Research Article
Optimization of Media Formulations for Callus Induction, Shoot Regeneration and Root Induction in Nicotiana benthamiana
Sundararaman Geethalakshmi1*, Balakrishnan Hemalatha2, Natarajan Saranya3
1Department of Biotechnology, Nehru Arts and Science College, Coimbatore, India
2Department of Biotechnology, Karpagam University, Coimbatore, India
3Department of Microbiology, Nehru Arts and Science College, Coimbatore, India
Corresponding author: Geethalakshmi Sundararaman, Department of Biotechnology, Nehru Arts and ScienceCollege, Coimbatore, India; E-mail: s.geethalakshmi@gmail.com
Citation: Geethalakshmi S, Hemalatha B, Saranya N. Optimization of Media Formulations for Callus Induction, Shoot Regeneration and Root Induction in Nicotiana benthamiana. J Plant Sci Res. 2016;3(1): 150.
Copyright © 2016 Geethalakshmi et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Plant Science & Research | ISSN: 2349-2805 | Volume: 3, Issue: 1
Submission: 16/05/2016; Accepted: 06/06/2016; Published: 11/06/2016
Abstract
Nicotiana tabacum and N. benthamiana have served as model species for the plant sciences for many years. For the studies involving transformation analysis using Agrobacterium, Nicotiana benthamiana has been gaining importance in recent years due to its ability to express auto fluorescent protein for detection of transformed genes and its efficiency in gene expression compared to other plants. This study aims at optimizing the culture media procedures for the in vitro culturing of this model plant. BAP (1 mg/l), IAA (0.1 mg/l) was suitable for callus induction in 20 days. This result was in accordance with previous studies. BAP (2 mg/l), IAA (1 mg/l), Kinetin (0.5 mg/l) was suitable for shoot regeneration in 25 days. This result differed from that of previousstudies where BAP (1 mg/l) and NAA (0.1 mg/l) was used for both callus induction and shoot regeneration. Thus, this study forms the basis for invitro rising of Nicotiana benthamiana for transformation and genetic engineering studies.
Keywords: Agrobacterium; Transformation; Growth regulators; In vitro culture; Nicotiana benthamiana.
Introduction
Tobacco is a natural allotetraploid formed through hybridizationbetween two diploid (2n=24) progenitors, N. sylvestris and N.tomentosiformis [1]. Within 3 months of time, a tobacco plant goes from seed to next generation seed and generates up to a million seed per plant. Scaling up to hundreds or thousands of acres is very rapid.
Earlier studies indicating the uptake of naked DNA to the recentvector mediated gene transfer. The first transgenic plants are producedin tobacco. Also, the experiments related to plant transformation,gene expression and gene stability have all been worked out usingtobacco. Currently, this plant used in studies of the production of useful recombinant proteins, antibodies and special chemicals for usein medicine and industry [2].
The genus Nicotiana includes 76 species which grows in tropicaland subtropical areas in the world. Nicotiana tabacum and N.benthamiana have served as model species for the plant sciencesfor more than 20 yr. Consequently N. benthamiana has become acommon source for the study of host/pathogen interactions in plants[3].
The technology that popularized N. benthamiana as a researchmodel was agroinfiltration in which the binary vector containinggenes to proteins of interest can be often expressed as functions to autofluorescent proteins, to be expressed transiently in plant cellswhich are well suited for high throughput studies [3].
Agro infiltration is a simple and useful technique, but for transientexpression this techniques has lots of complexities. A suspension ofAgrobacterium tumefaciens cells carrying binary plasmid vectorsdesigned for proteinexpression is infiltrated into the intercellular space within a leaf using. After 24 to 48 h of incubation, sectionsof infiltrated leaves can be sampled for microscopy or biochemicalanalyses. Agro infiltration techniques works exceptionally well inN. benthamiana but poorly in other plants, including Arabidopsisthaliana. Thus, advances for manipulating protein and geneexpression in plant cells are best suited to N. benthamiana [3].
Several studies have devised media for tissue culturing of N.benthamiana for specific purposes like viral transformation, host/virus interaction studies [4-6]. But an optimized media for callusinduction and shoot regeneration in this model plant has not yetbeen devised. This study aims at optimizing the culture conditions forNicotiana benthamiana, the results of which can form a foundationfor tissue culture and genetic engineering studies using this modelplant.
Materials and Methods
Seeds of Nicotiana benthamiana were sowed in plastic potscontaining soil: manure (10:1 w/w) and maintained under standardgreenhouse conditions at Karpagam University, Coimbatore, India.
Surface sterilization of the explants
Leaf, shoot, nodes and seed of N. benthamiana were taken asexplants for both callus induction and shoot regeneration. Surfacesterilization of the explants was performed according to [7] withmodifications. The explants were collected from the plant, washed inrunning tap water to remove the dust and other contaminants presenton the surface of the explants. It was then rinsed with distilled waterfor one minute and this step was repeated three times. The explantswere then taken into laminar hood for further surface sterilizationsteps. The explants were transferred into a conical flask containingethanol (70%) and rinsed for 30 seconds. This step was followedthree times followed by rinsing with distilled water for one minutethree times. This step was followed by treating the explants mercuricchloride (0.1%) for three minutes followed by rinsing with distilledwater. The explants were then blot dried using a blotting paper andtransferred to inoculation medium for callus induction and shootregeneration.
Callus induction and Shoot regeneration
For callus induction, different media compositions were usedindividually. All the compositions were prepared using basal MSmedia [8] supplemented with different concentrations of IAA (1.0 mg/l - 3.0 mg/l), BAP (0.25 mg/l - 1.0 mg/l) and Kinetin (0.5 mg/l -2.0 mg/l) with 30 gm/l of sucrose. Agar (8%) was used as solidifyingagent for all the composition. Earlier studies [9] used NAA (0.1 mg/l)instead of IAA as the auxin source. In the present study also, a callusinduction media was formulated with NAA (0.1 mg/l - 0.5 mg/l). Theculture media was sterilized using standard sterilization procedure.The surface sterilized explants were transferred aseptically to all themedia compositions and labelled accordingly.
Healthy callus grown after 20 days were inoculated in shootregeneration media formulated in different concentrations: BAP(0.5 mg/l - 2.5 mg/l), IAA (0.5 mg/l - 2.0 mg/l), Kinetin (0.5 mg/l -2.5 mg/l). Nearly equal amount of callus was transferred into all themedia. All the cultures were kept under 16 hrs photo period providedby white fluorescent tube lights (1000 lux) at 25±1 °C for four weeks.
Results and Discussion
The important criteria for plant tissue culture is to avoidcontamination and establish aseptic culture using explants whichprovide high risk of internal and external contamination [10]. Surfacesterilization of the explants was performed to obtain contaminationfree culture.
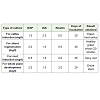
Of the various media formulations, healthy, hard green colourcallus was observed in the media containing BAP 1.0 mg/l and IAA2.0 mg/l. Kinetin had no effect on callus induction. The optimalincubation period was 20 days (Figure 1). This healthy callus was sub cultured in different shoot regeneration media. High number of shoot emergence (20 shoots) was observed in media containing BAP 2.0 mg/l, IAA 1.0 mg/l and Kinetin 0.5 mg/l. The optimal incubation period was 28 days for shoot regeneration from the callus (Figure 2). In this composition, minor amount of roots emerged but the root hairs were not healthy. Instead, in the media containing BAP (0.5mg/l, IAA 1.0 mg/l, Kinetin 2.0 mg/l, root hairs emerged after 28 daysof incubation but the shoots developed were not healthy (Figure 3).
Figure 1: Callus development from the explants. The calli were developed on the optimized media after 20 days of incubation.
Figure 2: Shoot regeneration from the callus. (A) Development of shoot from the inoculated callus in the shoot regeneration media. (B) Development of multiple shoots in the shoot regeneration media.
For rising an entire plant in the tissue culture media, theoptimum media formulation was found to be BAP 2.0 mg/l, IAA 1.0mg/l, kinetin 2.0 mg/l and the incubation period was 28 days (Figure 4). In all the cases, after the optimal incubation period, the shootsbecame transparent, roots became brown in colour and the colour ofthe callus became brownish indicating the degradation of the culture.Similarly, in certain media compositions, the explant and the callusbecame dry, indicating that these compositions were not suitablefor culture (Figure 5). Thus, Table 1 summarizes the optimal culture conditions for the in vitro rising of Nicotiana benthamiana.
Figure 5: Figure shows the results of unsuitable media formulations. (a) Browing of callus in non-suitable media. (b) Development of transparent leaf from callus. (c) Non responding inoculum.
Further, using this culture conditions, entire plant of Nicotianabenthamiana can be raised and taken for hardening.
Conclusion
Very few studies have been carried out for the culture conditionidentification and optimization for this model plant [11]. This planthas been gaining importance recently for genetic studies, especially inoverexpression analyses which can be mediated using Agrobacterium.Studies are rapidly increasing that develops genomic tools for N.Benthamiana for host-pathogen studies, over expression analysis andalso in virology [12,13]. It is evidently clear that this model plant could be an indispensable tool for plant systems biology in the coming days. Thus, this study describes quick, effective and reproducible procedure for in vitro culture and propagation of N. benthamiana in order to use it either for transformation or other genetic studies of desired genes.
References
- Okamura JK, Goldberg RG (1985) Tobacco single copy DNA is highly homologous to sequences present in genomes of its diploid progenitors. Mol Gen Genet 198: 290-298.
- Ganapathi TK, Suprasanna P, Rao PS, Bapat VA (2004) Tobacco (Nicotiana tabacum L,)-A model system for tissue culture interventions and genetic engineering. Int J Biotechnol 3: 171-184.
- Goodin MM, Dietzgen RG, Schichnes D, Ruzin S, Jackson AO (2002) pGD vectors: Versatile tools for the expression of green and red fluorescent protein fusions in agroinfiltrated plant leaves. Plant J 31: 375-383.
- Deo PC, Dugdale B, Harding RM, Kato M, Dale J (2015) In vitro micro propagation of Nicotiana benthamiana via axillary shoots. South Pacific J Natural App Sci 32: 55-60.
- Messinese E, Mun JH, Yeun LH, Jayaraman D, Rouge P, et al. (2007) A novel nuclear protein interacts with the symbiotic DMI3 calcium- and calmodulin-dependent protein kinase of Medicago truncatula. Mol Plant-Microbe Interact 20: 912-921.
- Tardif G, Kane NA, Adam H, Labrie L, Major G, et al. (2007) Interaction network of proteins associated with abiotic stress response and development in wheat. Plant Mol Biol 63: 703-718.
- Niedz RP, Bausher MG (2002) Control of in vitro contamination of explants from greenhouse and field-grown trees. In Vitro Cell Dev Biol 38: 468-471.
- Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiologia Plantarum 15: 473-497.
- Li B, Huang W, Bass T (2003) Shoot production per responsive leaf explant increases exponentially with explant organogenic potential in Nicotiana species. Plant Cell Rep 22: 231-238.
- Misaghi IJ, Donndelinger CR (1990) Endophytic bacteria in symptom free cotton plants. Phytopathology 80: 808-811.
- Clemente T (2006) Nicotiana (Nicotiana tabaccum, Nicotiana benthamiana), Methods in Molecular Biology: Agrobacterium Protocols (Ed 2) 1: 143-154.
- Caplan JL, Mamillapalli P, Burch-Smith TM, Czymmek K, Dinesh Kumar SP (2008) Chloroplastic protein NRIP1 mediates innate immune receptor recognition of a viral effector. Cell 132: 449-462.
- Takahashi Y, Nasir KH, Ito A, Kanzaki H, Matsumura H, et al. (2007) A high-throughput screen of cell-death-inducing factors in Nicotiana benthamiana identifies a novel MAPKK that mediates INF1-induced cell death signalling and non-host resistance to Pseudomonas cichorii. Plant J 49: 1030-1040.