Research Article
Effect of Leaf Powder Treatment on Root-Knot Nematode (Meloidogyne incognita) and Proximate Composition of Yam Tubers; Implication on Food Security
Aghale, Duke Nduka1*, Umeh, Ogechi Jubilant2, Anyim, Alozie3
1,2Michael Okpara University ofAgriculture, Umudike, Nigeria
3Abia State Ministry of Agriculture, Umuahia, Nigeria
Corresponding author: Aghale, Duke Nduka, Michael Okpara University of Agriculture, Umudike, Nigeria, Tel No:08134005735; E-mail: aghalenduka@yahoo.com
Citation: Aghale DN, Jubilant UO, Alozie A. Effect of Leaf Powder Treatment on Root-Knot Nematode (Meloidogyne incognita) and Proximate Composition of Yam Tubers; Implication on Food Security. J Plant Sci Res. 2016;3(1): 144.
Copyright © 2016 Aghale DN, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Plant Science & Research | ISSN: 2349-2805 | Volume: 3, Issue: 1
Submission: 11/02/2016; Accepted: 16/03/2016; Published: 25/03/2016
Abstract
Farmers face several challenges in yam production occasioned by incidents of pest and pathogens among which are nematodes, termites, beetles. The effect of leaf powder of five plants, Ocimum basilicum L. (Basil plant), Vernonia amygdalina.L., (bitter leaf) Azadirachta indica L. (neem) Moringa oleifera Lam. (horseradish) and Piper nigrum Schum and Thonn (bush pepper), were assessed for their effect on root-knot nematode, Meloidogyne incognita in yam and their influence on tuber proximate composition. The trial was conducted in Abia State, Nigeria laid out in completely randomized design, replicated three times. Data were collected and analyzed using descriptive statistics and analysis of variance (ANOVA). Results indicate that carbohydrate content increased by 10.4% in Basil plant, 21.4% in Bitter leaf, 8.1% in Neem, 11.1% in horseradish and 12.8% in Bush pepper treated yam tubers at 0.01t/ha when compared to the control. The protein composition increase was not significant (P > 0.05). Application of leaf powder at 0.01t/ha beforeplanting and during tuber initiation suppressed root knot nematode population and consequently, improved growth and yield of yam, improved the dietary value of the yam tubers which has the potentials for enhanced nutrition and food security of the Nigerian citizens and it is therefore recommended for wide spread farmers’ awareness and adoption.
Keywords: Nutritive values; Yam tubers; Meloidogyne incognita; Plant leaf powder; Awareness; Adoption
Introduction
Dioscorea species are consumed widely as staples in West Africa, Central Africa and the Caribbean [1]. Dioscorea rotundata is a major staple food in Africa with Benin Republic (346 calorie) as highestconsumer followed by Cote d Ivoire (342calorie), Ghana (296 calorie)and Nigeria (258 calorie) daily respectively [1]. Ninety eight percentof the world’s yam production is in Africa and West Africa accountsfor 90 - 95 % of the total world production. Nigeria is the largestproducer of the crop, producing about 33.57 million metric tons in 2008 [2] but consumes the entire product in the domestic market.Ghana which is the second largest world producer with 6.9 metrictons in 2008 is the highest exporter of the product [3].
Yams occupy the largest area in the production per hectare aftercassava. Yam is used as food as well as industrial raw material likeother root crops, such as sweet potato and cassava thus mountingpressure on yam as the most important staple which is needed tobe reposition as a food security crop [4]. Yams are major source ofrelatively cheaper caloric energy as starchy food that contains other useful nutrients. They are excellent source of carbohydrate and alsorelatively nutritious, providing some vitamins, minerals and dietaryprotein [5]. Yams play a significant role in the socio-cultural traditions of the producing areas, and are consumed in various food formsacross those producing regions either as boiled, mashed, pounded,fried or roasted.
Yams are affected by pests and pathogens among which arenematodes, termites, beetles, etc [6]. The root-knot nematodes(Meloidogyne species), and other nematodes such as Scutellonemabradys, Pratylenchus species (lesion nematodes) have been identifiedas the most economic pests of yam in Nigeria, which has resulted intoyield losses of 25-38% in major yam producing areas [7]. Onyenobiet al. noted that in most cases, nematode-infested yams in the fieldare without visible foliar symptoms and infestation by root-knotnematode on yam has contributed greatly into post-harvest biodeterioration [8]. It was observed that early senescence occurs inheavily infested minisett yam plant with galled yam tubers that rottedcompletely in storage [8]. Aghale noted that nematode damage onyam is an important factor which affects the tuber quality, resulting intuber yield reduction and general losses in the field and in storage [9].Meloidogyne species are group of plant-parasitic nematodes that havebeen recognized as pests of worldwide concern, constituting a majorthreat to world food production [7].
Several control measures such as cultural, biological, chemical andintegrated control methods, have been used in reducing populationsof the nematode in crop fields [9]. Synthetic nematicides are mostlyused to control yam nematodes. However, most of these nematicideshave been banned due to their carcinogenic effect on human lives andon the environment. High cost of the nematicides has also precludedresource-poor farmers from using them. The use of natural plantproducts has been considered as a way out of these chemical-relatedproblems in yam production since some plants have been identified topossess nematicidal properties, which has been proven to be efficientand easily accessible as seed dressing agents.
Aghale et al. [10] reported the efficacy ofnine plants (Caricapapaya, Chromolaena odorata, Cymopogon citratus, Mangiferaindica, Ocimum basilicum, Vernonia amygdalina, Azairachta indica,Moringa oleifera, and Piper nigrum) which were promising even in lowconcentrations in controlling the root knot nematode (Meloidogynespecies) in yam production [10]. Aghale noted strong nematicidalactivity of some plant extracts on nematode infested yams. However,in South East Nigeria, there is little or no information on the efficacyof bionematicidal materials on the levels of proximate content ofthe edible part of yam, the tubers [11]. This informed the design ofthe present investigation to determine the effect of the selected fiveplant leaf powder on the control of root knot nematode (Meloidogynespecies) and also on the proximate composition of yam tuber.
Material and Methods
Sampling Site
The study was conducted on the experimental research field ofthe Faculty of Agriculture, Abia State University, Nigeria, the PlantProtection Laboratory and the Biochemistry Laboratory of NationalRoot Crops Research Institute, Umudike, Nigeria. Five different typesof plants species used in the trials were collected from different partsof Umuahia in Abia state and these included Basil plant, Bitter leaf Neem, Horseradish and bush pepper. Healthy leaves of these plantswere harvested and spread separately on polythene sheets raised ona platform in the laboratory for three weeks to air dry at 28-+2 °C.Air-dried leaves were ground separately to fine particles (powder) ina domestic blender (model QBL-15L40) and applied at the rate of 5gper pot (0.01t/hectare).
D. rotundata (cv.Obiaturugo), a hybrid (89/19158) which is wellknown to be susceptible to M. incognita was used for the study [6].The size of yam sett used was 30 g. Prior to planting, the yam sets weretreated with the different plant leaf powder at the rates of 0.01t/ha ina jute bag tied at the top end and shaken properly before air drying ona raised platform. The sets were allowed for 24 hours for the powderto adhere properly on the sets before planting into the plastic pots.The yam sets were manually planted in 36 pots placed in open fieldwith one yam sett per pot. At 12th weeks after planting (WAP), the leafpowder was applied by ring method to the yam stands in the 30 potswhile the remaining six pots served as untreated controls. Earlier 10WAP, the yam plants in 30 pots were inoculated with 5000-secondstage pre-parasitic juvenile of M. incognita during tuber initiationwhen it is easy for the juveniles to penetrate the yam cortical cellsusing a 10ml syringe while the other six untreated control pots werenot inoculated.
The experiment was laid out in completely randomized designwith five treatments in three replicates. The eggs / juveniles that wereused for the inoculation were cultured and extracted using Husseyand Barker procedure [12]. Data were collected on sprouting at 4,8 and 12 WAP. At 28 WAP the experiment was terminated and thefollowing parameters were collected. Fresh tuber weight (g), proximatecomposition of the harvested tubers on carbohydrate, protein, fiber,ash, and fat using the gravimetric method of different laboratoryanalysis for the specific tests at Food/Biochemistry laboratory ofNational Root Crops Research Institute, Umudike [13]. Also, data ongall tuber formation was collected. The gall index (GI) were assessedon a 0-5 scale 0 = no infection, 1 = 1- 20% of tuber galled, 2 = 21- 40%of tuber galled, 3 = 41-60% tuber galled, 4 = 61- 80% and 5 = 81-100% of tuber galled. The data obtained were subjected to analysis ofvariance (ANOVA). Significant means were separated using DuncanMultiple Range Test (DNMRT)
Determination of protein
The proximate composition of the harvested yam tuber wasdone by the gravimetric method [13]. Yam flour obtained fromharvested yam tubers in pots treated with Basil plant, Bitter leaf,neem, Horseradish and Bush pepper were used for the proximatecomposition study. To obtain the flour, the harvested tubers werepeeled, chipped, dried and milled into flour. Five grams of flour wasmixed with 10 ml of concentrated H2SO4 in a digestion flask, and 1gtablet of selenium was added to the mixture as the catalyst beforeheating under a fume cupboard till a clear solution was obtained. Thedigest was diluted with water to 100 ml in a volumetric flask and usedfor the analysis. Ten ml of the digest was mixed with equal volumeof 45 % NaOH solution in a distillation apparatus and was furtherdistilled into another 10 ml of 4 % boric acid with three drops ofmixed indicator (BromocressolGreen/Methylred). Fifty milliters ofthe distillates was collected and titrated against 0.02N EDTA from green to deep red end point. The protein content of each treated yamtuber was calculated using the formula (6).
% protein = % N2 x 6.25, N2 = (100 x N x 14 vt) T.BW x 1000 x va.
W = weight of treated yam flour, N = normality of titrant (0.02NH2SO4), Vt = total digest volume, Va = volume of digest analyzed, T =sample titrate value, B = blank titrate value.
Ash Determination
Five grams of the yam flour obtained from harvested yam tubersin treated pots were measured into a previously weighed porcelaincrucible and burnt to ashes in a muffle furnace at 550°C. Followingthis the ash was cooled and the weight was determined by thedifferences in weight before and after burning to ash and expressed aspercentage of the sample analyzed.
Fiber Determination
The fiber composition was determined by method described byArora, 1981 [14]. Five grams of the harvested yam tubers in treatedpots were measured and boiled in 150 mL of 1.25 % H2SO4 solutionfor 30 minutes and washed in hot water using a twofold muslin clothto trap the particles. It was returned to the flask and boiled in 150mL of 1.25 % NaOH for 30 minutes. Washing in several portions ofhot water, the dust were allowed to drain dry before weighing in acrucible where it was oven dried at 105°C to a constant weight. Thiswas further burnt in a muffle till only the ashes were left. The weightof the fiber was obtained and expressed as percentage of the weight ofsample analyzed 90 [15-17].
Fat content Determination
Five grams of the yam flour obtained from harvested yam tubersin treated pots were wrapped in filter paper and put in a thimblewhich was placed in soxlet reflux flak and mounted into a previouslyweighed extraction flask containing 200 mL of petrol ether. The upperend of the flask was connected to a water condenser, while the contentswere heated and vaporized. The vapour condensed into the refluxflask, and was converted into the solvent that extracted the oil (fat).The oil extracts were oven dried at 60 °C for 30 minutes to removeany residual solvent. This was further cooled in a desiccator andweighed. The difference in the weight of oil extract was determinedand expressed as a percentage weight of sampled yam flour.
Carbohydrate Determination
Total carbohydrate was calculated by the method described inAOAC [13]. This was done using the formula:
Total carbohydrate = 100 - % protein + fat + fiber + ash + MC.
Results and Discussion
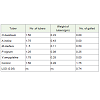
Sprouting counts at 4, 8, and 12 weeks after planting waspresented in Table 1. There was no statistical difference (P > 0.05)between treatments and the control on sprouting. This implied thatnone of the plant leaf powder evaluated had adverse effect on the timethe seed yams sprouted.
Table 1: Effect of five plant leaf powder on sprouting of yam setts at 4, 8, and 12 weeks after planting.
The effect of five plant leaf powder on yam tuber yield and gallformation is given in Table 2. A. indica and V. amygdalinarecordedthe highest number of tubers (1.75) while the untreated control hadsignificant (P>0.05) number of mean galled tubers (1.75). This showsthat the botanicals used in the treatment of seed yams effectivelycontrolled the proliferation of galled tubers which is a visual means ofthe presence of root-knot nematode on yam tubers. This indicates thatthe use of some botanicals (Basil plant, Bitter leaf, neem, Horseradishand Bush pepper) in the production of root-knot free yam tuberscould be a good local substitute to seed dressing with syntheticnematicides in yam production.
The proximate composition of the harvested yam tubers aftertreatment is presented in Table 3. The protein composition of theyam tubers was not affected by the treatments as compared with theuntreated control. There were no significant differences (P > 0.05) onthe protein levels in all the treatments and the control. This was inline with [9] who reported on the effect of botanicals on the proteincomposition of food yam. There were significant differences (P < 0.05)on the carbohydrate composition of the treated yam tubers whencompared with the untreated control. V. amygdalina had the highestlevel of carbohydrate (27.02 mg/100) followed by P. nigrum (25.12mg/100) compared to the control which was the least. The applicationof the leaf powder increased the carbohydrate level of the treatedyams by 10.4%, 21.4%, 8.1%, 11.1 %, and 12.8% in Basil, Bitter leaf,neem, Horseradish and Bush pepper, respectively when comparedwith control. The fat composition of the yam tubers indicated that O.basilicum (0.51 mg/100g) was significantly different (P < 0.05) fromV.amygdalina(0.44 mg/100g) and A. indica (0.41 mg/g). Besides, theleaf powder of bitter leaf, neem, and horseradish increased the fat content by 22%, 13%, and 5.5%, respectively when compared withcontrol. There were no significant differences (P > 0.05) between thefat levels in P. nigrumand M. oleifera. However, all the treatmentswere significantly different (P < 0.05) from the untreated control. Thehighest level of ash content was derived from the tubers pre-treatedwith P. nigrum(3.63 mg/100g), O. basilicum(2.96 mg/100g) and theuntreated control. There were significant differences (P < 0.05) in theash composition among A. indica, V.amygdalina, and M. oleifera.The untreated control and M. oleiferarecorded the highest levels offiber contents in the yam tubers and were significantly different (P< 0.05) from the other treatments. The nutritional compositions ofthe yam tubers were affected by the treatments as compared withuntreated control. Any treatment that inhibits the damaging effect ofnematodes on yam without adversely affecting the nutritive value ofthe yam tubers will be considered as a way forward in organic foodproduction.
The use of botanicals improved the dietary level of the yamtubers as a food security crop in hunger intervention, althoughthe treatments are in high demand as spices, they appear to have ahigher potential in the control of M. incognita and have proved to befavorable alternatives to synthetic pesticides in yam production. Allthe plants used in this study are locally available and used in ethnomedicinal practices without toxicological residue effects and as suchcould serve as alternatives to a sustainable organic farming system tomeet global demand for food production.
A total of 376(38.0%) were recorded in the Monday, while 611(61.9%) were recorded during Thursday exposure (Table 2).
Conclusion
The study provides empirical data confirming the use of botanicals for improved dietary level of yam tubers as a food security crop in hunger intervention, although the treatments are in high demand as spices, they appear to have a higher potential in the control of M.incognita and have proved to be favorable alternatives to syntheticpesticides in yam production. All the plants in this study are locallyused in ethno medicinal practices without toxicological residueeffects and as such could serve as alternatives in a sustainable organicfarming system to meet global demand in food production.
Therefore, the study recommends adoption of the technique byyam farmers in a larger field.
References
- FAO (2005) FAO year book, Rome Italy.
- NAERLS (2009) Annual Agric performance survey Report of Nigeria. National Report 2009.
- IITA (2010) International Institute of Tropical Agriculture (2010).Annual Report, IITA Ibadan, Nigeria.
- Onwueme IC (1978) Tropical root and tuber crops, Cassava, Sweet potato Cocoyam. John Wiley and sons. N.Y 224pp.
- Asiedu R, Wanyera NM, Ng SYC, Ng NQ (1997) Yams In: Biodiversty in Trust. D. Fuccillo, L.Sear & P.Stapleton, Nematrropica 1: 39-40.
- Emehute JK, Ikotun UT, Nwauzor EC, Nwokocha HN (1997) Crop protection. In: Food yams; Advances in Research. GC Orkwor, R Asiedu, IJ Ekanayake. (ed). NRCRI /Nigeria. PP143-186.
- Onyenobi FI (1992) Root knot nematode Meloidogyne. An economic Problem in Nigeria. In: the biology and control of nematode of food crops in Nigeria. Pp 37-48.
- Onyenobi FI, Aghale DN, Okorocha EA (2006) Post -harvest studies of yam pre-treated with bio and synthetic pesticides to control Meloidogyne incognita at cultivation. Proceedings of the 40th Conference of the Agriculture Society of Nigeria, Abia Pg 463-466.
- Aghale DN (2011) Evaluation of Botanicals in the control of Root Knot nematode M. incognita (Kofoid& Whitehead, 1919). Ph.D Thesis, Dept of Crop Production/Protection, Abia State University, Uturu, Nigeria.
- Aghale DN, Okorocha A, Njoku, Nwanyieze (2010) Evaluation of Different Botanicals for root knot nematode control. Journal of Agric Biology & Food Research 3: 23 -240.
- Aghale DN (2011) Efficacy of crude plant extract at different rates of application in the control of root knot nematode (M. incognita) in yam production. International Journal of Agric food & Ecosystem Research 3: 200-215.
- Husssey RS, Barker KB (1973) “A comparison of methods of collecting inocula of Meloidogyne spp., including a new techniqueâ€. Pl Dis Reptr 57: 1025-1028.
- AOAC (1990) Official method of Analysis. 15th Edition, Association of official Analytical Chemist, Washington D.C USA.
- Arora SK (1981) Analytical Technique for quality Evaluation. Forage Research 7: 196-213.
- Nwauzor EC, Fawole B (1985) Root knot nematode Meloidogynesppon edible yam Dioscoreaspp in Nigeria. Effect on tuber yield. 15th Ann Conf. of NSPP, NRCRI, Umudike, March 3-8.
- National Root Crops Research Institute (2005) Annual report, 2005. 260pp.
- SAS (1999) Statistical Analysis System. The SAS system for Windows version 8. SAS Institute Inc. Cary, NC, USA.