Ilex macropoda, and Clerodendrum trichotomum, no significant differences in the fiber diameter were identified between reaction and opposite woods. Chow reported that tension wood fibers were narrower than those of normal wood in beech [12]. However, Onaka pointed out a larger tangential diameter of tension wood fiber in some species [1]. Almost no differences were found in the diameter of the fiber tracheid among the upper, lateral, and lower sides of inclined trunks in two Magnolia species [5]. The present experiment suggested that variation in the change of wood fiber diameter depends on the species because the unique nature of the trend was not observed in the fiber diameter changes in the reaction wood of all nine species studied here.
Changes of fiber wall thickness in reaction wood
The thickness of the fiber wall significantly (p ≤ 0.01) increased in reaction wood of Euscaphis japonica and Ilex macropoda, while it significantly (p ≤ 0.01) decreased only in Daphne odora with the formation of reaction wood. The highest IR and DR were observed in Euscaphis japonica (77.4%) and Daphne odora (30.4%), respectively. In other species, significant changes in the thickness of fiber wall were not observed in reaction wood (Table 1). Conspicuously thickwalled fiber in reaction wood with G-fiber was found in arborescent angiosperms [13]; in contrast, thick-walled fiber was observed in normal wood of Fagus sylvatica when reaction wood fibers contained a G-layer [12]. On the other hand, almost no differences in the thickness of fiber tracheids without a G-layer were found among the upper, lateral, and lower sides of two Magnolia species [5]. In the present study, the fiber wall thickness of all nine species without a G-layer varied from species to species. Based on the observation of the present investigation, it is suggested that changes of fiber wall thickness are not always associated with either the presence or absence of G-layer in reaction wood fiber.
Changes of length of vessel element and wood fiber in reaction wood
The length of vessel element significantly decreased with 20.7% only in reaction wood of Daphne odora among nine species examined (Table 1). The contrast observation was reported in Populus euramericana by Jourez, et al. [14], who observed that the vessel elements were longer in tension wood than in opposite wood.
The length of wood fiber significantly increased in Enkianthus subsessilis and Clerodendrum trichotomum with the formation of reaction wood, the highest increased rate (13.3%) being found in Enkianthus subsessilis. On the other hand, the length of wood fiber decreased significantly in Euscaphis japonica and Daphne odora, and the highest DR of 38.2% was observed in Daphne odora. No significant differences in the fiber length between reaction and opposite woods were observed in the other five species (Table 1). In a comparison of normal wood fiber length, tension wood fibers were sometimes reported to be longer, sometimes shorter, and sometimes with no marked difference [14-16]. On the basis of an experiment on fiber length in tension wood of some Eucalyptus species, Wardrop reported that eccentric growth is generated with more formation of shorter length wood fibers [17]. In a previous report, we demonstrated that the growth increment was the highest in Daphne odora [6]; on the other hand, the shortest wood fiber formation in reaction wood of this species was observed in the present experiment. Therefore, our observation in the case of Daphne odora is in agreement with the findings of Wardrop [17].
Changes of microfibril angle of S2 layer in reaction wood fiber
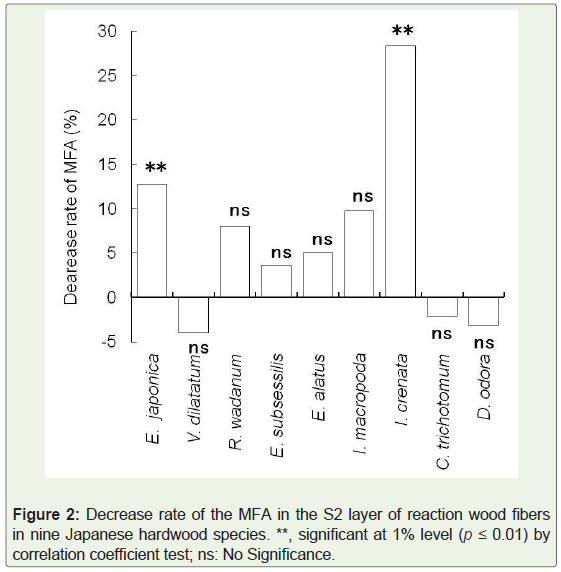
In a previous study, we demonstrated that the microfibril arrangement of the S3 layer in both reaction and opposite wood fibers was a flat S-helix [6]. In the present study, we examined the changes of the MFA in the S2 layer of wood fibers owing to reaction wood formation. The MFA of the S2 layer in wood fibers significantly (p ≤ 0.01) decreased in Euscaphis japonica and Ilex crenata. The highest DR (28.3%) was observed in Ilex crenata. In other species, almost no significant differences in the MFA change of S2 layer were observed between reaction and opposite wood fibers (Figure 2).
It has been reported that the microfibrils of a G-layer oriented parallel or nearly parallel to the longitudinal axis of a cell in many angiosperm trees [18]. The tendency of decreasing MFA has also been observed in the S2 layer of reaction wood where G-layer is absent. This trend is in agreement with the observation of Yoshizawa, et al. in some primitive angiosperm trees lacking either G-layer or S3 layer in the secondary wall of reaction wood fiber [5]. This tendency showing the decrease of MFA in the S2 layer of reaction wood fibers corresponds to a phenomenon in which tensile stress at longitudinal direction increases in reaction wood that maintains or recovers the position of the inclined stems or branches to appropriate orientation [7]. The present results confirmed that the MFA of the S2 layer in wood fibers might not always decrease due to reaction wood formation in some species having an S3 layer but lacking a G-layer in reaction wood fibers. Therefore, the mechanisms are unknown for these species to maintain an appropriate position for a leaning stem or branch. Therefore, a more detailed investigation is needed to clarify the changes in the MFA in the S2 layer in comparison to those in the other species with a three-layered secondary wall in wood fiber without a G-layer.
Relationship among reaction wood characteristics
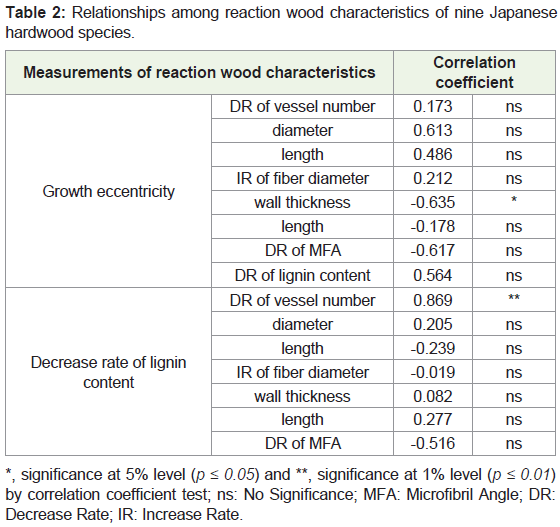
In the nine species studied here, we previously examined growth eccentricity, change in vessel number, and lignin content by the formation of reaction wood [6]. In the present study, we investigated changes in other complementary reaction wood characteristics, such as vessel diameter and length, fiber diameter, wall thickness and length, and MFA in the S2 layer of wood fiber. Using all these reaction wood characteristics of nine species, relationships were established in a) growth eccentricity and other reaction characteristics and b) the decrease rate of lignin and other reaction characteristics (Table 2).
*, significance at 5% level (p ≤ 0.05) and **, significance at 1% level (p ≤ 0.01) by correlation coefficient test; ns: No Significance; MFA: Microfibril Angle; DR: Decrease Rate; IR: Increase Rate.
Relationships between growth eccentricity and other reaction characteristics: A significant negative (r = -0.635, significant at 5% level) relationship was found between eccentric growth and IR of fiber wall thickness (Table 2). There are some reports that indicated a relationship between fiber wall thickness and growth stress. Fang, et al. revealed that the correlation between double cell wall thickness of fiber and growth stress was negative, which has also been confirmed by another study [8]. Although several studies pointed out the relationship between fiber cell wall thickness and growth stress, the present result revealed a negative correlation between growth eccentricity and cell wall thickness of wood fiber.
Relatively high positive correlations were found between the growth eccentricity and DR of the vessel diameter (r = 0.613) and length (r = 0.486). Correlations between growth eccentricity and fiber diameter and fiber length have been observed [14,17], but not in the nine species without G-fiber in their reaction wood, which showed relatively high relationships between the growth eccentricity and DR of the vessel diameter and length.
A positive but not significant correlation was also observed between the growth eccentricity and DR of lignin content (r = 0.564). This fact is a common phenomenon in the reaction wood anatomy of many angiosperms. For example, Yoshizawa, et al. reported that tension wood showed a relatively low lignin content when a growth increment was observed on the upper side of a leaning stem or branch of Magnolia obovata and M. kobus [5].
On the other hand, as shown in Table 2, a high negative correlation was observed between the growth eccentricity and DR of MFA (r = -0.617), although it was not significant. This result was opposite to a report by Yoshizawa, et al. [5], who speculated that eccentric growth occurred on a leaning stem of Magnolia species having neither an S3 layer nor a G-layer in their reaction wood fiber but MFA decreases in the S2 layer of reaction wood fiber, which means that the relationship was positive between eccentric growth and DR of MFA in the S2 layer. In the present study, this opposite trend of reaction wood characteristics is considered to occur with the presence of the S3 layer in reaction wood fiber.
Relationships between DR of lignin content and other reaction characteristics: A significant positive correlation coefficient (0.869, p ≤ 0.01) was observed between the DR of lignin content and that of vessel number (Table 2). In previous studies, there was a common phenomenon in which tension wood contained a lower proportion of vessels than normal wood [5]. The present study suggests that the decrease in the number of vessels due to reaction wood formation is a reason for the decreasing lignin content. Xu, et al. reported that the vessel wall and middle lamella are more highly lignified than the secondary wall of wood fiber; therefore, it is possible that the decrease in the number of vessels resulted in the decreased lignin content in reaction wood [18].
In the present study, a relatively high negative correlation coefficient was recognized between the DR of lignin content and DR of MFA (r = -0.516, not significant). The opposite result was observed in the relationship between the DR of lignin content and that of the MFA in reaction wood of Liriodendron tulipifera with G-layer-like fiber [6,8]. These researchers observed that the lignin content in wood fibers of tension wood decreased with decreasing MFA, i.e., a positive correlation exists between those of two characteristics.
Conclusion
Some reaction wood characteristics, such as vessel diameter and length, fiber diameter, fiber wall thickness, and fiber length, and MFA in the S2 layer of fiber were examined in nine Japanese hardwood species without a G-layer but with a three-layered (S1+S2+S3) structure in their reaction wood fibers. Although a unique nature in the traits of reaction vessels was observed, this was not found in the characteristics of reaction fibers. The G-layer was absent in reaction wood fiber, but the MFA in the S2 layer decreased significantly in Euscaphis japonica and Ilex crenata. The relationships among anatomical features in reaction wood were discussed. A significant negative correlation was observed between the growth eccentricity and IR of fiber wall thickness, and a positive correlation was observed between the DR of lignin content and that of vessel number. It is likely that the lignin content was reduced in the reaction wood of the nine species because of the reduction in the number of vessels.
References
- Onaka F (1949) Studies on compression-wood and tension-wood. Wood Res 1: 1-88.
- Day AC (1965) Anatomy and ultrastructure of reaction wood. In: Côté WA (Eds) Cellular Ultrastructure of Woody Plants. Syracuse University Press, New York, pp. 391-418.
- Wardrop AB (1965) The formation and function of reaction wood. In: Côté WA (Eds) Cellular Ultrastructure of Woody Plants. Syracuse University Press, New York, pp. 371-390.
- Yoshizawa N (1987) Cambial responses to the stimulus of inclination and structural variations of compression wood tracheids in gymnosperms. Bull Utsunomiya Univ Forests 23: 23-141.
- Yoshizawa N, Inami A, Miyake S, Ishiguri F, Yokota S (2000) Anatomy and lignin distribution of reaction wood in two Magnolia species. Wood Sci Technol 34: 183-196.
- Sultana RS, Ishiguri F, Yokota S, Iizuka K, Hiraiwa T, et al. (2010) Wood Anatomy of nine Japanese Hardwood species forming reaction wood without gelatinous fibers. IAWA Journal 31: 191-202.
- Okuyama T, Yamamoto H, Yoshida M, Hattori Y, Archer RR (1994) Growth stresses in tension wood: Role of microfibrils and lignification. Ann For Sci 51: 291-300.
- Yoshida M, Ohta H, Yamamoto H, Okuyama T (2002) Tensile growth stress and lignin distribution in the cell walls of yellow poplar, Liriodendron tulipifera Linn. Trees 16: 457-464.
- Fang CH, Guibal D, Clair B, Gril J, Liu YM, et al. (2008) Relationships between growth stress and wood properties in poplar I-69 (Populus deltoides Bartr. cv. “Lux” ex I-69/55) Ann For Sci 65: 1-9.
- Kobayashi Y (1952) A simple method of demonstrating the fibrillar orientation in lignified walls. Japanese Forestry Soc 34: 392-393.
- Hiraiwa T, Yamamoto Y, Ishiguri F, Iizukz K, Yokota S, et al. (2007) Cell wall structure and lignin distribution in the reaction wood fiber of Osmanthus fragrans var. a urantia cus Makino. Cellulose Chem Technol 41: 537-543.
- Chow KY (1947) A comparative study of the structure and chemical composition of tension wood and normal wood in beech (Fagus sylvatica L). Forestry 20: 62-77.
- Jourez B, Riboux A, Leclercq A (2001) Anatomical characteristics of tension wood and opposite wood in young inclined stems of poplar (Popular euramericana cv ‘Ghoy’). IAWA Journal 22: 133-157.
- Wardrop AB, Dadswell HE (1955) The nature of reaction wood. IV. Variations in cell wall organization of tension wood fibres. Australian J Bot 3: 177-189.
- Zimmermann MH (1964) The formation of wood in forest trees (Eds). Academic Press, New York 145: 562.
- Dadswell HE, Wardrop AB (1955) The structure and properties of tension wood. Holzforchung 9: 97-104.
- Wardrop AB (1956) The nature of reaction wood. V. The distribution and formation of tension wood in some species of Eucalyptus. Australian J Bot 4: 152-166.
- Xu F, Zhonga XC, Sunb RC, Luc Q (2006) Anatomy, ultrastructure and lignin distribution in cell wall of Caragana Korshinskii. Industrial Crops Prod 24: 186-193.