A Protocol for the Control of Spongy Tissue Disorder in Alphonso Mango by Pre-HarvestTreatments
Shivashankar S* and Sumathi M
Corresponding author: Shivashankar S. Indian Institute of Horticultural Research, Bangalore-560089, India,; E-mail: drsshivashankariihr@yahoo.co.in
Division of Plant Physiology and Biochemistry, Indian Institute of Horticultural Research, Hesaraghatta Lake P.O., Bangalore560089, India
Citation: Shivashankar S, Sumathi M. A Protocol for the Control of Spongy Tissue Disorder in Alphonso Mango by Pre-Harvest Treatments. J Plant Sci Res. 2014;1(4): 114.
Copyright © 2014 Shivashankar S et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Plant Science & Research | ISSN: 2349-2805 | Volume: 1, Issue: 4
Submission: 01/10/2014; Accepted: 22/12/2014; Published: 23/12/2014
Reviewed & Approved by: Dr. Mohar Singh, Scientist, Division of Germplasm Evaluation, National Bureau of Plant Genetic Resources, India
Abstract
Spongy tissue (ST) of Alphonso mango is the most important physiological disorder which causes grave loss of fruit quality in ripe fruits without showing external symptoms. Until recently, there was no remedy to overcome the malady, as there were speculations regarding the nature of its origin. However, following the crucial findings that the disorder arises due to the premature onset of germinationassociated events in the seed of the developing fruit, field experiments were initiated to develop a protocol to treat the disorder. Results clearly showed that application of salt solutions as a pre-harvest spray between 60 and 70% fruit maturity gave significant reduction in the incidence of the malady. Among the treatments, application of nutrient mix containing a mixture of macro and micronutrients and sea water produced fruits with an ST incidence level of < 5%. Analysis of data revealed that the osmotic effect generated by hypertonic salt solutions was primarily responsible for withholding the movement of water from mesocarp to seed thus preventing the onset of germination-associated events and consequently a reduced incidence of spongy tissue. Pre-harvest application of the nutrient mix containing macro and micronutrients or sea water on developing fruits between 60 and 70% maturity stage effectively reduced the incidence of ST to < 5% compared to 54.3% in control, besides improving the shelf life and fruit quality. The study also demonstrated that treatment of fruits before reaching 60% maturity or after crossing 70% maturity was not as effective in reducing ST incidence. The method offers an easy, effective, economical and eco-friendly solution to overcome the dreaded disorder at the field level thus allowing farmers to produce superior quality Alphonso fruits free from the malady.
Keywords: Control of spongy tissue; Alphonso mango; Preharvest treatment
Introduction
Spongy tissue (ST) of Alphonso mango fruit is a serious physiological disorder in which the edible quality of the ripe fruit is severely affected making it unfit for consumption at the advanced stage of the malady. The problem is of serious concern as the consignment consisting of spongy tissue affected fruits is liable to be rejected at the destination by the importing country causing huge losses to exporters. However, as far as the domestic market is concerned, the consumers are the worst hit since fruits with spongy tissue do not show external symptoms even in the advanced stage of the disorder. Due to the high rate of incidence of spongy tissue in mature fruits exported from India, many countries including the US and Europe have not been showing interest in importing the fruit from India, in spite of the fact that the internal demand for Alphonso fruit in those countries is considerable and is on the rise with each passing year. The principal reason for our failure in addressing this major concern of exporters in the past was due to the fact that the underlying cause of the disorder had not been understood even though spongy tissue disorder was identified and described as early as 1934 and as suchthere was no definite cure for the malady.
The insights gained over the last few years on the mechanism of development of the disorder have helped unravel the mystery surrounding the spongy tissue disorder which had defied an answer for long [1]. The fortuitous discovery that pre-germination associated events in the developing seed play the central role in theorigin of the disorder gave precious insights into the mechanism of development of the disorder. The detailed understanding of the physiological processes leading to the formation of spongy tissue and the critical stage of fruit growth at which the developing seed slipsinto germination phase have provided invaluable data with regard to the possibility of prevention of the incidence of the disorder by appropriate interventions during fruit growth. The crucial finding in our studies which revealed that the incidence of spongy tissue could be brought down significantly by application of paclobutrazol (PBZ)to developing fruits around 60-70% maturity gave strong indications with regard to the possible control of the disorder by pre-harvest sprays [2]. However, the suspected residual toxicity of PBZ in the harvested fruits [3] posed a serious limitation in extending the processfor field control. Keeping this in view, experiments were designed to develop a pre-harvest spray schedule using non-toxic chemicals for the prevention of the malady. The results of the study which clearly showed that the spongy tissue disorder is preventable are presented in this paper.
Materials and Methods
Plant material
Adult trees of Alphonso mango (25- year-old) grown in experimental orchards of the Indian Institute of Horticultural Research, Hessaraghatta, Bangalore constituted the material for the study. Trees showing profuse flowering and good fruit set during Feb-Mar were selected for the experiments.
Fruit treatments
Developing fruits on the tree from 60% -70% maturity (65-75 days after fruit set) were dipped in the following experimental treatment solutions, namely (a) sodium chloride, 3% and 5% (b) calcium chloride, 2% and 3% (c) potassium phosphate, 3% (d) nutrient mix ( Table 3) and (e) sea water along with a non-ionic adjuvant (APSA-80) @ 0.03% for 30 seconds. Water with adjuvant acted as control. In order to determine the critical stage of maturity for pre-treatment, fruits at 50,60,70,80 and 90% maturity levels were treated with nutrient mix and tested for ST incidence at full maturity.
Scoring for spongy tissue incidence
Fruits were harvested at maturity and ripened at an ambient temperature of 25 °C± 2 °C and RH of 70±5%. The percent incidence of spongy tissue was determined based on visual scoring of 100 ripe fruits from each treatment after cutting them open.
Biochemical analyses
Total carotenoids in the pulp were extracted in 80% acetone estimated spectrophotometrically by recording the absorbance of the clear extract at 470, 644.8 and 661.6 nm against the pure solvent as blank [4]. Total soluble solids (TSS) in the pulp was measured using arefractometer and expressed as % sucrose.
Statistical analysis
The experiment was laid out in a completely random block design with five replications (single tree replications). Data were subjected to analysis of variance (ANOVA) using the MSTAT statistical programme (Michigan State University, East Lansing, MI, USA) andare presented as means ± standard error (SE). Differences in fruit quality characters and shelf life between control and nutrient mix treated samples were tested by student’s t- test.
Results and Discussion
The incidence of spongy tissue was found to reduce significantly from 54.3% in control to 12.8% and 4.2% in fruits treated with 1g or 3g/l of PBZ respectively (Figure 1). On the contrary, pre-harvest application of gibberellic acid (GA3) to developing fruits was found to enhance seed germination with a proportionate increase in the rate of spongy tissue incidence [2]. Spongy tissue in Alphonso mango occurred from an increased metabolic activity of seed due to the onset of germination-associated events in the ripening fruit [5]. Since PBZ is an inhibitor of the biosynthesis of gibberellins and promotes the production of the hormone abscisic acid leading to inhibition of seed germination [6], it was obvious that PBZ reduced the spongy tissue incidence by the down-regulation of seed metabolic activity. Thus, the present results confirmed our previous findings and firmly established that spongy tissue incidence in Alphonso mango is regulated by the activity of seed. However, due to the suspected residual toxicity effects of PBZ on humans [3] and its influence on physiological processes in mango, application of PBZ for field control of spongy tissue is not considered the preferred option.
Figure 1: Effect of application of paclobutrazol (PBZ) on the incidence (%) ofspongy tissue in Alphonso mango.
Studies were therefore, conducted to delay seed germination by using inorganic salts which are known to induce seed dormancy both in glycophytes and halophytes [7,8] and inhibit germination in a reversible manner. Abdallah Atia et al [9] showed that salt concentrations exceeding 50 mM NaCl delayed and reduced seed germination in sea fennel (Crithmum maritimum L.) without altering seed viability, These authors further reported that salt-induced stress could influence and/or block some physiological processes and concluded that reversibility of inhibition of salt-induced germination indicated that salt solutions may inhibit the germination mostly via an osmotic effect preventing the imbibition phase to occur.
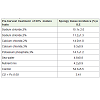
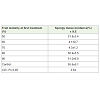
Pre-harvest treatment of developing fruits at 60% maturity with sodium chloride solution of 5% and 3% w/v concentrations significantly reduced spongy tissue incidence ranging from 11.2-15.1% respectively compared to 54.3% in control (Table 1). However,these fruits exhibited surface shrinkage and early ripening symptoms on the tree. In view of this, experiments were conducted with salt solutions containing CaCl2, potassium phosphate, mixture of macro and micronutrients and sea water. It was interesting to note that there was a significant reduction in the spongy tissue incidence in all these salt treatments compared to control (Table 2).The lowest incidence of ST was obtained by treatment with nutrient mix (4.2%) closely followed by treatment with sea water (4.8%) coupled with better fruit quality characteristics in terms of TSS, shelf life and pulp color in ripe fruits (Table 3). It was also apparent from the resultspresented in Table 4 that pre-harvest treatment of fruits between 60 and 70% maturity was most effective in reducing the ST incidence while treatment before 60% or after 70% fruit maturity was not as effective in reducing the ST incidence in fruits (Table 4).
Previous work by Cracknell Torres and Sauco [10] and Young et al [11] had shown that CaCl2 was used to control internal breakdown. Cracknell Torres et al [12] showed that the incidence of internal fruit breakdown was significantly and positively correlated with the fruit N content and negatively related to the Ca content. Based on these, the above authors assumed that spongy tissue was caused by a deficiency of calcium. Accordingly, they attributed the reduction of spongy tissue incidence by CaCl2 as due to the reversal of calcium deficiency in treated fruits. However, past work in our lab had provided irrefutable evidence for the fact that the observed Ca deficiency in the mesocarp of ST affected fruit was due to movement of Ca away from pulp into seed for meeting the increasing demand for Ca caused dueto the onset of germination events and not due to the reduced uptake of the mineral [5]. Therefore, it was apparent that the effect of CaCl2 in reducing ST incidence in Alphonso mango was through a different mechanism of action. Since treatment with solutions of sodium chloride, CaCl2, potassium phosphate, nutrient mix and sea water (Table 1) could significantly reduce the spongy tissue incidence, it was evident that these salt solutions applied to fruits acted possibly by the osmotic stress caused by the presence of salts in the treatments.
It is known that the effect of salts on plants leads to physiological drought [13]. Salt solutions having 3% salts typically have more negative water potentials, relative to the water in the mesocarp. When a salt solution is applied on the fruit, water molecules in the mesocarp experience a strong pull from the more negative water potential of the salt solution from across the peel tissue along an osmotic gradient due to which the flow of water in to seed is prevented by the opposing force. Since the process of spongy tissue formation is initiated only in those fruits which are physiologically cut off from the supplies of water and nutrients due to funiculus damage [1], the prevention of flow of water from mesocarp to seed by application of salt solution leads to the development of moisture stress in the seed. Consequently, abscisicacid (ABA) level in the seed increases [14] causing seed dormancy. As the process of initiation of seed germination events is dependent on the initial hydration of seed followed by an uninterrupted flow of water into seed, the commencement of pre-germination-associated events in the seed are temporarily suspended, thereby preventing seedgermination and consequently leading to fruits with lesser incidence of spongy tissue disorder. Potassium phosphate at low concentrations has the ability to act as an systemic induced resistance elicitor leading to an increase in the synthesis of jasmonic acid (JA) [15] whichacts similar to ABA, thus inducing seed dormancy, delayed seed germination [16] and a reduced level of ST incidence.
Results presented in Table 3 further showed that fruits treated with nutrient mix and sea water showed improved fruit quality characteristics in terms of TSS, shelf life and pulp color in ripe fruits although the differences were not significant (Table 3). These observations indicated that the micronutrients in these treatments could be playing a role in enhancing fruit quality. Sea water is known to contain total dissolved solids in the range of 35,000 ppm in which the major components are sodium and chloride ions along with other micronutrient ions, such as calcium, boron, zinc, sulphur etc., [17]. Ca is known to stabilize cell membranes and plays a key role in maintaining cell structure and turgidity thus giving rise to fruits of superior quality. The increase in post-harvest life in fruits treated with the nutrient mix (Table 3) could be possibly related to the uptake of calcium by fruit tissue as reported in apple [18]. The improvement in pulp color as reflected in the higher carotenoid content of the pulp and fruit quality attributes (Table 3) could be due to copper, an essential micronutrient element involved in the formation of carotenoids and vitamin A in plants. A deficiency of copper is reported to lead to small fruits with poor color and quality [19]. Fe in nutrient mix could have improved fruit characters as a deficiency of the element leads to the production of smaller sized fruits with poor fruit quality parameters such as firmness and acidity [20]. Boron is essential for activation of dehydrogenase enzymes, sugar translocation, synthesis of nucleic acids and plant hormones [21]. Besides, manganese is involved in processes such as photosynthesis and metabolism of nitrogen and carbohydrates which contribute to fruit yield and quality. Zinc is a component of a wide array of enzymes and has a role in the production of the growth hormone, indolyl acetic acid. Considering the various functions of micronutrients in plants, it was apparent that the beneficial effect of the nutrient mix and sea water on fruit quality parameters reported in this study was evidently linked to the numerous vital functions of microelements and their synergistic actions in plants [22,23].
Data presented in Table 4 showed that spongy tissue incidence was significantly reduced when fruits of 60-70% maturity were treated while fruits treated beyond 70% maturity did not. These results clearly demonstrated that the imposition of the treatment before fruits reached 70% maturity was critical for regulation of spongy tissue incidence in fruits. Previous studies had shown that 70% fruit maturity is the critical stage at which the shift of seed in to germination phase occurs due to a greatly enhanced competition for resources among the developing fruits [1]. In order to prevent spongy tissue formation, it is, therefore, essential to avoid or delay the shift of seed into germination before it sets in. This conclusion was further corroborated from the finding that treatment of fruits past the critical stage was not beneficial due to the irreversible nature of the process of germination. Thus, the results of the present study involving the application of simple inorganic salts as a pre-harvest spray on developing fruits clearly showed that spongy tissue incidence could be prevented effectively by delaying seed germination events.
In conclusion, the study clearly established that treatment of developing fruits of Alphonso with nutrient mix or sea water between 60 and 70% fruit maturity was able to prevent spongy tissue formation in mature ripe fruits. Pre-harvest spray of fruits between 60 and 70% maturity for 2-3 times was essential to obtain effective control of the ST disorder. It was also apparent from the study that treatment of fruits before 50% maturity was not quite effective in controlling ST incidence and may also likely slow down the rate of fruit growth by retarding seed metabolism while treatment beyond 70% fruit maturity did not reduce spongy tissue incidence due to the irreversible nature of the process of germination. Inorganic salts are considered to belong to the category of substances classified as GRAS (Generally Recognized As Safe) by the United States Environmental Protection Agency (USEPA) under the Food Quality Protection Act. The salts employed for the control of ST are plant nutrients with no phyto-toxic effects, no adverse impact on the environment and public health, easy to apply and pose no health risks to farm workers, unlike pesticide chemicals. Therefore, this method produces fruits safe for consumption, besides being highly effective, economical and environment-friendly. It is also likely that treatment with salt solution may also provide a fair degree of protection against fungal diseases of mango fruits with little possibility for the pathogen to build up resistance. Thus, the protocol described here offers a simple and biosafe technology in the hands of farmers to produce superior quality Alphonso mango fruits, both for domestic consumption and export of fruits as well. Thus, the technology of pre-harvest treatment of Alphonso mango fruits for the control of spongy tissue disorder which had defied an answer for more than 75 years after it was first described by Cheema and Dani [24] represents a breakthrough achievement. This is also the world’s first report on the successful management of spongy tissue in Alphonso mango by using commonly available salts as pre-harvest spray application on developing fruits which could be gainfully applied by Alphonso mango growers for field control of the malady.
Acknowledgments
Thanks are due to the Director, IIHR, Bangalore for providing all facilities for the work. Grateful thanks are due to CSIR, New Delhi for financial grants under the Extra Mural Research project.
References
- Shivashankar S (2014) Physiological disorders of mango fruit. Hort Reviews 42: 313-347.
- Shivashankar S, Ravindra V, Louis L (2007) Biochemical changes in seed and mesocarp of mango (Mangifera indica L.) cv. ’Alphonso’ and their significance during the development of Spongy tissue . J Hort Sci & Biotechnol 82: 35-40.
- Paclobutrazol - Review Conducted by MDAR and Mass DEP for Use in Sensitive Areas of Rights-of-Way in Massachusetts (2012). The Commonwealth of Massachusetts Executive Office of Energy and Environmental Affairs, Department of Agricultural Resources, Boston, MA 02114.
- Litchenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148: 350-382.
- Ravindra V, Shivashankar S (2004) Spongy tissue in Alphonso mango- significance of in situ seed germination events. Curr Sci 87: 1045-1049.
- Chaney WR (2005) Growth retardants: A promising tool for managing urban trees. Purdue Extension document FNR-252-W. Accessed on May 11, 2011.
- Bozcuk S (1980) Effects of kinetin and salinity on germination of tomato, barley and cotton seeds. Ann Bot 48: 81-84.
- Sosa L, LlanesA, Reinoso H, ReginatoM, LunaV (2005) Osmotic and specific ion effect on the germination of Prospis strombulifera. Ann Bot 96: 261-267.
- Atia A, Debez A, Rabhi M, Athar HR, Abdelly C (2006) Alleviation of salt-induced seed dormancy in the perennial halophyte Crithmum maritimum L (apiaceae). Pak J Bot 38: 1367-1372.
- Torres CA, BallarÃn CMC, Monzón SAR, Galván FD, GarcÃa RP, et al. (2004) Effects of nitrogen and calcium supply on the incidence of internal fruit breakdown in ‘Tommy Atkins’ mangoes (Mangifera indica L.) grown in a soilless system. Acta Hort (ISHS) 645: 387-393.
- Young TW, Koo RCJ, Miner JT (1962) Effects of nitrogen, potassium and calcium fertilization on Kent mangoes in deep, acid, and sandy soil. P Fl St Hort Soc 70: 280-283.
- Cracknell Torres A, Galan Sauco V (2004) The study of the problem of mango (Mangiferaindica L.) internal breakdown In: Pinto A C Q, Pereira M E C, Alves R E. Int Soci Hortl Sci (ISHS): 167-169.
- Bojović B, Äelić G, Topuzović M, Stanković M (2010) Effects of NaCl on seed germination in some species from families brassicaceae and solanaceae. Kragujevac J Sci 32: 83-87.
- Benech Arnold RL, Fenner M, Edwards PJ (1991) Changes in germinability, ABA content and ABA embryonic sensitivity in developing seeds of Sorghum bicolor (L.) Moench. induced by water stress during grain filling. New Phytologist 118: 339-347.
- IIHR Annu Rep 2006-07 Research achievements. Efficacy and mode of action of potassium phosphate in the control of rose powdery mildew. 57
- Creelman RA, Mullet JE (1995) Jasmonic acid distribution and action in plants: Regulation during development and response to biotic and abiotic stress. Proc Natl Acad Sci U S A 92: 4114-4141.
- Anthoni J F (2006) The chemical composition of seawater.
- Asgharzade A, Babaeian M (2012) Foliar application of calcium borate and micronutrients effects on some characters of apple fruits in Shirvan region. Ann Biol Res 3: 527-533.
- Lovatt CJ (1999) Timing citrus and avocado foliar nutrient applications to increase fruit set and size. Hort Tech 9: 607-612.
- Fernandez V, Orera I, AbadÃa J, AbadÃa A (2009) Foliar iron-fertilisation of fruit trees: present knowledge and future perspectives - a review. J Hort Sci Biotech 84: 1-6.
- Brady NC, Weil RR (1996) The nature and properties of soils. Prentice Hall Inc. New Jersey.
- Ahmed F F, Morsy MH (2001) Response of Anna apple trees grown in the new reclaimed and to application of some nutrients and ascorbic acid. The Fifth Arabian Horticulture Conf Ismailia, Egypt, March 24-27, pp. 27-34.
- Fawzi MIF, Eman Abd El Moneim AA (2004) The effect of foliar application of some trace elements and active dry yeast on vegetative growth, yield, fruit quality and leaf chemical composition of Flame seedless grapevines grown in calcareous soil conditions . Minufiya J Agri Res 2: 463-478.
- Cheema GS, Dani PG (1934) Report on the export of mangoes to Europe in 1932 and 1933. Department of Agriculture, Bombay Bulletin. 170: 1-31.