Research Article
Effect of NaCl on Root Proliferation of Musa Species through Tissue Culture
Nibedita Chhatoi, Bikram Pradhan and Bandita Deo*
Corresponding author: Bandita Deo, Plant Physiology and Biochemistry division, Regional Plant Resource Centre, Nayapalli, Bhubaneswar- 751015, Odisha, India, E-mail: bdeo2008@gmail.com
Citation: Chhatoi N, Pradhan B, Deo B. Effect of NaCl on Root Proliferation of Musa Species through Tissue Culture. J Plant Sci Res. 2016;3(2): 163.
Copyright © Nibedita Chhatoi, et al. 2016. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Plant Science & Research | ISSN: 2349-2805 | Volume: 3, Issue: 2
Submission: 01/10/2016; Accepted: 04/11/2016; Published: 12/11/2016
Abstract
The present study was conducted to observe the morphological effect in saline condition through development of roots during rooting culture of Musa paradisiaca cv. Bantal. Various parameters including days of response, percentage of response, average numbers of roots, etc; were taken after root formation from shoots in Murashige & Skoog medium containing different concentration of Sodium Chloride (NaCl). It was observed that NaCl used in low concentration (100 mg/l) produce healthy and thick roots. But in case of higher concentration (500 mg/l) the formations of roots were thin, short and unhealthy whereas the proliferation of roots was also reduced.
Keywords: Musa paradisiaca; Murashige and Skoog medium; Tissue culture; NaCl
Introduction
Bananas and plantains are giant herbs belong to the familyMusaceae and mostly cultivated in the tropical and sub-tropical regions. With an annual productivity of 100 million metric tons [1], banana is the world's fourth most essential global food crop. India is the leading producer of banana with an annual production of nearly 30 million tons followed by China and Uganda
.
Salinity is an important abiotic stress factor severely affectingproductivity and survival of plant species. Scientists reported that salinity stress causes many adverse effects on the growth and development of millets [2]. Salinity causes inhibitory effects on plant growth, morphology, and survival because of the toxicity of excessive Na+ and Cl- to the absorbance of water. It is revealed that salt stress causes an imbalance of the cellular ions resulting in ion toxicity and osmotic stress [3,4].
Tissue culture techniques have been applied to the plant speciesto produce new clones and cultivars with improved characteristics.Thus, numbers of researchers have suggested that cultured tissues andcells may prove useful both in selections of the salt-tolerant plants and in studies of the physiological basis for salinity tolerance [5,6]. In vitro culture can also be used for studying the effects of different abiotic stress factors, like drought [7] and heavy metals [8].
The aim of the present study was to observe the different morphological abnormalities of banana shoots and roots grown on Murashige and Skoog medium containing low NaCl concentration (100 mg/L) and high NaCl concentration (500 mg/L).
Material and Method
Plant material
The shoots for the present study were collected from the multiplication phase of Bantal grown in vitro at Banana tissue culturelab of Regional Plant Resource Centre (RPRC), Bhubaneswar. The healthy shoots with 2 - 3 leafs were obtained at 4th and 5th subculture through the tissue culture of the meristem on MS medium containing growth regulators.
Culture medium
In our study the medium used for banana tissue culture was Murashige and Skoog Medium [9]. The phytohormone used for the root culture studies was Indole -3-acetic acid (IAA).
Aseptic transfer of the explants: The working area of the laminar airflow was first wiped with cotton moistened with 70% ethanol and then irradiated with ultraviolet light for 30 minutes beforeinoculation. The shoots were taken out to autoclaved petridish and shoots were isolated using sterilized scalpel and forceps. The shoots were inoculated in the culture vessel containing induction medium. The culture vessels containing the shoots were kept in culture rack maintained at 24 °C - 26 °C for 16 hours of photo period with intensity of 35-50 μEm-2s-1 intensity provided by cool white fluorescent tubes in the culture room of Banana Tissue Culture Lab.
Results
Development of Roots at Different Salt Concentration in Rooting Culture
In the present study the selected shoots of Mus asp cv. Bantalwere cultured in media containing Sodium Chloride (NaCl) at twodifferent concentration 100 mg/L and 500 mg/L. From the experiment it was found that shoots cultured in rooting medium containing 1 mg/L IAA and 100 mg/l NaCl showed better result in comparison to medium containing 500 mg/l NaCl having same quantity of IAA [Figure 1].
Figure 1: Figure shows the roots development in shoots grown on MS medium with NaCl 100 mg/L (1a) and 500 mg/L (1b) after 21 days.
Here 1 mg/L IAA in MS medium was considered as the control forall the experiments. After 7 days no remarkable growth was marked in the rooting culture and also after 15 days the response of growth in culture media was not adequate. After 21 days it was observed that shoots grown on control medium produced healthy and long roots. The percentage of response was 100%, the average number of roots was 3.3 and the days of response were in short duration.
1. a. Effect of sodium chloride at 100 mg/L: From the data it was observed that NaCl used in low concentration produce healthy and thick roots but the length of the roots were reduced. The percentage of response was 100% but the average number of roots reduced to 2.7 which was less in comparison to control [Figure 2].
Figure 2: Figure shows the comparative results (average days of response and average number of roots) of shoots grown on control and MS medium containing NaCl (100 mg/L and 500 mg/L).
1. b. Effect of sodium chloride at 500 mg/L: The percentage of response of root growth was 80% and the average number of roots was 2.3 in shoots grown on medium containing 500 mg/L NaCl (Figure2). It was depicted that with the increase in the salt concentration theroot proliferation was reduced. The formations of roots were thin, short and unhealthy. The days of response of shoots were seen to be longer than those cultured on control as well as medium containing 100 mg/L NaCl.
Discussion
1- Study of initiation of roots using IAA
Auxins mostly utilized in the medium to induce rooting are in theform of IAA, IBA, and NAA. During this experiment it was observed that shoots grown in 1 mg/L IAA had developed good numbers of roots in short duration. Generally higher auxin concentrations are required during the induction phase, whereas during the later stage it becomes inhibitory. This effect is also marked when micro cuttings are cultured continuously on media with auxins, studied by De Klerk et al. in apple microcuttings previously exposed for 3 weeks to IAA and which rooted ex vitro more efficiently than those exposed to IBA or NAA [10].
2- Development of roots at different salt concentration inrooting culture
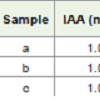
In the present experiment it was revealed that the percentage of root germination and growth was 100% in case of control but with increase in NaCl concentration (100 mg/L and 500 mg/L) the germination percentage and growth rate was decreased [Tables 1-3].
Growth inhibition is a common response to salinity and plantgrowth is one of the most important agricultural indices of salt stresstolerance as indicated by different studies [11]. With the increase in sodium chloride concentration the percentage or response of roots decreased in this experiment. It was observed that roots formation at high NaCl concentration were unhealthy, weak and very short in compared to the roots grown in low salt concentration. Growthreduction was also observed with the increase of NaCl concentrationin three pea species grown through in vitro culture [12].
Conclusion
In the present experiment the activity of NaCl was found to be less in case of 100 mg/L and it increased as the concentration of NaCl increased (500 mg/L). Exposure of plant to abiotic stress like NaCl imparts the ionic toxic effects on plant morphology like shoot and root growth. Here under high stressed condition the growth of the roots, days of response, average root number, percentages of response were remarkably affected. Almost all the parameters were considered to be affected significantly in both stressed condition in in vitro culture of Musa paradisiaca cv. Bantal. For better assessment it needs further study.
Acknowledgments
The authors are thankful to Regional Plant Resource Centre,Department of Forest and Environment, Odisha, India for providinglaboratory facility and funding.
References
- FAOSTAT (2010) Food and Agriculture Organization of the United Nation Statistical Database.
- Hussain J, Ullah R, Rehman N, Khan AL, Muhammad Z, et al. (2010) Endogenous transitional metal and proximate analysis of selected medicinal plants from Pakistan. J Med Plants Res 4: 267-270.
- Khan MN, Siddiqui MH, Mohammad F, Naeem M, Khan MMA (2010) Calcium chloride and gibberellic acid protect linseed (Linum usitatissimum) from NaCl stress by inducing antioxidative defence system and osmoprotectant accumulation. Acta Physiol Plan 32:121.
- Munns R, James RA, Luchli A (2006) Approaches to increasing the salt tolerance of wheat and other cereals. J Exp Bot 57: 1025-1043.
- Chen Y, Zahavi E, Barek P, Umiel N (1980) Effect of salinity stresses on tobacco: I. The growth of Nicotiana tabacum callus cultures under sea water, NaCl and Mannitol stress. Z Pflanzenphysiol 98:141-143.
- Umiel N, Zahavi E, Chen Y (1980) Effects of salinity stresses on tobacco. 2. Short term kinetics of Na+ and K+ uptake by callus cultures grown on media containing NaCl. Z Pflanzenphysiol 100: 363-367.
- Wang W, Vinocur B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218: 1-14.
- Deo B, Nayak PK (2011) Study of copper phytotoxicity on in vitro culture of Musa acuminate cv. Bantala. J Agric Biotech Sustain Dev 3: 136-140.
- Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15: 473-497.
- De Klerk GJ, Ter Bruge J, Marinova S (1997) Effectiveness of indoleacetic acid, indolebutyric acid and naphthalene acetic acid during adventitious root formation in vitro in Malus ‘Jork 9’. Plant Cell Tiss Organ Cult 49: 39-44.
- Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60: 324-349.
- Miljuš-Djukić J, Stanisavljević N, Radović S, Jovanović 1Ž, Mikić A, et al. (2013) Differential response of three contrasting pea (Pisum arvense, P. sativum and P. fulvum) species to salt stress: assessment of variation in antioxidative defence and miRNA expression. Aust J Crop Scie 7: 2145-2153.