Review Article
Study on the Floristic Diversity and Threatened Plant Species in Babile Elephant Sanctuary in East Hararge, Ethiopia: Review Paper
Fentahun Y**and Abdala T
Corresponding author: Yeneahayu Fentahun, Ethiopian Biodiversity Institute, Harar Biodiversity Center, Harar, Ethiopia, Tel: +251912857979; E-Mail: yeneayehu07@gmail.com
Citation: Fentahun Y, Abdala T. Study on the Floristic Diversity and Threatened Plant Species in Babile Elephant Sanctuary in East Hararge, Ethiopia: 02 Review Paper. J Plant Sci Res. 2017;4(1): 163.
Copyright © Fentahun Y, et al. 2017. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Plant Science & Research | ISSN: 2349-2805 | Volume: 4, Issue: 1
Submission: 02/02/2017; Accepted: 15/03/2017; Published: 20/03/2017
Abstract
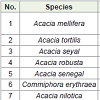
Babile Elephant Sanctuary (BES), in the semi-arid part of eastern Ethiopia, has long been known for comprising one of the globally threatened, ecologically isolated and possibly distinctive subsp. of elephant population (Loxodonta Africana orleans). As this review (In the present communication) a total number of 55 identified plant species comprising 35 genera and 24 families has been studied/described. Out of these, shrubs have accounted for 94.9% while trees constituted only 5.1% of the total density. Fabaceae, Tiliaceae and Capparidaceae are the most dominant families’ 1st, 2nd and 3rd dominant, respectively. Browsing elephant, anthropogenic and invasive alien are the major threat of plant species in BES. Acacia robusta, Acacia senegal, Commiphora erythraea, Acacia nilotica, Balanites glabra, Acacia mellifera, A. tortilis and A. seyalare the major useful plant and impacted species by human use, browsing elephant and invasive. Therefore, future research should focus on detailed assessment of current vegetation status in the sanctuary; rehabilitation by plantation especially the indigenous multi-purpose tree species that are highly threatened but still available in the valley should be also given emphasis.
Keywords: Cut flowers; Clarkia amoena; Defoliation; Pulsing; Silverthiosulphate; Vase life
Abbreviations: African elephant; Babile Elephant Sanctuary (BES); Plant diversity; Threatened plants
Introduction
Establishment of protected areas is vital for in situ conservation of diverse native species. These activities are undertaken for the purpose of education, research, and recreation. Moreover, these areas provide essential items such as fuel wood, building materials, forage, traditional medicines and wild foods. Up to now, no area has beenformally protected in the country to conserve an ecosystem or habitat important for plant species, although, Ethiopia is known for its high diversity of plant species [1]. Protected areas cover approximately 16.4% of the country’s surface area [2]. There are 20 national parks, 3 sanctuaries, 2 wildlife reserves and 17 controlled hunting areas, 7 open hunting areas and 3 community conservation areas covering an area of about 65,531.4 km2 [3]. However, except few, most of the protected areas exist on paper only and have declined in size and quality.
BES, located in the semi-arid part of eastern Ethiopia, is a part of the Somali-Masai Centre of Endemism [4]. This sanctuary is one of the protected areas in the country established to protect the only viable elephant population in the Horn of Africa. These elephants have been separated from other populations in Ethiopia for more than eight decades. Despite the establishment of the Sanctuary in 1970, their range of distribution has shrunk considerably. As a result of mass influx of a large number of farmers and their livestock from the east and north, the home range of elephants of Babile has shrunk by about 65.5 percent since 1976 [4]. Moreover, a recent year 10,000 hectares of the north western part of the land given to a private
company engaged in the cultivation of castor used for production of biofuel [4] is being described as a calamity to the already declining elephant population and other wildlife. Of the total 10,000 hectares of land granted to the company, 87.4% was proved to fall within the boundary of the sanctuary, and of this 79.2% were within the present elephant ranges movement corridors and regular feeding grounds for elephants [4]. Local communities around the area are highly dependent on vegetation for fencing, medicine, construction and fuel wood [5].
Floristic Diversity
Plant diversity
Biological diversity or simply “Biodiversity” is defined as the total variability of all living organisms and the ecological complexes in which they occur. “Species diversity” refers to the number of species found within a given area. “Genetic diversity” refers to the variety of genes within a particular species variety or breed [6,7]. “Plant diversity’’ refers totality and variability of all plants and their ecosystems.
The diversity of higher plant species increases as one move from the pole to the equator. According to Groom bridge, 40-100 tree species may occur in one hectare of tropical moist forest in Latin America, compared to 10-30 per hectare in Northeastern America [8]. The estimated number of Angiosperm species in the world and Ethiopia is 250,000 and 6000-7000, respectively. However, due to biotic and/or a biotic factors species and ecosystems are being lost [6]. The recognition of this loss at a global level has led scientists and concerned groups to draw up world conservation strategy and the Convention on Biological Diversity held at the Earth Summit in Rio de Janeiro, Brazil in June, 1992. At the national level, Biodiversity Institute and National Conservation Strategy have been established. In addition, various government, non-governmental organizations and Peasants Associations are involved in conservation practices [10].
Diversity in ecological research could refer to species diversity, habitat diversity or genetic diversity indicating variability betweenspecies, habitat and individuals, respectively [11]. Species diversity could be viewed from different approaches in terms of alpha (α), Beta (β) and gamma (γ) diversity [12]. Alpha diversity refers to the diversity of species within particular habitat or community. Beta diversity is a measure of the rate and the extent of changes in species along gradient from one habitat to another. It is diversity between habitats that measures the turnover rate. Gamma diversity on the other hand is the diversity of species in comparable habitat along geographical gradients and is independent of the two.
Diversity indices are simple mathematical expressions that summarize a lot of data recorded in one or sets of figures. Among the different coefficients widely used to compute diversity of a community are species richness, evenness and heterogeneity [13]. Species richness is the simplest concept of species diversity implying the number of species in a community. Heterogeneity is the measure of the probability of which two individuals randomly picked from a community belong to a different group or species [14]. Evenness is the measure of equitability. It attempts to quantify the unequal representation of species in a community against a hypothetical community in which all species are equally common [15].
Assessments of plant diversity have been frequently used to judge the success of conservation efforts besides being a criterion for the identification of potential conservation sites [16]. Therefore, it is important to assess the floristic composition and dynamics of the forest communities after protected area establishments in order to comprehend the conservation efforts made so far [17-19].
Composition and population characteristics of woodyplant species in BES
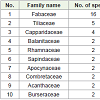
A total of the 55 woody plant species sampled in the riverine and woodland habitats, the impacts of elephants were recorded on 28 (50.9%) species belonging to 12 families. Out of these, 13 (45.8%) were trees and 15 (54.2%) shrubs. The total density of these 28 woody plant species was found to be 11,169.3 individuals/ha where the mean density was 399. Whereas the maximum density was 3,842.7 individuals/ha (Opuntia ficus-indica), the minimum density 3.7 individuals/ha was recorded for Combretum molle. The total density of trees was 558.7 individual/ha and shrubs 10,610.6 individual/ha. Furthermore, shrubs have accounted for 94.9% while trees constituted only 5.1% of the total density [20].
Threat of Plant Species in Babile Elephant Sanctuary(BES)
Impact of elephant on woody plant species
Population characteristics of impacted woody plant species: Consequences of the feeding and foraging behavior of elephant populations are important for woody species, especially whendeveloping conservation management options [21]. Elephants spend 16 hours a day feeding, with peaks in the morning, afternoon, and around midnight. An elephant’s diet requirement is usually met by forest-edge and woodland, and these habitats also provide theelephants with valuable shades [22]. Elephant damage was not randomly distributed for riverine and woodland and habitats inBabile Elephant Sanctuary. Shrubs accounted for 94.9% of the total density of the 28 woody species impacted by elephants, whereas trees only 5.1%. This is because shrubs are the dominant components of the floristic composition of the study area. As elephants prefer trees to shrubs, meeting the high feed demand of elephants of the BES could be a future challenge [22].
As many uncommon species were less frequently impacted than some of the more abundant species, any change in woody vegetation composition that might result from these impacts is likely to benefit species diversity in BES. Elephants are known to be physical ecosystem engineers with an ability to directly or indirectly control the availability of resources to other organisms [23]. Pamo and Tchambahave also shown that elephants have varied effects on the vegetation; they can cause detrimental changes to some species, at the same time increasing the abundance of other species [24]. Whitehouse and Schoeman found that elephant foraging resulted in the reduction of plant biomass and abundance at Serengeti National Park [25]. However, Midgley et al. contradicted these results by showing that the density of woody plants in the Addo Elephant National Park increased with elephant defoliation and attributed this to the fact that the removal of the canopy allows for coppicing to occur [2,22]. In both of the riverine and woodland habitat types, the frequency of elephant damage varied according to the woody plant species and size classes. Density was calculated for woody species impacted by elephants, by calculating the average individual tree and shrubs, in terms of the number of individuals per hectare in both areas (Table 1) [20,29]. The highest density of shrubs is due to the domination of small-sized shrubs in the floristic composition in both communities. Opuntia stricta, Acacia brevispica, Kleinia squarrosa and Acacia mellifera constitute about 50% of the total density. The most probable reason for the domination of these species may be specialization of the different species to different dispersal agents. Wind can carry light seeds with thin cotyledons, e.g. Acacia mellifera for a considerable distance [27]. Some of the plant species may have a wide range of dispersal mechanisms and/or rapid reduction strategies. Cowling and Kerley also suggested that since most of the shrubs produce birddispersed fruits, they are able to recolonize areas successfully (Table 2) [20,23,28,29].
According to the informant report, out of the ten top woody species browsed by elephants, Acacia tortilis, Acacia nilotica, Acacia etbaica, Balanites aegyptiaca, Balanites glabra, Berchemia discolor Oncobas pinosa, Salvadora persica and Tamarindus indica accounted for only 1.4% of the total density, suggesting that they are highly threatened [29].
Anthropogenic impact on woody plant species
Anthropogenic changes in vegetation over the past century need to be accounted for when assessing elephant impact. The harvesting and consumption of plant products from natural vegetation is known to account for a large proportion of the livelihood of people living close to such habitats. At the global level, socio-economic and political forces that determine the mode of development in many developing countries play an important role in the processes of vegetation degradation and destruction [30].
The major anthropogenic impacts in the BES: Temporaryand permanent settlement, charcoal making, firewood collection, overgrazing, deliberate fire to clear the vegetation for agriculture, andselective tree cutting for construction were the main threats in the sanctuary. Agriculture scored 5 as a major threat to the sanctuary, followed by human settlement and overgrazing 4, charcoal making and tree cutting 3, invasive species 2, and honey collection 1. Approximately, 400 ha of land along the Erer River have been used for agriculture illegally. A total of 31 small villages were counted in the sanctuary. Even during the data collection period, about 20 ha of woodland were cleared (burned) in the different part of the Erer Valleymand prepared for agriculture illegally. However, crops cultivated near protected areas are attractive to elephants as an alternative source of food, which has a potential to create human-elephant conflicts [31].
Use of riverine and woodland resources: Woody plant species such as Acacia mellifera, Acacia tortilis, Balanites glabra and Acacia senegal were used for a variety of purposes in relatively large quantities and at higher frequencies. These species are relatively widespread at Babile Elephant Sanctuary. They represent some of the dominant and large woody plants on the sanctuary, the branches, bark, leaves and trunks of which can be used for a variety of purposes and in satisfactory quantities. Even though they are used in relatively largerquantities, these plant species are still considered relatively available. This is because they are characterized by relatively large basal and crown cover and once the saplings grow fast beyond the browsingrange of the local livestock, while their thorns discourage browsing by a variety of free-ranging domestic and wild ungulates that inhabit the rangelands [32].
The sapling of Acacia mellifera is characterized by large multiplestemstands, they easily out-competes other plant species, to the extent of dominating most of the landscape. It successfully colonizes degraded soils and sites due to its natural ability to fix atmospheric nitrogen, which is a limiting nutrient in such soils [33,34]. This species was also preferred because of its durability and high wood density. A. mellifera has the ability to recover quickly, especially if only the crown is harvested, and has short seed germination duration of 2-14 days and 50-80% seed viability [35]. Balanites glabra is also successful in the BES, partly because of its ability to survive in the alkaline soils [36]. A large crown dominates the growth form of Acacia tortilis, with a deep and widespread root system which enables it to access waterin the lower strata of the soil horizon. These potentially increase itsability to withstand dry land conditions. Its pods are widely consumedby a variety of domestic and wild herbivores, which facilitates thebreaking of seed dormancy as well as enhancing germination rate andpromoting high seed dispersal (Table 3) [20,37].
Table 3: Some of the important species used by human being and their mainuses [20].
Expansion of invasive species
Lantana camara: The invasive species Lantana camara was observed as the dangerous species for both plants and animals during the field observation of this study. Lantana camara is an angiosperm, the origin of diversity of which is America. It is a small perennial shrub which can grow up to 2 meters in height and form dense tickets in a variety of environments. One of the highest problems of Lantana camara is that it breeds by both sexual and asexual means. Munir reported that up to 12000 (twelve thousand) seeds per plant per year can be produced by each plant which are then eaten by birds and other animals which can spread the seed over large distances facilitating the spread of Lantana camara [38]. This situation is creating a seriousproblem for plants and animals. This is due to its significance of being a dangerous weed found in a variety of environment including grassland, farmland, forest margins and gaps in both natural an semi natural areas of forest. Therefore, Lantana camara is considered as invasive species by dominating other native fauna and flora. It prevents the growth of other trees and plants. Desalegn Desissa and Sanders were also of the same opinion [39,40]. Lantana is a weed not used by the animals as fodder or human being for any productivepurpose but it is rapidly covering mainly the grazing land of the sanctuary. It is also becoming a major threat to the grassland-the main feed source for the elephants. Lantana camara is also reducing the crop production. This invasive species is less prone tobe eaten by elephants and livestock due to its toxicity. According to Desalegn Desissa, this toxicity also inhibits competing plant species [39]. And it is the shelter for threatening wild animals like wild cats, hyena, warthog and others. According to the people inhabiting the studyarea, Lantana camara was creating problems since it was introduced during 1980s.
Conclusion and Recommendation
Babile Elephant Sanctuary (BES), in the semi-arid part of eastern Ethiopia, has long been known for comprising one of the globally threatened, ecologically isolated and possibly distinctive subsp. of elephant population (Loxodonta Africana orleans). As this review describe a total 55 plant species composed of 33 genera and 24 families were identified. Out of these, shrubs have accounted for 94.9% while trees constituted only 5.1% of the total density. Fabaceae, Tiliaceae and Capparidaceae are the most dominant family 1st, 2ndand 3rd dominant respectively. Browsing elephant, anthropogenic and invasive alien are the major threat of plant species in Babile elephant sanctuary. Acacia robusta, Acacia senegal, Commiphoraerythraea, Acacia nilotica, Balanites glabra, Acacia mellifera, A. tortilis and A. seyal are the major useful plant and impacted species by human use, browsing elephant and invasive. Therefore, following recommendation needed for further conservation:
• Future research should also focus on detailed assessment of current vegetation status in the Sanctuary; emphasis should be given on the rehabilitation by plantation especially the indigenous multi-purpose tree species that are highly threatened but still available in the valley.
• To use the area as an ecotourism site in order to attract visitorsdue to its biodiversity potential and topographic features. This can generate income that can be used to the benefits of the sanctuary and the local community if better management of the sanctuary is planned and implemented.
• Rehabilitating more impacted indigenous tree to maximize their regeneration status.
• Babile Elephant Sanctuary (BES) should be afforded the highest protection possible as a matter of urgency before it is too late.
• To demarcate the area from expansion of agricultural land and over grazing.
• To eradicate invasion through community mobilization and appropriate measure.
References
- Gole TW (2003) Vegetation of the Yayu forest SW Ethiopia: impacts of human use and implication for in situ conservation of wild Coffea arabica L. populations. Ecology and Development series No. 10.
- United Nations Environment Programme (2003) World Conservation Monitoring Centre (UNEPWCMC). World Database on Protected Areas (WDPA) Version 6. Compiled by the World Database on Protected Areas Consortium. Cambridge, UK.
- (2011) Ethiopian Wildlife Conservation Authority.
- Demeke Y, Marilyn BR, Roger VS, Richard FB (2006) The undisclosed facts about the relic elephant population in the horn of Africa. In: Proceeding of the biological society of Ethiopia, 16th Annual Conference and Workshop. pp. 13.
- Belayneh A, Workeye F (2008) “Conservation and investment: Observation from the Babile Elephant Sanctuary.†IBCR Newsletter. Addis Ababa, Ethiopia.
- WRI-IUCN-UNEP (1992) Global diodiversity strategy.
- ECPGR (1993) Diversity for development: the new strategy of the international plant genetic resource institute. IPGRI, Rome, Italy, pp. 69.
- Groombridge B (1992) Global diodiversity: Status of the Earth’s living resources. World Conservation Monitoring Center, Chapman and Hall, London.
- Tsidell CA (1991) Economics of environmental conservation: economics for environmental and ecology management. Elsevier, Amsterdam, pp. 233.
- Watson RT Heywood VH, Baste I, Dias B, Gamez R et al. (1995) Global biodiversity assessment. United Nations Environmental Program, Cambridge University.
- Spellerberg IF (1994) Monitoring ecological change. New York Cambridge University Press.
- Rosenzweig ML (1995) Species diversity in space and time. Cambridge University Press, pp. 436.
- Krebs CJ (1989) Ecological methodology. Harper and Row, 654.
- Hurlbert SH (1971) The non-concept of species diversity: a critique and alternative parameters. Ecology 52: 577-586.
- Lioyd M, Ghelardi RJ (1964) A table for calculating the ‘Equitability’ component of species diversity. J. Anim Ecol 33: 217-225.
- Hall DL, Willig MR (1994) Mammalian species composition, diversity and succession in conservation reserve program grasslands. South West Nature 39: 1-10.
- Geilbach FR (1975) Investigation, evaluation and priority ranking of natural areas. Biol Conesrv 8: 79-88.
- Margules C, Usher MB (1981) Criteria used in assessing wildlife conservation potential: A review. Biol Conserv 21: 79-109
- Baskin Y (1994) Ecosystem functions of biodiversity: Regarding ideal levels of species richness, the data are few and unclear. BioScience 44: 657-660.
- Wodu Z (2007) Elephant and anthropogenic impacts on wood plant species in Babile Elephant Sanctuary, Eastern Ethiopia.
- Wiseman R, Page BR, O’Connor TG (2004) Woody vegetation change in response to browsing in Ithala Game Reserve, South Africa. S Afr J Wild Res. 34: 25-37.
- Croze H (1974) The Seronera bull problem: The elephants. Afr J Ecol 12: 1-27.
- Cowling R, Kerley G (2002) Impacts of elephants on the flora and vegetation of subtropical thicket in the Eastern Cape. In: Kerley G, Wilson S, Massey A (Eds). Elephant conservation and management in the Eastern cape: Workshop proceedings. Terrestrial Ecology Research Unit, University of Port Elizabeth, South Africa 35: 55-72.
- Pamo ET, Tchamba MN (2001) Elephants and vegetation change in the Sahelo-Soudanian region of Cameroon. J Arid Environ 48: 243-253.
- Whitehouse AM, Schoeman DS (2003) Ranging behavior of elephants within a small, fenced area in Addo Elephant National Park, South Africa. Afr Zool. 38: 95-108.
- Midgley JJ, Balfour D, Kerley GI (2005) Why do elephants damage savanna trees? : Commentary. S Afr J Sci 101: 213-215.
- Tybrik K (1991) Regeneration of woody legumes in Sahel. Aarhus University Press (DK). 81.
- Tesemma AB (1993) Useful trees and shrubs for Ethiopia: Identification, propagation and management for agricultural and pastoral communities. Regional Soil Conservation Unit, SIDA, pp. 474.
- Belayneh A, Demissew S (2011) Diversity and population structure of woody species browsed by elephants in Babile Elephant Sanctuary, eastern Ethiopia: an implication for conservation. Agric. For. 3: Pp. 20-32.
- Sindiga I, Chacha CN, Kanunah MP (1995) Traditional medicine in Africa. East African Educational Publishers, pp. 197.
- Demeke Y (2008) The Ecology and Conservation of the relic elephant population in the Horn of Africa. Thesis University of Melbourne, pp. 32-41.
- Mbuya LP, Msanga HP, Ruffo CK, Birnie A and Tengnas B (1994) Useful trees and shrubs for Tanzania: identification, propagation, and management for agricultural and pastoral communities. Regional Soil Conservation Unit (Nairobi), 542.
- International Centre for Research in Agro-forestry (1992) A selection of useful trees and shrubs for Kenya: notes on their identification, propagation, and management for use by agricultural and pastoral communities. Nairobi, Kenya, pp. 313.
- Moore R, Clark WD, Vodopich DS (1998) Botany (2nd edn). McGraw-Hill, Boston. pp. 919.
- Argaw M, Teketay D, Olsson M (1999) Soil seed flora, germination and regeneration pattern of woody species in an Acacia woodland of Rift Valley in Ethiopia. J Arid Environ 43: 411-435.
- Brady NC, Weil RR (1999) The nature and properties of soils. Prentice Hall, New Jersey. pp. 881.
- Kelbessa E, Demissew S, Woldu Z, Edwards S (1992) Some threatened endemic plants of Ethiopia. In: Edwards S, Asfaweds Z (Eds). Botany 2000: East and central Africa. NAPRECA Monograph, pp. 35-55.
- Munir AA (1996) A Taxonomic Reviews of Lantana Camara L. and L. Montevidensis (SPRENG.) Briq. (VERBENACEAE) in Australia. J Adlaide Bot gardens 17: 1- 27.
- Binggli P, Desissa D (2003) Ethiopian wildlife and natural history society. Newsletter: 4-6.
- Sanders RW (2012) Taxonomy of Lantana sect. Lantana (VERBENACEAE): II. Taxonomic revision. J Bot Res Inst Texas 6: 403-442.