Review Article
Plant Genetics A View on the Effect of Laser Irradiation on Cell Division
Norah M Al Aboud*
Department of Biology, Umm Alqura University, Makkah, Saudi Arabia
*Corresponding author: Norah M. Al Aboud Department of Biology, Umm Alqura University, Makkah, Saudi Arabia
E-mail : nmaboud@uqu.edu.sa
Copyright: © Norah M Al Aboud 2023. This is an open access article distributed under the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 10/12/2022; Accepted: 09/01/2023; Published: 11/01/2023
Abstract
The effect of lasers of wave length in the visible region 660-680 nm on mitotic cell divisions, germination and growth were studied in Vicia faba. The
study indicated that lasers could be mutagenic causing aberration in the mitotic cell divisions while also producing changes in germination and growth of the
plant. A wide range of chromosomal aberration were observed in all the four stages of mitotic cell division. The most important stickiness and non disjuction
of changes observed were clumping chromosomes, bridges, laggards, micro nucleate, binucleate and elongated nucleate cells. In addition to these some
other types of aberration such as inter phase with unequal sized nuclei, polyploid cells, chromatin droplets, nuclear bridges, nuclear polymorphism and multi
nucleate conditions were also encountered in low frequencies. Laser exposure at 660-680 nm for 5 min caused increase in germination index in V. faba
while germination index induced by exposures at 660-680 nm for 10 min was similar with control group.The change in plant height over a period of time or
the growth rate of mutagen treated samples was compared with that of the control (untreated) samples. Growth rate induced by 5 min of laser exposure was
the highest one. The study suggests that laser may used as a bio stimulator in agriculture. Further studies are required for elucidating the exact mechanisms
by which lasers cause mutations.
Keywords
Laser irradiation; Viciafaba; Mutagenesis breeding; chromosomal aberration
Introduction
Nature of Mutations:
Mutations are of different types. Mutations produced by changes
in base sequence of genes as a result of base pair transition or trans
version, deletion, duplication or inversion etc. are known as gene
mutations. Those produced by changes in chromosome structure
or even in number are termed as chromosomal mutations. Gross
chromosomal changes such as changes in chromosome number,
translocations, inversions, large deletions and duplications are
detectable under the microscope. In cytoplasmic mutation, the
mutant character shows cytoplasmic or extra nuclear inheritance.
Bud mutations or somatic mutations occur in buds or somatic tissues
which are used for propagation, e.g. in clonal crops.New alleles are rarely produced in induced mutations but they
produce alleles which are already known to occur spontaneously or
may be discovered if an extensive search is made. The effects and the variability produced by induced and spontaneous mutations
are comparable. The great advantage of induced mutations over the
spontaneous ones is that they occur at a relatively higher frequency so
that it is practical to work with them.
Mutations have certain general characteristics such as they are
generally recessive, but dominant mutations also occur, a small
portion (0.1%) are beneficial but a majority of mutations are generally
harmful to the organism, mutations may occur at random in any
gene. However, some genes show higher mutation rates than the
others and the same mutation may occur again and again, that is, they
are recurrent.
Effects of Mutation:
In general, mutations have harmful effects on organisms. The
viability of the individuals that carry mutations is usually reduced.
Mutations are classified into four groups, based on their viability.
They are:Lethal mutations: This type of mutation normally kills all
individuals that carry them. Dominant lethal mutations affect
even heterozygous individuals, while recessive lethals kill only the
individuals which carry them in the homozygous state.
Sub lethal and Sub vital mutations: Do not kill all the individuals
that carry them but reduce the viability. Sub lethals kill more than
50% of the individuals while sub vitals kill much less than 50%. This
type of mutations is of no value although a vast majority are of these
type.
Vital mutations: The viability of the individuals carrying this
type of mutation is not reduced. This class of mutations occur in a
much lower frequency than the other three types, but are the only
ones that can be utilized in crop improvement.
Induction of mutations:
Treatments with certain agents known as mutagens can be used
to induce mutations at relatively higher frequencies. Mutagens
may be different kinds of radiations (physical mutagens), these
include various kinds of radiations such as Ionising radiations and
Non ionizing radiations. Also we can induce mutations by certain
chemicals mutagens (chemical mutagens) such as ethidium bromide
and nitrous acid.Mechanism of action of radiations:
Radiations are direct as well as indirect in their effects. Energy
is transferred directly by the radiation to a molecule in direct effect
but in in direct effect it is mediated by free radical formation : the
radicals transfer their energy to other molecules. The indirect effect
is particularly important in presence of water since ionised water
molecules produce free radicals [1].According to Evans [2] radiation does not produce direct
breakage in chromosomes, but initiates a lesion requiring DNA
synthesis for repair. An exchange would arise as a consequence of
mis repair of the lesions. Revell [3] have stated that all aberration
are a consequence of exchange following a process of mis repair of
primary lesions. Variations in radio sensitivity at different stages of
the cell cycle are due to differences in the time available for repair and
to changes in chromatid structure during chromosomal replication.
Radiations have been extensively utilized for many years to
cause mutations and chromosomal damage for experimental
purposes. They can induce a change in the molecular organisation of
protoplasm. The change maybe expressed as a mutation, a break in
a chromosome, or an alternation in the physiological activity of the
cell [4]. The manner in which the yield of structural changes increases
with increase of the dose of radiation has been extensively studied,
and the results of these studies form the main basis on which theories
of the mechanism of induction of these changes are built [5].
Effect of light in the visible range on pla:
A number of processes in plants such as photosynthesis,
protoplasmic streaming, flower induction, seed germination,
chlorophyll biogenesis, bending of organs and numerous other
growth reactions are affected or controlled by radiant energy. Under
optimum conditions these processes are normal. But variations above a certain range induces stress symptoms in plant [6]. Radiation
between 510 and 610nm (green yellow) has minimal effect on plant
growth where as radiation between 400 and 510 nm (blue) will have
the following effects - chlorophyll and other photosynthetic pigments
such as phycocyanin, phycoerythrin and carote in have peaks in
absorption in these wavelengths. Phototropic movements of plants
are promoted by absorption of radiant energy of these wavelengths.
It has been thought that visible radiation will not be mutagenic since
most of the biological molecules have optical absorption in the UV
region. Since the discovery of laser in 1960, the idea that the visible
light may be mutagenic has been mooted. Putative mutagenicity has
been attributed to the coherence and intensity of laser beams, due to
which it can interact with bio molecules in anon-linear way.Modern laser techniques provide a wide range of variation of
radiation parameters such as frequency, intensity and pulse duration
thus making it possible to carry out investigations on selective action
on substances. Molecules or part of molecules of the same type may
undergo considerable change caused by photo ionisation or photo
dissociation with sub sequent chemical reactions.
Objectives of the present study:
Genetic improvement of crops is dependent on the availability of
genetic variability. Sources to induce such variability include the use
of physical and chemical mutagens, tissue culture etc. Although lasers
have recently been suggested to be useful in inducing mutations,
their use has been limited in the absence of any systematic study to
establish their precise mutagenic nature. In recognition of this, the
specific objectives of the present study were as follows
1. T o assess the efficiency and effectiveness of laser ray mutagens
in faba bean.
2. T o study the effect of laser radiation in inducing chromosomal
aberration during mitotic cell division by analysing the root
tip squashes of Vicia faba.
3. To study the effect of laser radiation on mitotic index.
4. To study the effect of laser radiation on Germination and
growth.Review of Literature
Mutation Research in Plant Breeding:
In mutation breeding, mutants are produced by inducing
variations using either chemical or physical mutagens. The methods
of experimental mutation research are utilised in plant breeding
since about 40 years. Induced mutations in crop plants contribute by
increasing genetic variability.Effective treatments are essential for economical use of
mutagensas tools for the induction of heritable changes in qualitative
and quantitative characters of crop plants. A number of useful
varieties of food crops and ornamentals have been developed by
making use of mutations [7].
Both epigenetic and genetic changes have been found to be useful
in plant improvement. Epigenetic changes increasing growth, yield
or secondary metabolite production in vegetatively propagated crops and genetic mutations affecting increased yield, stress tolerance,
disease resistance, protein quantity or quality, etc. are of use in crop
plants.
Mutagenic effectiveness and efficiency:
“Effectiveness” is a measure of gene mutation in relation to dose
and “Efficiency” is an estimate of biological effects induced such as,
lethality, injury and sterility [8]. According to Blixt [9] the sensitivity
of an organism depends up on the mutagen employed and its genetic
makeup. The usefulness of any mutagen depends upon not only its
effectiveness but also to a large extent upon its efficiency. Effective
mutagenesis is brought about by the production of useful mutation
with minimum undesirable changes.The different response of varieties to different mutagens have
been reported by Prasad and Das [10,11]. The type of mutagens, plant
genotype and the physical state of the organism are considered to be
important factors which contribute to the difference in frequency and
spectrum of induced mutations [10,1113]. The difference in mode of
action of mutagen [14] differential penetration of the mutagen to the
target [15] efficiency of repair process [16]. and factors affecting the
expression of concerned mutation [17] might also be playing a role in
inducing mutations‘with varying frequency and spectrum. [18] and
[19] are of the opinion that the difference in mutagenic effectiveness
and efficiency are due to the amount of DNA and its replication time
in the initial stages. It might be due to the physiological stage of the
cell, ability to repair the damage or several other physical factors
[20,21,17,22,23].
Index in determining the biological effects of various mutagens:
Germination, survival and seedling growth are widely used as
indices in determining the biological effects of various mutagens. The
reduction of these parameters were prominent in EMS treatments
either alone or in combination. Such an inhibitory effect of various
mutagens was reported in several crops [24-26]. Reduction of these
parameters has been attributed to various factors including changes
in the balance of growth regulators and metabolic activity [27]
physiological changes including inhibition of DNA synthesis [28]
or inhibition of mitotic proliferation [29] Increase in seedling injury
could be due to mitotic irregularities [30]. Sterility in pollen is mainly
due to interchanges between nonhomologous
chromosomes and
detectable chromosomal aberration [31].Chromosomal aberration studies:
One of the oldest, simplest and least expensive methods for
studying the induction of chromosomal aberration utilises plant root
tips as experimental material. Of all the plants, where root tip mitosis
has been studied for induced chromosomal aberration, only few, for
instance Vicia faba, A. cepa, A. proliferum, A. fistulosum have proved
to be favourable materials [2,32-35]. The suitability of these plants for
cytological studies has been attributed to their large chromosomes,
low chromosome number, supplemented by easier cultivation and
availability throughout the year. The merits of these materials were
realised by radiobiologists in 1930’s [33]. Several types of studies have been done in this field so far for instance, [36] have studied the
molecular mechanism in the production of chromosomal aberration
with the 5 - Bromo deoxyuridine labelling method in V. faba while
[37] studied the localisation of chemically induced chromosomal
aberration in three different karyo types of V. faba.Cytogenetics and mutational effects of laser radiation:
Laser irradiation with the wavelength equal to 337 nm on
Alliumfistulosum and Hordeum vulgare cells causes an appearance
of chromosome aberration. In the presence of chromophores, the
cytogenetic effect may be explained by direct effect of laser irradiation
on chromatin DNA. Such a possibility has been demonstrated in
experiments with pBR 322 DNA in the presence of ethidium bromide
and riboflavin. Chromophores absorb the energy of laser irradiation
according to two-quantum mechanism. The following energy
migration from donor (chromosphore) to acceptor (DNA) produces
breakage of phosphodiester bonds [38] carried out investigations for
the purpose of assessing the mutagenic effect of laser irradiation of
cvs Auralia and Doukat. The treated seeds were dry, soaked in tap
water (15-18 hours) or soaked in a solution of the stain Rhodamine
B(RhB) pea seeds. Various doses of helium neon laser (lambda - 631.8
nm) andargon laser (lamda -457.9, 488 and 514 nm) were applied.
The experimental data showed cytogenetic effect of laser irradiation
depending on doses,wavelength, metabolic state and cultivar of the
seeds. Dry seed irradiation ofcv. Auralia with helium-neon laser
produced higher percentage of mutation changes at doses 0.43 and
1.72 J/cm-2. The spectrum was wider at does 1.28and 1.72 J/cm-2.
Irradiation effect was higher on seeds soaked in tap water and highest
in seeds soaked in the RhB stain. Mutation frequency increased with
the rise of dose and the spectrum was wider at doses 0.86 and 1.28 J/
cm-2. In cv. Douk at the effect of helium - neon irradiation was slightly
expressed. Argonlaser irradiation with lambda - 488 nm produced
higher effect on cv. Auralia dry seeds at the higher doses 20 and
26.74 J/cm-2. cv. Auralia proved more sensitive to this treatment and
manifested higher mutability. Electro phoretic analysis of per oxidase
showed that cv. Auralia reacted faster to the applied irradiation
(lambda 488 nm).[40] conducted a study to determine the laser irradiation effect on
Gossypium seeds. Pre sowing irradiation of seeds had a stimulating
effect on M1 plants. The subsequent generations revealed a who
lerange of mutations typical of Gossypium. The number of mutants
depended on the irradiation intensity and some of them can be used
for breeding.
Effect of lasers on germination and growth:
Influence of laser beam of three different wavelengths - 337.1nm,
510 nm and 632.8 nm on germinating maize seeds was carried out to
study some metabolic process in seedlings[41]. The results showed
that during the period of investigation (1-6 days), the laser irradiation
of 632.8 and 510 nm wavelengths performed in the 24th hour of
germination did not modify the protein content of either the embryo
or the endosperm,compared with control seeds. Whereas, the light
of 337.1 nm increased the soluble protein content in the embryo,
depending on the degree of dose. RNA and DNA contents were not
modified by any of these irradiations.[42] used laser irradiation of rated power density (about 5
mW cm’2 and a wavelength lambda = 632.8 nm) to improve the
of propagation of false acacia forms (Robina pseudoacacia L). An
additional irradiation of the upper two or three axillary buds of the
cutting not only improved the rooting rate, but also increased the
numbers of the adventitious roots and of the root hairs.
In 1986-1988 the effect of seed irradiation by laser on the
vegetative and reproductive manifestations of the plants of the small
fruit cultivar and pobeda cucumbers was investigated. Irradiation was
performed with helium - neon laser of 632 nm wavelength and 20
mw power at the output, and with variants of one, three, five, seven
and nine times. The strongest stimulation effect was obtained with
five and seven - fold irradiation .with these variants the plants formed
over ground vegetative mass by 10.1 and15% higher and leaf surface
by 25.3 and 28% higher. The higher standard yield fruits are of a
length from 3 upto 12 cm) of 20.213 tons/ha (exceeding the control
by 16%), was obtained at seven - fold irradiation. The seed irradiation
increased the content of dry mater, total sugars and vitamin C in the
fruits and plastid pigments in their skin [43].
The effect of nitrogen laser (337.1 nm) and argon ion laser
514.5nm) irradiation on physiological response in the green gram
Vigna radiata L. seedlings was studied by [44]. The shoot and root
lengths and fresh dry weights of the seedlings increased with 30 min
exposure to nitrogen laser and 5 min exposure to argon ion laser.
Protein content was maximum with 20 min exposure to N laser and
5 min exposure to argon iron laser, while RNA and DNA contents
were maximum at 5 min exposure with oth the laser treatments. [45]
reported the results of pot and field experiments related to studies of
the influence of laser irradiation on winter wheat, spring earley and pea
over the years 1986-1988 using the LA 1001 Ne-He laser and coherent
laser beam, at 632.8 nm wavelength, has a biologically stimulant
influence resulting in increased emergence velocity as well as in the
related dynamics of the beginning of growth. Laser irradiation had no
effect upon yields of the studied species and no varietal dependence
was found. It is assumed interaction with that the described effects
of laser irradiation occur in deteriorated environmental conditions.
Experiments were conducted in 1987-1988 by [46] with alfalfa
and a grass mixture (alfalfa, red clover and burr reed) in pots with a
capacity of 14 Kg dry soil. Single, repeated and triple seed irradiation
with helium - neon laser wavelength 632.8 nm was tested. It was
found that pre-seeding laser irradiation of the seeds increased by
85%, the number of alfalfa stems and up to 66% of grass mixture
stems as compared to the control. Laser irradiation had no significant
effect on plant height and leaf area. As a result of laser treatment
accumulation of dry matter increased 27.3% in alfalfa and 16.1% in
the grass mixtures. Single laser treatment of the seeds proved more
efficient, it increased the root mass of alfalfa (34.5%) and of the grass
mixture (17.9%) at the end of the experiments as compared to the
control.
Materials and Methods
Materials:
Taxonomically diverse plant species suitable for cytological analysis, namely Vicia faba (faba bean) belonging to the family
Leguminosae,sub family Fabaceae (family Leguminosae), was
selected as the experimental material. Seeds of V. faba were obtained
from local market.Irradiation of samples by physical mutagens:
The treatment included Laser irradiation at 660-680 nm (viz.
5 min exposures at power density 5 mW and 10 min exposure at
the same power density). Laser source (5mW) was used for the
irradiation V. faba seeds were soaked over night in tap water and
decoated before irradiation. Seeds were divide into three groups after
soaking, the first group was exposed to 5mW of laser radiation for
5 minute, the second group was also exposed to the same power for
10 minute and the therd group was the control group. Immediately
after the various treatments, the faba bean germinated on moist filter
paper in petridishes at 25+ 2°C. Faba bean root tips of the same size
were collected between 10 and 11 A.M after 3-5 days of treatments.Fixation of root tips:
The root tips were fixed in a mixture of 3 parts of absolute alcohol
and 1 part of glacial acetic acid. Fixative was prepared fresh every time.
Root tips could be kept in the fixative up to 15 days in the refrigerator.Preparation of root tip squashes:
From the fixative, the roots were transferred to distilled water and
were washed twice. They were then hydrolysed in 1N Hydrochloric
acid at about 60°C for a few seconds. After hydrolysis, the root tips
were washed twice in distilled water and then transferred to distilled
water. On a clean slide, the tips were separated from the rest of the
root and crushed in a drop of 2% acetocarmine with the flat end of a
rod and squashed under a cover slip. The pressure was applied under
several thickness of blotting paper.Scoring of slides:
For scoring of cytological aberration, temporary slides were used.
Atleast 3 slides were prepared from actively dividing root tips in each
dose and 9 fields (approximately 50-70 cells) were scored. Different
structural changes of chromosomes were scored at metaphase and
anaphase. Micronuclei were scored at anaphase/telophase. Savage’s
(1975) classification of various types of chromosomal aberration
was used for scoring the aberration. The percentage of prophase,
metaphase, anaphase and telophase were calculated, also Mitotic
index (M.I.) and total aberration were calculated as below:Mitotic index M.I. % =Number of dividing cells /Total No. of
cells× 100
Total abnormality % =Total No. of abnormal cells /Total No. of
dividing cells× 100
Germination/Sprouting index studies:
Germination/sprouting index was monitored in treatment of the
mutagen used in this study, the germination/sprouting index was
computed using the equation:Germination index =No. of seedsshowing seedling(sprouting) /
Total No. of seeds
Results
Mitotic chromosome complement of the materials:
The mitotic chromosome complement of V. faba contains
12chromosomes which could be classified into five pairs of
nearly equally long chromosomes with sub terminal centromeres
(chromosomes II - VI, also referred to as the S chromosomes) and
one pair with median contromere(chromosomes I, also called the M
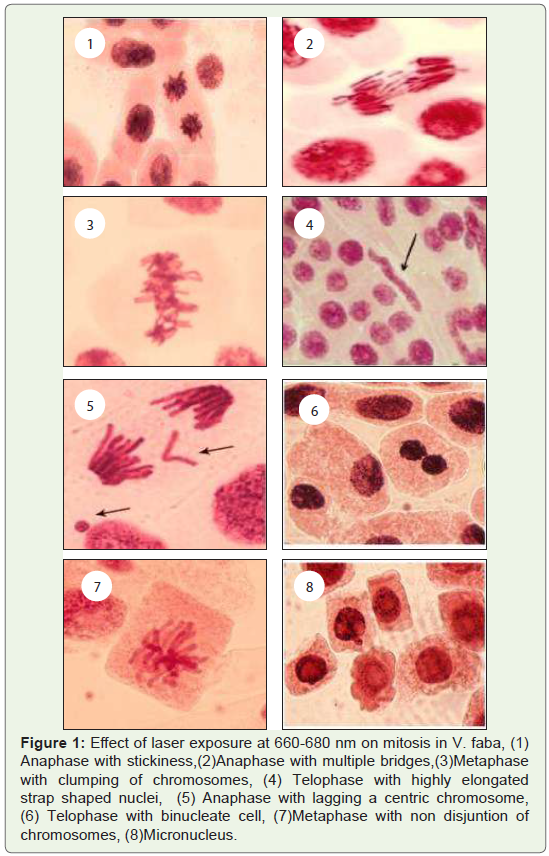
chromosome).Mitotic chromosomal aberration:
In control (untreated) samples mitosis was normal with only
0.17% of aberration observed. In treated samples, a wide range of
chromosomal aberration were observed in all the four stages of mitotic
cell division. The most important stickiness and non disjuction of
changes observed were clumping chromosomes, bridges, laggards,
micro nucleate, binucleate and elongated nucleate cells (Figure 1). In
addition to these some other types of aberration such as inter phase
with unequal sized nuclei, polyploid cells, chromatin droplets, nuclear
bridges, nuclear polymorphism and multi nulceate conditions were
also encountered in low frequencies.
Figure 1: Effect of laser exposure at 660-680 nm on mitosis in V. faba, (1)
Anaphase with stickiness,(2)Anaphase with multiple bridges,(3)Metaphase
with clumping of chromosomes, (4) Telophase with highly elongated
strap shaped nuclei, (5) Anaphase with lagging a centric chromosome,
(6) Telophase with binucleate cell, (7)Metaphase with non disjuntion of
chromosomes, (8)Micronucleus.
Prophase aberration:
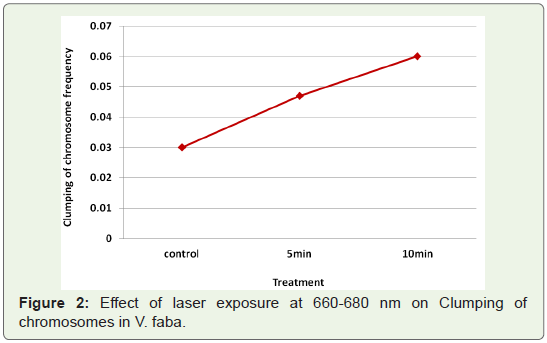
Clumping of chromosomes: The most important type of
chromosomal aberration observed at prophase was clumping of
chromosomes (Table 1). The frequency of this change was very low
(0.03%) in control (untreated) samples. Laser exposures at 660-680
nm (5 mW) induced an increase in clumping of chromosomes with
increase in exposure time (Figure 2).
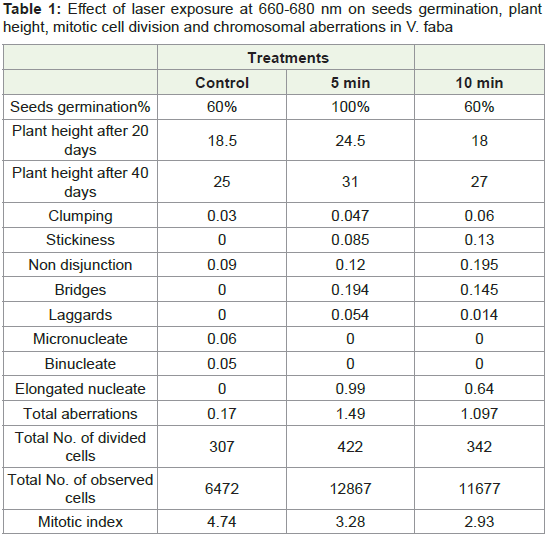
Table 1: Effect of laser exposure at 660-680 nm on seeds germination, plant
height, mitotic cell division and chromosomal aberrations in V. faba
Metaphase aberration:
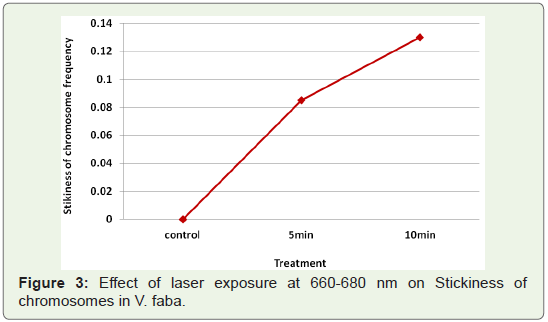
Stickiness of chromosomes: The most common type of
chromosomal aberration observed at metaphase was stickiness of
chromosomes (Table 1). It was totally absent in the control but was
encountered in all the mutagen treatments. Laser exposure at 660-680
nm (5 mW) induced an increase in stickiness of chromosomes with
increase in exposure time (Figure 3).
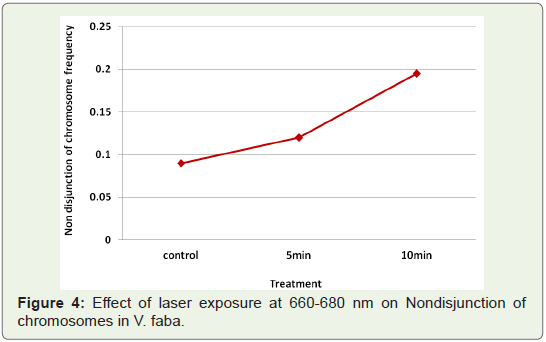
Nondisjunction of chromosomes: Another common type
of aberration observed at metaphase was non disjunction of
chromosomes (Table 1). The frequency of this change was very low
in control (untreated) samples (0.09%). Laser exposures at 660-680 (Figure 4).
Anaphase aberration:
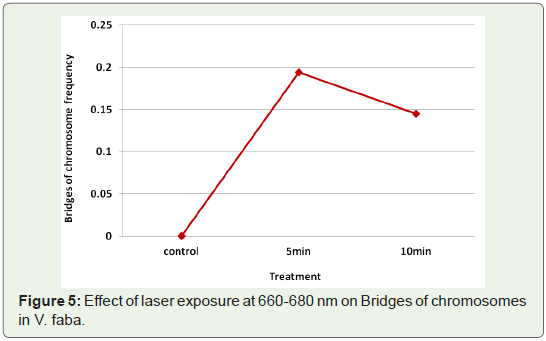
Bridges: The common type of aberration observed at anaphase
were chromosome bridges (Table 1). Bridges were not found in
control samples, while no time dependence could be established for
the change in bridge frequency caused by laser exposures at 660-680
nm (Figure 5).
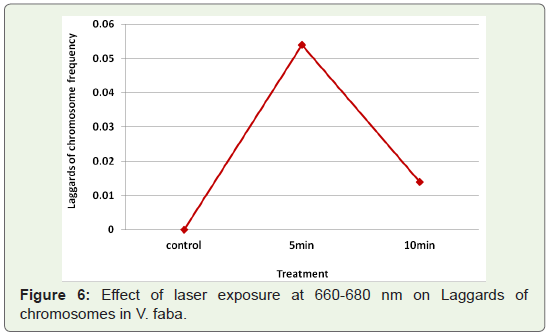
Laggards: Another type of aberration found at anaphase in low
frequencies were chromosomal Iaggards (Table 1). This was not
found in controls, and no time dependence could be established for
the change in bridge frequency caused by laser exposures at 660-680
nm (Figure 6).
Telophase aberration:
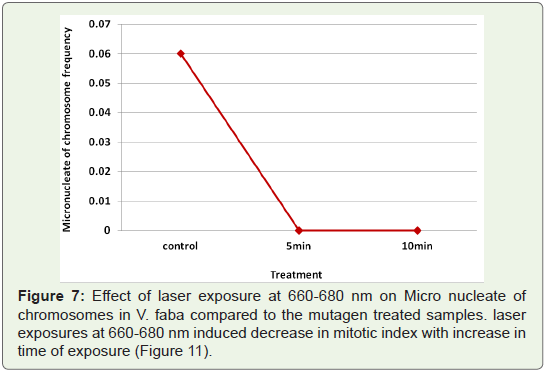
Micronucleate cells: One of the aberration observed at telophase
was cells with micronuclei (Figure 7). It was seen in low frequency in
the untreated controls.
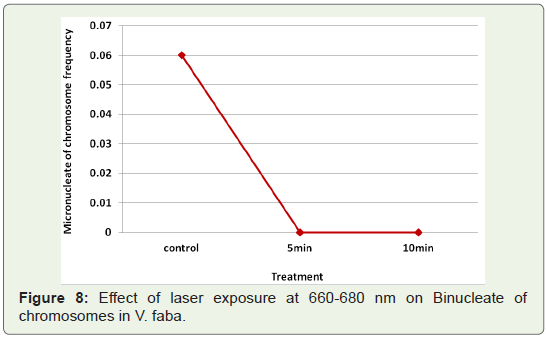
Figure 7: Effect of laser exposure at 660-680 nm on Micro nucleate of
chromosomes in V. faba compared to the mutagen treated samples. laser
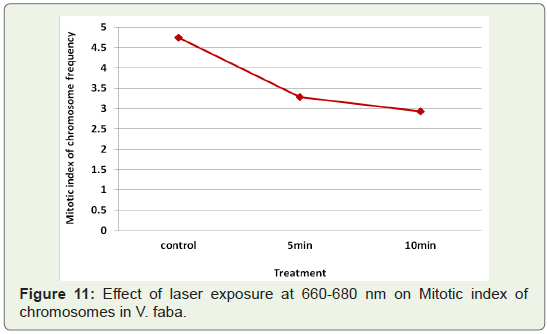
exposures at 660-680 nm induced decrease in mitotic index with increase in
time of exposure (Figure 11).
Binucleate cells: Another type of aberration commonly found in
most of the cases at telophase was the formation of binucleate cells
(Figure 8). It was seen in low frequency in the untreated controls.
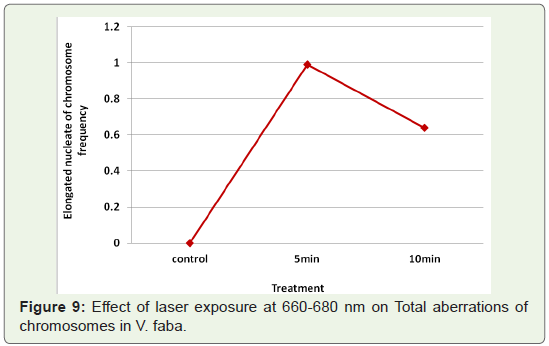
Elongated nucleate cells: The presence of cells with highly
elongated nuclei were another aberration seen at telophase (Table 1). It was absent in controls, laser exposures at 660-680 nm induced increase in the frequency of the formation of elongated nuclei at the
minimum exposure time (Figure 9).
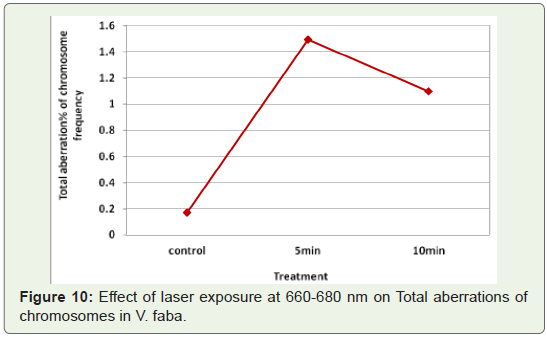
Percentage of total mitotic aberration:
The sum total of the different types of aberration induced by the
mutagen treatments in four stages of mitotic cell division was used
to determine the percentage of aberration. Laser exposure at 660-680
nm induced increase in total aberration frequency at the minimum
exposure time (Figure 10).Mitotic index:
Mitotic index is used as a measure to denote the number of cells
undergoing mitotic cell division. Mitotic index was found to be
highest in control as (Figure 11).Germination/sprouting index:
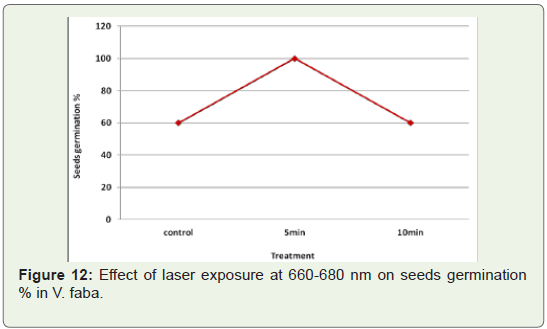
Laser exposure at 660-680 nm for 5 min caused increase in
germination index in V. faba. While germination index induced by
exposures at 660-680 nm for 10 min was similar with control group
(Figure 12,13).Growth:
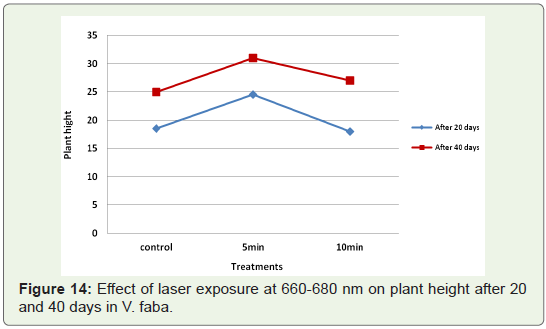
The change in plant height over a period of time or the growth
rate of mutagen treated samples was compared with that of the
control (untreated) samples (Figure 14,15).Growth rate induced by 5
min of laser exposure was the highest one.Discussion
Induced mutagenesis has been recently recognized as the
most efficient tool for improving nutritional properties. Induced
mutagenesis has played a significant role in successful development of
new varieties by upgrading a specific trait without altering the original
genetic makeup of the cultivar[47]. A single plant can contain a large
number of different mutations resulting a manageable population
sizes [48].
As per the reports of FAO, 2017 there are about 20 mutant
varieties developed to date and the recently released mutant variety,
Geca5, has improved attributes including good quality and early
maturity.
Mutagenic effectiveness and efficiency have much importance
in mutation breeding experiments. The measure of frequency
of mutations induced by unit dose of mutagen is considered as
mutagenic effectiveness; whereas the mutagenic efficiency represents
the mutations on the basis of biological damage i.e. sterility, injury
and lethality. In mutation breeding programmes, the mutagenic
effectiveness and efficiency are necessary for obtaining desirable
mutations in plants [49]. Mutagenic effectiveness and efficiency
depends upon the type of genotype used and the mutagen applied
on it.
Different workers have reported different effectiveness and
efficiency of mutagens on different plants, e.g. grass pea [50,51]
lentil [52,53] clusterbean [54] chickpea [55] black cumin [56] cowpea
[57,58] garden pea [59] and mungbean [60]. Recently, laser irradiation
has also been employed to meet agricultural demands and attain the
goal of sustainable development [61,62].
Lasers of 5 mW (with wavelengths in the visible region 660-680
nm) caused aberration in the early stages of mitotic cell division,
the early stages of mitotic cell division are prophase and metaphase.
The most important type of chromosomal aberration observed at
prophase was clumping of chromosomes and at metaphase was
stickiness and non disjunction of chromosomes. According to [63]
the clumping of chromosomes may be attributed to the inhibition of
protein synthesis.
Stickiness and non disjunction are due to disruption of bonds
between protein and nucleic acid constituents or physical adhesion
of proteinaceous matrix resulting in failure of chromosome
condensation in prophase [4,63,64]. According to [65] stickiness
is the result of the breaks and chromosome exchanges during prophase contraction. Apparently lasers affect the process of normal
chromosome condensation.
Laser also apparently cause chromosome breaks, which are
usually formed as a result of breaks in each arm of two adjacent
chromosomes and their reunion was the most commonly observed
type of stickiness. Multiple kinetochore chromosomes such as
tricentric have been rarely observed, which apparently arise from
breaks in more than two chromosomes. Polyploid cells were also
observed rarely at metaphase. The spindle abnormality, polyploidy
like ‘regular orientation and scattered distribution of chromosomes
at metaphase due to non synchronisation of division at centromere.
Thus distribution of chromosomes is more confined to the periphery
of the cell.
The later stages of mitotic cell division are anaphase and
telophase. The aberration commonly seen at anaphase were bridges
and Laggards and at telophase were micronucleate, binucleate and
elongated nucleate cells. Bridges probably result from laser induced
stickiness and bridge breakage fusion cycle [4]. In very rare cases star
shaped anaphase was also observed.
Another type of aberration noticed at anaphase was Laggards.
Those chromosomes which are not taking part in bridge formation
sometimes may get detached from the group and are seen lagging in
the cell. This aberration was induced in very low frequency in laser
treated samples. The frequency of Laggards was higher in samples that
exposed to 5 min than those exposed to 10 min. One of the aberration
observed at telophase was cells with micronuclei, it was seen in low
frequency in the untreated controls.
Another common aberration seen in telophase is the formation
of binucleate cells. It was seen in low frequency in the untreated
controls.The formation of binucleate cells is attributed to the absence
of cytokinesis [66] which may be due to the inhibition of cell plate
formation. The continuous fiber forms the spindle of cytokinesis
upon which the cell plate formation occurs.
According to [67] changes in nucleic acid and protein synthesis
changes cell volume or nucleus volume in mammalian cells.
In addition to these changes, The increased exposure time of
treatments resulted in chromatin droplets in some cells and very rarely
nuclear polymorphism and nuclear bridges were also encountered in
some treatments. [66] suggested the term pseudonuclei for chromatin
dropletes resulted due to chromatin disintegration which remains
scattered in the cytoplasm.
The total mitotic aberration induced by laser exposures was
maximum at 5 min. Differences in the cytogenetic and mutational
effects of different wavelengths of laser irradiation has been reported
by several workers. For instance, [40] reported the dependence of
mutation frequency Gossypium seeds on the irradiation intensity and
[68] reported differences in mutation frequency in wheat dependent
on time of irradiation with CO2 lasers.
Previous study by Abdel-Fatah (2005) mentioned that moisture
improved the effect of laser treatments on seed germination. The
pre-sowing seed treatment with laser radiation stimulates the
physiological and biochemical changes in the seed [69]. The treated seeds with leaser light can be applied to improve seed germination
[70,71].Similar results was observed on amaranth plant by [72]
they mentioned that pre-sowing of seeds with laser light resulted
in a significant increase of germination. Also, [73] reported that the
germination of He-Ne laser irradiated seeds significantly increased as
compared with control.
Many researchers reported that the stimulation effects of laser
light are visible in sprouting seeds [74,75]. The laser light has a
positive influence on enzyme activity and the concentration of free
radicals in seeds [76,77,78] observed increasing in the activity of
some phytohormones, especially indole-3-acetic acid (IAA) in the
irradiated seeds.
Pre-sowing irradiation of seeds with laser light results in a
faster uptake of water and achieving the larger mass during seed
imbibing [79]. Also, amylolitic enzymes were increased in seeds
after irradiation of the seeds with laser light improved germination
[80]. Optimal doses of He-Ne laser irradiation may enhance FGP of
Negilla sativa. Pre-sowing laser of seeds has imposed beneficial effects
on germination, seedling growth and yield of various crops [62].
Laser irradiations had a stimulatory effect on growth and yield in
faba. Growth rate induced by laser exposure of 5 min was the highest.
Mutations affecting the plant height have been reported by several
workers [81,82,83,84,85]. The plant height in mutants was reported to
be affected by internodal length and alternations in number of nodes
by Weber and [86] Gottschalk (1973) while [87] Bloustein and Gale
(1984) proposed that the inter nodal length was probably affected by
cell number, cell length or both.
Reduced growth in mutagen treatments due to auxin destruction,
changes in ascorbic acid content and physiological and biochemical
disturbances was noticed by several workers [88-91]. Chromosome
breakage during mitotic inhibition [92] and the effect of the mutagens
on the metabolism of the organism [93] could be the reason for
retarded plant growth, sterility and death in higher doses of mutagen
treatments.
References
Citation
Norah M Al Aboud. Plant Genetics A View on the Effect of Laser Irradiation on Cell Division. J Plant Sci Res. 2023;10(1): 237