Review Article
The Evolving Function of IFITs in Innate Immunity
Damuka Naresh, Prachi Yadav, Parul Sahu, Deepak Sachan, Paramananda Saikia*
Corresponding author: Dr. Paramananda Saikia, Department of Biotechnology & Bioinformatics, University of Hyderabad, Prof. C.R.Rao Road, Hyderabad-500046, India; E-mail: spsl@uohyd.ernet.in
Citation: Quamar MF, Bera SK. Ethno-Medico-Botanical Studies of Plant Resources of Hoshangabad District, Madhya Pradesh, India: Retrospect and 03 Prospects. J Plant Sci Res. 2014;1(1): 101.
Copyright © 2014 Paramananda Saikia et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Enzymology and Metabolism | Volume: 1, Issue: 1
Submission: 10/02/2014; Accepted: 12/03/2014; Published: 03/04/2014
Abstract
Virus infections are common threat to the human population. The interferons (IFN) are a large family of multifunctional proteins involved in antiviral defense, cell growth regulation and immune activation. Type 1 IFNs are produced in direct response to virus infection. IFN mediates its antiviral actions through the products of IFN stimulated genes (ISGs) which numbers in hundreds. Human ISG56, official gene symbol IFIT1, is the first ISGs to be discovered and cloned almost three decades ago. IFIT1 is highly induced in response to IFN, dsRNA (a mimic of virus replication intermediate) and many viruses. Recent researchers have shown that different IFITs specifically recognized pathogen associated molecular pattern molecules (PAMPs) and inhibits virus replication by inhibiting specific stages of virus replication or limiting multiplication by inducing early apoptosis of host cells. In this paper, we represent a review of evidence that IFITs play a significant role in the antiviral defense of IFN. More information of IFITs will promote a better understanding of cellular molecular mechanism of antiviral defense and provide a new insight for the development of therapeutics.
Keywords: Interferon; antiviral; innate immunity; virus; interferon stimulated gene products; IFITs
Introduction
Issac and Lindemann, in 1957 [1] reported that chick cells infected by Influenza virus produced a factor that mediates the transfer of a viral resistant state. They baptized this factor as Interferon (IFN) anddemonstrated its activity to induce interference of viral replication incells [1]. Later on, infection of human white blood cells with viruses induced the expression of human IFNs in response to virus infection which are secreted to circulation and act on the uninfected cells to activate a global antiviral state. These findings lead to the discovery of interferon as a large family of multifunctional secreted glycoproteins involved in antiviral defense, cell growth regulation and immune activation.
The IFNs are the subset of class 2 α helical cytokine that have existed in early chordates for about 450 million years ago and represent early elements in innate and adaptive immunity. About 11 mammalian IFN species have been discovered so far but only eight are found in humans. The functions of IFNs are represented by their three major biological activities viz. antiviral, antiproliferative and immunoregulatory effects. IFNs stimulate the innate cell mediated responses and then participate in the transition of initial host innate response into a potent adaptive immune response. Moreover they help to maintain Th1/Th2 balance for an effective host defense. Thus IFNs are the mediators of cellular homeostatic response towards virus infection. They are produced in response to viral infection and inhibit their replication thereby creating a negative feedback loop. The antiviral functions of IFNs are mediated by hundreds of interferon-Stimulated Gene products(ISGs) [2]. IFN induce their synthesis by activating the JAK-STAT pathway, an archetype of cell signaling used by many cytokines and growth factors. They bind to the extracellular domains of specific cell-surface receptors and transcriptionally activate the ISGs whose products inhibit various stages of virus replication. These ISGs may include IRF7, RIG-I, MDA5, OAS, PKR, MxA, ISG56, ISG20, iNOS, Viperin, ISG15 and many more. Among these ISG56/IFIT1 is the first to be discovered as antiviral and is most widely studied [3,4].
ISG56 family members genes also known as IFN-induced tetratricopeptide repeats 1(IFIT1) are clustered in locus on human chromosome 10q23.3 and is composed of ISG56/IFIT1, ISG54/ IFIT2, ISG60/IFIT3 and ISG58/IFIT5. These genes are evolutionaryconserved from mammals to amphibians which is evident from the presence of their homologs in other mammals (like monkey, rat, cow, sheep, hamster, horse and dog), in birds (chicken), in lizards, in fish (gold fish and puffer fish) and even in amphibians like xenopus [5]. The proteins of this family were characterized by the presence of 34 amino acids long TPR helix turn helix motifs present in cassettes of multiple repeats. These motifs mediate variety of protein-protein interactions and assembly of large protein complexes [6].
Structure of IFIT1 Gene Family Proteins
In human four IFIT1 family member have been characterized, namely, ISG56/IFIT1, ISG54/IFIT2, ISG60/IFIT3, and ISG58/IFIT5. The gene of this family consists of only 2 exons separated by an intron of few kilo bases in length. The first exon encodes only 5′ UTR and the start codon whereas second exon encodes the rest of the mRNA. Moreover, the promoter of these genes contain two IFN- stimulated response elements(ISRE) as only exclusive cis-acting element located within 200bp upstream of TATA box promoter (human ISG58 being the exception with 3 ISREs) [7]. These ISREs are identified by Interferon Regulatory Factors (IRF) that are activated by a variety of stimuli and thus induces the transcription of the corresponding mRNAs.
The structural hallmark of the IFIT proteins is the presence of multiple tetratricopeptide repeats (TPR) motifs dispersed throughout the whole sequence. These degenerate 34 amino acid motifs contain two anti-parallel helices, helix-A and helix-B. The interaction between helix-A and helix-B is mediated by eight loosely conserved residues W4, L7, G8, y11, A20, F24, A27 and P32 at stringently conservedpositions [6]. These anti-parallel helix-turn-helix arrays generates a right handed helical channel where helix-A is located inside the channel while helix-B is located on the outside of the channel, providing a corresponding binding facade for other proteins [8,9]. The amino acid present in the channel of this super helical structure often changes significantly, influencing the peculiarity of the protein for its protein substrate [6,8]. However, as predicted by sequence analysis, different IFIT family members have discrete number of TPR motifs which dictate their specific functions. For example, IFIT1 and IFIT2 were predicted to have six and four TPR motif, respectively [10].
Among the IFIT family member, IFIT2 is the first protein whose structure was elucidated by X-ray crystallography at a resolution of 2.8 Å[11]. The detailed structure revealed that IFIT2 monomers actually have 9 TPR motifs and form domain swapped dimers. The structure also revealed the presence of an exclusive positively charged C-terminus nucleotide binding channel attributing to its property of specifically binding to AU- rich RNA motif [12]. Mutation or deletion of amino acid residues in this region alters its viral RNA binding and have negative effect on its antiviral function. Thus homo or heterodimerization by domain swapping in these multi-TPR proteins imparts the IFIT protein with their distinct antiviral function to bind with viral RNA structures and proteins. But on the contrary, the fourth member of IFIT family IFIT5/ISG58 lacks any strongly interacting protein partner [13] and recognizes cellular RNA, including transfer RNAs. The crystal structure of human IFIT5 (residues 7-279) at 2.1Å resolution reveal a helical domain. Out of 24 α helices, 18 form canonical TPRs and the remaining six helices intercede between the TPRs which disturbs their regular association. This results in three discrete bundles of TPRs (subdomain I, II and III) forming a deep pocket which is 28 Å deep and 15 Å wide. This pocket is lined with extensive compilation of positively charged residues well suited for its binding with nucleic acids [14]. The detailed secondary structure also revealed a convoluted intramolecular packing of TPRs as a fold termed as TPR eddy. IFIT5 fold autonomously as a monomer which harbor a deep cleft with positive surface potential [15]. Thus IFIT5 readily recognizes cellular RNA rather than only viral-hallmark RNA structures and has evolved independent biochemical functions and distinct antiviral activity [16].
Evoluton of IFIT Gene Family
As evident from the literature, IFIT1 gene family is conserved inmammals, amphibians and fish but doesn’t exist in lower animals, like Drosophila, Caenorhabditis, Saccharomyces or in plants [3,4]. IFIT1 gene family is known to originate from a common vertebrate ancestor about 450 million years ago as IFIT genes are identified in genome databases from the jawed vertebrates including the cartilaginous elephant shark (C.milli), the phylogenetically oldest species of living jawed vertebrates [17]. But IFIT genes were absent from non-vertebrates such as sea squirt, lancelet and acorn worm. A constant number of 4 members of IFIT1 gene family ( IFIT1, 2, 3 & 5) are present in all mammalian species except in rat ( Rattus Norvegicus) and mouse lacking IFIT5, horse ( Equus Caballus) lacking IFIT1 and gibbons (Nomascus leucogenys) lacking IFIT3. Whereas opossum (Monodelphis domestica), chicken (Gallus gallus), frog (Xenopus tropicalis) and zebrafish (Danio rerio) possesses IFIT5 only. The genes from IFIT1 family have conserved gene structure and gene arrangement. The expansion of this gene family as studied by phylogenetic analyses has been occurred through lineage-specific and species-specific gene duplication. Much evidence has been found by genomic structure analysis of mammalian IFIT gene family which reveals a common evolutionary origin possibly by gene duplication. These evidence include.
1. 4 human IFIT members, including IFIT1, 2, 3&5 are tandem located in a locus on chromosome 10 and consistently 3 mouse homologues of IFIT members are also clustered together [5].
2. All mammalian IFIT proteins are classified into 4 subgroups with clear orthologous relationship [10,18,19].
3. Most of the mammalian gene shows a similar exon-intron organization with 2 exons separated by an intron of few kilo bases in length [20,21].
4. These genes possess a similar promoter structure characterized by two or three ISRE motif normally located within 200 bp upstream of TATA box [22,23].
Thus comprehensive analyses of genomic structure andphylogenetic tree indicated that evolutionary origin of IFIT gene is temporally associated with the appearance of jawed vertebrate and family members are involved in an active expansion by lineagespecific duplication [22,20,21,].
IFIT1 Proteins: Expression and Localization
Most cell type does not express IFIT family proteins under basal condition, with the exception of some myeloid cell subsets such as primary macrophage [24]. They are significantly induced by type1 and type 3 IFNs, especially IFNα/β [2]. In addition to IFN treatment, expression of these families of gene is triggered by virus infection, dsRNA and lipopolysaccharides; implying a crucial role under various cellular stresses. Thus IFIT genes have been described as viralstress inducible genes [10]. These genes are also induced directly at transcriptional level by Interferon Regulatory Factors (IRF-1, IRF-3, IRF-5 and IRF-7) which are activated soon after virus infection but before the induction of type1 IFN [25,26]. IRF-3 is a key transcriptional factor that signals the host cell to control viral infection by initiating IFN response in different cell types. The differential cell-specific expression of pathogen recognition receptors (PRRs) and selective IFITs against a particular viral infection alters the baseline antiviral state of cell and thus explains the induction of particular set of ISGs against a specific viral infection [24]. IFIT gene expression can also be induced independently through signal generated after the binding of cytoplasmic or endosome receptors such as TLR3, TLR4, MDA-5 and RIG-I by PAMPs (such as LPS, viral dsRNA,poly (dI:dC), etc.). Retinoic acid is also associated with the induction of human IFIT genes although much than PAMP dependent induction [27].
IFIT1 family gene transcription is efficiently elicited by bothDNA and RNA viruses. Among DNA viruses Human Cytomegalovirus (HCMV) [28] and Adenovirus [29] stimulates the expression of IFIT2 and IFIT1 & IFIT2 in human fibroblasts, respectively. Simianvirus 40 (SV40) and Polyomavirus also trigger IFIT family gene expression. Whereas among RNA viruses, West Nile Virus(WNV) induces IFIT1 & IFIT2 in mice [30], Hantavirus infection results in strong induction of IFIT3 expression in human endothelial cells [31]and Porcine Reproductive and Respiratory Syndrome Virus (PRRSV) activates IFIT1 & 3 in porcine alveolar macrophages [31, 32]. During Influenza A virus infection IFIT1, 2 & 3 were up-regulated in human primary macrophages [32]. While IFIT2 was strongly expressed in peripheral blood during acute stage of the infection [33]. Other RNA viruses which stimulate IFIT family expression include Japanese Encephalitis Virus, Lymphocytic Choriomeningitis Virus (LCMV), Rabies and Dengue virus.
Inspite of the structural similarity in IFIT family genes, theirexpression patterns only partially overlap in vivo and in vitro. The upstream promoter regions of IFIT genes contain ISRE which is responsible for the expression pattern of the IFIT genes [7,20,21]. Furthermore the expression kinetics of IFIT genes is cell type and tissue specific as supported by the selective induction of subsets of IFIT genes after stimulation with viral infections or IFNs. Also the IFIT mRNA levels can be persistent or ephemeral depending upon the cell type [4,36,30,37]. It was reported that IFIT3 is extensively expressed in kidney cells, T and B cells, neurons, plasmacytoid dendritic cells and myeloid dendritic cells [30,37]. Moreover a recent study utilized intracranial LCMV infection in mice to study the distinct pattern of preferential expression of different genes by the anatomic distribution of IFIT family transcripts in the brain. The astrocytes and hippocampal CA1/CA2 pyramidal cells mainly express IFIT1, deep cerebellar neurons express all the IFITs whereas IFIT2 transcript levels are highest in olfactory bulb [30]. This differential expression of IFIT genes conferred the important nonredundant antiviral function against particular viral infection[36,30].
The function of the protein mainly depends on their sub-cellular location. Usually IFIT1 family proteins function in the cytoplasm. IFIT1 is primarily located in the cytosol. But Li and coworkers found that IFIT1 is also located in the mitochondria where it interacts with mitochondrial membrane protein MAVS [38]. IFIT3 is also located in mitochondria and cytoplasm [39]. Whereas IFIT2 also appears in microtubules in addition to cytoplasm and mitochondria [40]. Instead of general cytoplasmic distribution, IFIT5 concentrates in actin rich protrusions from apical cell surfaces co-localized with the RNA- binding Retinoic acid- inducible gene-I ( RIG-I) [15].
Antiviral Functions of IFIT Family Proteins
1) IFIT1 Antiviral Functions
a. Inhibition of Hepatitis C virus translation initiation
The cellular function of IFIT1 is the inhibition of translation initiation [41]. Hepatitis C virus (HCV) is a positive-stranded RNA virus with an internal ribosome entry site (IRES). IFIT1inhibits HCV infection by blocking translation of the HCV RNA [42]. Human IFIT1 bind to eukaryotic initiation factor 3e (eIF3e), a translation initiation factor and it fractionate with the initiator ribosome HCV RNA complex [43]. IFIT1 inhibits the function of the HCV IRES, but mutant IFIT1 protein lacking eIF3-binding activity failed to inhibit HCV replication. In addition, overexpression of IFIT1 decreased HCV infection in hepatocytes [44]. As a result, IFIT1 blocks HCV replication by targeting eIF3e-dependent steps in the viral RNA translation initiation process; these steps include the recognition of the 43S pre-initiation complex by the HCV IRES and the assembly of the 43S-mRNA complex [45].
b. Inhibition of Human Papilloma Virus (HPV) Replication
IFIT1 inhibit human papillomavirus (HPV) replication by binding to the viral helicase E1 [46]. E1 is a multifunctional viral phosphoprotein that shuttles between the nucleus and cytoplasm, and has ATPase and DNA helicase activities. IFIT1 binds directly to HPV E1 where it restricts E1in the cyto plasm, and also inhibits E1 enzymatic activity needed for HPV replication [47].
c. Inhibition of the Translation of Viral mRNAs
Many viruses have evolved with N7- and 2′-Omethyltransferases which methylate their mRNA cap structures. IFIT1 inhibits the translation of viral mRNAs that are not properly capped. Mutants of West Nile virus, Vaccinia and mouse Coronavirus virus, lacking 2′-O methyltransferase activity are highly sensitive to IFN and IFIT protein [48]. It has also been reported that IFIT1 binds uncapped triphosphorylated RNA, which is found at the 5′ end of some RNA viruses and viral transcripts [49]. Recent studies have shown that IFIT1 recognizes RNA of Japanese encephalitis virus (JEV) lacking 2′-O methyltransferase (MTase) activity. IFIT1 inhibits the translation of mRNA through direct binding to mRNA 5′ structures and thereby restricts the replication of JEV mutated in 2′-O methyltransferase [50]. However, some other recent work suggests that Parainfluenza virus type 5 (PIV5) mRNAs are methylated at the hydroxyl group but IFIT1 inhibits the translation of PIV5 mRNA by unrecognized mechanism [51].
d. Activation of Innate Immunity
The antiviral innate immunity relies on the recognition of various microbial patterns by several transmembrane (Toll like Receptors) and cytoplasmic receptors (e.g. RIG-I and Mda-5). Homology modelling and site-specific mutational analyses demonstrated that IFIT1 is likely to recognize unique ‘non-self’ signature of 5′PPPRNA, which occurs during life cycle of large number of viruses, through its TPR motif. IFIT1 binds to PPP-RNA with nanomolar affinity and thus is central to antiviral activity against 5′ PPP-RNA generating viruses such as VSV and influenza A virus [49]. In the absence of IFIT1, the growth and pathogenicity of such viruses was much higher. Thus IFIT1 antagonize virus by specifically sequestering viral nucleic acid. IFIT1 has also been validated as the important bottleneck in controlling innate immune functions initiated by the toll like receptors as evident from the fact that it’s silencing resulted in the suppression of predicted downstream genes thus exerting a positive regulatory effect on these downstream genes [52]. It has also been shown to be a negative feedback regulator of virus-triggered signaling because over expression of IFIT1 inhibited the Sendai virus triggered activation of IRF3 and IFN β promoter and reversed the inhibition of VSV replication induced by poly(I:C) [38]. IFIT1 is also rapidly produced in response to stress induced by bacterial lipopolysaccharides and IL-1 and may involve in cell death pathways by interacting with eukaryotic elongation factor-1A [53]. Many studies revealed the association of IFIT1 in the pathogenesis of systemic lupus erythomatous (SLE) as it interacts with Rho/Rac guanine nucleotide exchange factor and regulate their activation. Thus inhibition of the expression of IFIT1 may become novel therapeutic approach to SLE [54]. Also IFIT1 serves as an important molecule to restrict Hepatitis C virus infection and may have implications in the development of therapeutic modalities. Six TPR motif of IFIT1 interact with ribosomal protein RPL15. RPL15 protein overexpress on the gastric cancer cells and contribute the antiproliferative capacity on. This interaction may facilitate the development of several new anticancer regimes [55].
e. Diagnostic marker
IFIT1 is also a potential diagnostic marker for ongoing viremia during Hepatitis E infection and acts as a marker for several diseases like myelodysplastic syndrome and chronic graft vs host disease [56].
2) IFIT2 Antiviral Functions:
IFIT1 and IFIT2 proteins are encoded by two closely related genes but their induction kinetics in response to IFNβ were quite different [57]. IFIT1 and IFIT2 bind with eukaryotic initiation factor 3e containing PCI motif to inhibit translation initiation of Hepatitis C virus [57]. IFIT2 interacts with both subunits of eIF3 whereas IFIT1 interacts with ‘e’ and not the ‘c’ subunit of the initiation factor. The resolved X-ray crystallographic structure of IFIT2 defined it as a novel RNA binding structure which interacts specifically with AU-rich RNA region and cellular RNA motif possessing AU-rich element [11]. This RNA binding ability seems to be associated with its antiviral activity. IFIT2 expression plays a significant role in the induction of cellular apoptosis in response to viral infection or IFN signaling by interacting with multiprotein complex such as IFIT1 and IFIT3 [58]. Also IFIT2 shows antiviral activity against West Nile virus and Corona virus by binding on the capped viral mRNA [59]. IFIT2 co-localizes with cytokeratin 18 and inhibit cell migration in oral squamous cell carcinoma (OSCC) cells. It was observed that with increase in the expression levels of IFIT2 the life span of OSCC patient enhances which is probably associated with the reduced migration of tumor cell [60]. Recent studies have shown that IFIT2 restricts WNV infection and pathogenesis in different tissue in a cell type specific manner, yet had small effect on the induction and extent of the innate and adaptive antiviral immune responses. When the results were compared to congenic wild-type control, Ifit2-/- mice showed enhanced WNV infection in a tissue-restricted manner with preferential replication in the central nervous system (CNS) of animals lacking IFIT2. The experiments also revealed no appreciable difference in B and T cell responses betweenIfit2-/- and wild-type mice during WNV infection. This indicates that the absence of IFIT2 does not influence the development of effective adaptive immunity during viral infection[45].
3) IFIT3 Antiviral Functions:
IFIT3 play important role in IFN-α antiviral activity. Over expression of IFIT3 in VERO cells decreases virus titers of VSV and Murine Encephalomyelitis virus (EMCV). The siRNA knockdown of IFIT3 confirms the decreased efficiency of IFN antiviral effect [61]. IFIT3 also serves as an important adaptor in innate immunity. IFIT3 interacts specifically with the N terminus of TBK1 (TANK binding kinase 1) which results in bridging TBK1 to MAVS (mitochondrial antiviral signaling) on the mitochondrion. As a result IRF3 gets phosphorylated and translocate into the nucleus to promote antiviral gene transcription such as IFNβ, ISG56, Rantes etc [62]. The induced expression of IFIT3 by Dengue virus infection was demonstrated in human dendritic cells and it play important protective roles in Dengue virus infection of human lung epithelial cells. Furthermore, the results demonstrated that knockdown of IFIT3 led to the expression of several apoptotic regulator such as caspase 3, caspase 8, caspase 9, and Bcl-2-associated X protein (BAX). These results reveal that IFIT3 might be critical for maintaining cell survival [63].
4) IFIT5 Antiviral Functions:
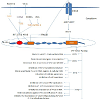
Human IFIT5 crystal structure reveals a novel arrangement of TPR domain that directly bind only PPP- single stranded RNA in a non-sequence specific manner. Functional analysis in human embryonic kidney (HEK) cells reveal a reduction of viral replication only in the presence of proper PPP-RNA binding by IFIT5 or IFIT1[14]. Recent studies have reported that both the mRNA and protein levels of IFIT5 are up-regulated in response to RNA virus infection or polyinosinic-cytidylic acid stimulation (polyI:C) stimulation. Ectopic expression of IFIT5 could synergize IRF3- and NF-κB-mediated gene expression whereas knockdown of IFIT5 impair the transcription of these genes. This study demonstrates that IFIT5 potentiate anti-viral response through enhancing innate immune signaling pathways [64] (Table 1, Figure 1).
Figure 1: Viral RNA and DNA are detected by cytosolic RIG-I like receptors (RLRs) or endosomal Toll like Receptors (TLRs) including TLR3, 7, 9. The downstream signaling leads to the activation of IRFs and produces IFNs which signals through IFN receptors (IFNAR) and JAK/STAT pathway followed by the activation of ISGF3 complex which translocate into the nucleus and activates ISREs. This activation of ISRE elements results in the transcription of IFIT1 gene family. These IFIT proteins thus generate antiviral state by functioning as antiviral molecules.
Acknowledgement
This review was supported by the Ramalingaswami Fellowship contingency grant, Department of Biotechnology, Govt. of India.
References
- Isaacs A, Lindenman J (1957) Virus interference. I. The interferon. Proc R Soc 147: 258-267.
- Der SD, Zhou A, Williams BR, Silverman RH (1998) Identification of genes differentially regulated by interferon alpha, beta, or gamma using oligonucleotide arrays. Proc Natl Acad Sci U S A 95: 15623-15628.
- Chebath J, Merlin G, Metz R, Benech P, Revel M (1983) Interferon-induced 56,000 Mr protein and its mRNA in human cells: molecular cloning and partial sequence of the cDNA. Nucleic Acids Res 11: 1213-1226.
- Kusari J, Sen, GC (1986) Regulation of synthesis and turnover of an interferon-inducible mRNA. Mol Cell Biol 6: 2062-2067.
- Fensterl V, Sen GC (2011) The ISG56/IFIT1 gene family. J Interferon Cytokine Res 31: 71-78.
- Lamb JR, Tugendreich S, Hieter P (1995) Tetratrico peptide repeat interactions: to TPR or not to TPR? Trends Biochem Sci 20: 257-259.
- Levy D, Larner A, Chaudhuri A, Babiss LE, Darnell JE jr (1986) Interferon-stimulated transcription: isolation of an inducible gene and identification of its regulatory region. Proc Natl Acad Sci USA 83: 8929-8933.
- D’Andrea LD, Regan L (2003) TPR proteins: the versatile helix. Trends BiochemSci 28: 655-662.
- Blatch GL, Lassle M (1999) The tetratricopeptide repeat: a structural motif mediating protein-protein interactions. Bioessays 21: 932-939.
- Sarkar SN, Sen GC (2004) Novel functions of proteins encoded by viral stress-inducible genes. Pharmacol Ther 103: 245-259.
- Yang Z, Liang H, Zhou Q, Li Y, Chen H, et al (2012) Crystal structure of ISG54 reveals a novel RNA binding structure and potential functional mechanisms. Cell Res 22: 1328-1338.
- Xu N, Chen CY, Shyu A B (1997) Modulation of the fate of cytoplasmicm RNA by AU-rich elements: key sequence features controlling mRNA deadenylation and decay. Mol Cell Biol 17: 4611-4621.
- Hogg, JR, Collins K (2007) RNA-based affinity purification reveals 7SK RNPs with distinct composition and regulation. RNA 13: 868-880.
- AbbasYM, Pichlmair A, Gorna M W, Superti-Furga G, Nagar B (2013) Structural basis for viral 5’-PPP-RNA recognition by human IFIT proteins. Nature 494: 60-64.
- Katibah GE, Lee HJ, Huizar JP, Vogan JM, Alber T, et al. (2013) tRNA Binding, TPR Eddy Fold, and Subcellular Localization of the Human Interferon-induced Protein, IFIT5. Mol Cell 49: 743-750.
- Schoggins JW, Wilson SJ, Panis M, Murphy MY, Jones CT, et al. (2011) A diverse range of gene products are effectors of the type I interferon antiviral response. Nature 472: 481-485.
- Venkatesh B, Kirkness EF, Loh YH, Halpern AL, Lee AP, et al. (2007) Survey sequencing and comparative analysis of the elephant shark (Callorhinchusmilii) genome. PLoS Biol 5: e101.
- Smith JB, Herschman HR (1996) The glucocorticoid attenuated response genes GARG-16, GARG-39, and GARG-49/IRG2 encode inducible proteins containing multiple tetratricopeptide repeat domains. Arch Biochem Biophys 330: 290-300.
- Zhang YB , Gui JF (2004) Identification and expression analysis of two IFN inducible genes in crucian carp (Carassiusauratus L.). Gene 325: 43-51.
- Bluyssen HA, Vlietstra RJ, Faber PW, Smit EM, Hagemeijer A, et al. (1994) Structure, chromosome localization, and regulation of expression of the Interferon-Regulated Mouse Ifi54/Ifi56 gene Family. Genomics 24: 137-148.
- de Veer MJ, Sim H, Whisstock JC, Devenish RJ, Ralph SJ (1998) IFI60/ISG60/ IFIT4, a new member of the human IFI54/IFIT2 family of interferon-stimulated genes. Genomics 54: 267-277.
- Xiao S, Li D, Zhu HQ, Song MG, Pan XR, et al. (2006) RIG-G as a key mediator of the antiproliferative activity of interferon-related pathways through enhancing p21 and p27 proteins. Proc Nat Acad Sci the U S A 103: 16448-16453.
- Wathelet MG, Clauss IM, Nols CB, Content J, Huez GA (1987) New Inducers Revealed by the Promoter Sequence-Analysis of 2 Interferon-Activated Human Genes. Eur J Biochem 169: 313-321.
- Daffis S, Samuel MA, Keller BC, Gale M Jr, Diamond MS (2007) Cell-specific IRF-3 responses protect against West Nile virus infection by interferon-dependent and-independent mechanisms. PLoS Pathog 3: e106.
- Grandvaux N, Servant MJ, tenoever B, Sen GC, Balachandran s, et al. (2002) Transcriptional profiling of interferon regulatory factor 3 target genes: direct involvement in the regulation of interferon-stimulated genes. J Virol 76: 5532-5539.
- Ogawa S, Lozach J, Benner C, Pascual G, Tangirala RK, et al. (2005) Molecular determinants of crosstalk between nuclear receptors and Toll-like receptors. Cell 122: 707-721.
- Yu M, Tong JH, Mao M, Kan LX, Liu MM, et al (1997) Cloning of a gene (RIG‑G) associated with retinoic acid-induced differentiation of acute promyelocytic leukemia cells and representing a new member of a family of interferon-stimulated genes. Proc Natl Acad Sci USA 94: 7406-7411.
- Zhu H, Cong JP, Shenk T (1997) Use of differential display analysis to assess the effect of human cytomegalovirus infection on the accumulation of cellular RNAs: induction of interferon-responsive RNAs. Proc Natl Acad Sci U SA 94: 13985-13990.
- Zhao H, Boije H, Granberg F, Pettersson U, Svensson C (2009) Activation of the interferon-induced STAT pathway during an adenovirus type 12 infection. Virology 392: 186-195.
- Wacher C, Muller M, Hofer MJ, Getts DR, Zabaras R, et al. (2007) Coordinated regulation and widespread cellular expression of interferon-stimulated genes (ISG) ISG-49, ISG-54, and ISG-56 in the central nervous system after infection with distinct viruses. J Virol 81: 860-871.
- Matthys V, Gorbunova EE, Gavrilovskaya IN, Pepini T, Mackow ER (2011) The C-terminal 42 residues of the Tula virus Gn protein regulate interferon induction. J Virol 85: 4752-4760.
- Xiao S, Mo D, Wang Q, Jia J, Qin L, et al. (2010) Aberrant host immune response induced by highly virulent PRRSV identified by digital gene expression tag profiling. BMC Genomics 11: 544.
- Zhou P, Zhai S, Zhou X, Lin P, Jiang T, et al. (2011) Molecular characterization of transcriptome-wide interactions between highly pathogenic porcine reproductive and respiratory syndrome virus and porcine alveolar macrophages in vivo. Int J Biol Sci 7: 947-959.
- Lietzen N, Ohman T, Rintahaka J, Julkunen I, Aittokallio T, et al. (2011) Quantitative subcellular proteome and secretome profiling of influenza A virus-infected human primary macrophages. PLoS Pathog 7: e1001340.
- Kawada J, Kimura H, Kamachi Y, Nishikawa K, Taniguchi M, et al. (2006) Analysis of gene-expression profiles by oligonucleotide microarray in children with influenza. J Gen Virol, 87: 1677-1683.
- Terenzi F, White C, Pal S, Williams BR, Sen GC (2007) Tissue-specific and inducer-specific differential induction of ISG56 and ISG54 in mice. J Virol 81: 8656-8665.
- Fensterl V, White CL, Yamashita M, Sen GC (2008) Novel characteristics of the function and induction of murine p56 family proteins. J Virol 82: 11045-11053.
- Li Y, Li C, Xue P, Zhong B, Mao AP, et al. (2009) ISG56 is a negative-feedback regulator of virus-triggered signaling and cellular antiviral response. Proc Natl Acad Sci U S A 106: 7945-7950.
- Liu XY, Chen W, Wei B, Shan YF, Wang C (2011) IFN-induced TPR protein IFIT3 potentiates antiviral signaling by bridging MAVS and TBK1. J Immunol 187: 2559-2568.
- Saha S, Sugumar P, Bhandari P, Rangarajan PN (2006) Identification of Japanese encephalitis virus-inducible genes in mouse brain and characterization of GARG39/IFIT2 as a microtubule-associated protein. J Gen Virol 87: 3285-3289.
- Guo J, Peters KL, Sen GC (2000) Induction of the human protein P56 by interferon, double-stranded RNA, or virus infection. Virology 267: 209–219.
- Sumpter R Jr, Wang C, Foy E, Loo YM, Gale M Jr (2004)Viral evolution and interferon resistance of hepatitis C virus RNA replication in a cell culture model. J Virol 78: 11591-11604.
- Wang C, Pflugheber J, Sumpter, JR, Sodora DL, Hui D (2003) Alpha interferon induces distinct translational control programs to suppress hepatitis C virus RNA replication. J Virol 77: 3898-3912.
- Raychoudhuri A, Shrivastava S, Steele R, Kim H, Ray R, et al. (2011) ISG56 and IFITM1 proteins inhibit hepatitis C virus replication. J. Virol. 85: 12881-12889.
- Cho H, Shrestha B, Sen GC, Diamond MS (2013) A Role for Iï¬t2 in Restricting West Nile Virus Infection in the Brain. J Virol 87: 8363-8371.
- Terenzi F, Saikia P, Sen GC (2008) Interferon-inducible protein, P56, inhibits HPV DNA replication by binding to the viral protein E1. EMBO J 27: 3311-3321.
- Saikia P, Fensterl V, Sen GC (2010) The inhibitory action of P56 on select functions of E1 mediates interferon’s effect on human papillomavirus DNA replication. J Virol 84: 13036-13039.
- Zust R, Cervantes-Barragan L, Habjan M, Maier R, Neuman BW, et al. (2011) Ribose 29-O-methylation provides a molecular signature for the distinction of self and non-self mRNA dependent on the RNA sensor Mda5. Nat Immunol 12: 137-143.
- Pichlmair A, Lassnig C, Eberle CA, Gorna MW, Baumann CL, et al. (2011) IFIT1 is an antiviral protein that recognizes 5’- triphosphate RNA. Nat Immunol 12: 624-630.
- Kimura T, Katoh H, Kayama H, Saiga H, Okuyama M, et al. (2013) Ifit1 inhibits Japanese encephalitis virus replication through binding to 5' capped 2'-O unmethylated RNA. J Virol 87: 9997-10003.
- Andrejeva J, Norsted H, Habjan M, Thiel V, Goodbourn S, Randall RE (2013) ISG56/IFIT1 is primarily responsible for interferon induced changes to patterns of parainfluenza virus type 5 transcription and protein synthesis. J GenVirol 94: 59-68.
- Guo J, Hui, DJ, Merrick WC, Sen GC (2000) A new pathway of translational regulation mediated by eukaryotic initiation factor 3. EMBO J 19: 6891-6899.
- Li HT, Su YP, Cheng TM, Xu JM, Liao J, et al. (2010) The interaction between interferon-induced protein with tetratricopeptide repeats-1 and eukaryotic elongation factor-1A. Mol Cell Biochem 337: 101-110.
- Tang JP, Gu YY, Shen N, Ye S, Qian J, et al. (2004) Interferon-inducible genes lymphocyte antigen 6 complex E and tetratricopeptide repeats 1 are correlated with clinical features of patients with systemic lupus erythematosus. Zhonghua Yi Xue Za Zhi 84: 1157-1160.
- Hsu YA, Lin HJ, Sheu JJ, Shieh FK, Chen SY, et al. (2011) A Novel interaction between interferon-inducible protein p56 and ribosomal protein L15 in gastric cancer cells. DNA Cell Biol 30: 671-679.
- http://www.ncbi.nlm.nih.gov/pubmed/?term=Chronic+Hepatitis+E+Virus+Infection+Is+Specifically+Associated+With+an+Interferon-+Related+Transcriptional+Program.
- Terenzi F, Hui DJ, Merrick WC, Sen GC (2006) Distinct induction patterns and functions of two closely related interferon-inducible human genes, ISG54 and ISG56. J Biol Chem 281: 34064-34071.
- Stawowczyk M, Van Scoy S, Kumar KP, Reich NC (2011) The interferon stimulated gene 54 promotes apoptosis. J Biol Chem 286: 7257-7266.
- Daffis, S, Szretter KJ, Schriewer J, Li J, Youn S (2010) 2’-O methylation of the viral mRNA cap evades host restriction by IFIT family members. Nature 468: 452-456.
- Lai KC, Chang KW, Liu CJ, Kao SY, Lee TC (2008) IFN-Induced Protein with Tetratricopeptide Repeats 2 Inhibits Migration Activity and Increases Survival of Oral Squamous Cell Carcinoma. Mol Cancer Res, 6: 1431-1439.
- Schmeisser H, Mejido J, Balinsky CA, Morrow AN, Clark CR, et al. (2010) Identification of alpha interferon-induced Genes associated with Antiviral Activity in Daudi Cells and Characterization of IFIT3 as a Novel Antiviral Gene. J Virol 84: 10671-10680.
- Liu XY, Chen W, Wei B, Shan YF, Wang C (2011) IFN-Induced TPR Protein IFIT3 Potentiates Antiviral Signaling by Bridging MAVS and TBK1. J Immunol 187: 2559-2568.
- Hsu YL, Shi SF, Wu WL, Ho LJ, Lai JH (2013) Protective Roles of Interferon Induced Protein with Tetratricopeptide Repeats 3 (IFIT3) in Dengue Virus Infection of Human Lung Epithelial Cells. PLoS One 8: e79518.
- Zhang B, Liu X, Chen W, Chen L (2013) IFIT5 potentiates anti-viral response through enhancing innate immune signaling pathways. Acta Biochim Biophys Sin (Shanghai) 45: 867-874.