Sequence Variation in Promoter Regionof Irf-4 Gene and the Risk of CML in the Kashmiri Population
Gulzar Bhat1, Ashaqullah Bhat1, Wani Aadil1, Ali Muzamil1, Samoon Jeelani2, Akbar Masood1, Bashir Ahmad Ganai1*
1Department of Biochemistry, University of Kashmir, Srinagar, India
2Department of Clinical Hematology, SKIMS, Srinagar, India
Corresponding author: Bashir Ahmad Ganai, Department of Biochemistry, University of Kashmir, Srinagar, India; E-mail: bbcganai@gmail.com
Citation: Bhat G, Bhat A, Aadil W, Muzamil A, Jeelani S, et al. Sequence Variation in Promoter Region of Irf-4 Gene and the Risk of CML in the Kashmiri02 Population. J Cancer Res Molecul Med. 2015;2(1): 102.
Copyright © 2015 Bhat G, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Cancer Research and Molecular Medicine | Volume: 2, Issue: 1
Submission: Submission: 24/09/2015; Accepted: 28/09/2015; Published: 03/10/2015
Abstract
A number of theories have been postulated to describe the etiology of Chronic Myeloid Leukemia (CML), such as genetic alterations and alterations in cytokine production. A combination of inflammatory cytokines has an important role in cancer development. The aim of this hospital based case-control study was to screen for variations located in promoter region of irf-4 gene sequence in CML cases and healthy controls of Kashmiri population. We included sixty confirmed CML cases and an equal number of age, district and gender matched controls in this study. Sequencing of both the CML case and control samples for any potential variation in the nucleotide sequence in the promoter region of irf-4 gene was done commercially. Sequence alignment with the wild type sequence revealed the substitution of T with G nucleotide at -991 position in the promoter region of irf-4 gene. The allelic frequency for normal andvariant allele in CML cases was 46.66% and 53.33% respectively. In controls, the allelic frequency observed for normal and variant allele was 86.66% and 13.33% respectively. Thus a statistically significant association was observed between the CML and the presence of variant allele (TG) (OR= 7.43, 95%CI: (3.9-14.1); p = 0.0001). Our study also revealed that CML is more prevalent in males as compared to females (ratio is 1.12:1, and the fruit consumption was very low in CML cases (72.33%). Furthermore 74.00% of the CML cases were found to be tobacco smokers. This is the first report on the sequence variation in promoter region of irf-4 gene and the risk to CML in the Kashmiri population. However, the understanding gained through this study needs to be furthersubstantiated by conducting a similar study on statistically significant sample size.
Keywords: CML; Cytokine; Philadelphia chromosome, IRF-4
Introduction
Chronic myelogenous leukemia (CML) is a clonal hematopoieticdisorder caused by an acquired genetic defect in a pluripotent stem cell. Incidence increases with age, being exceptionally rare in infancy at 0.7/million/year at ages 1 to 14 years and rising to 1.2/million/year in adolescents [1]. It is slightly more common in males than in females. In general male predominance has been estimated to be1.3-1.4 to 1. The hallmark genetic aberration of CML is a reciprocalchromosomal translocation t(9;22) leadingto expression of a BCR-ABL fusion gene, an aberrant activatedtyrosine kinase [2]. However there is mounting evidence that the deregulation of other genes, such as the transcription factor interferon regulatory factor 4 (IRF-4), MGMT, DAPK, p14ARF, p15INK4b, p16INK4a, BRCA1, CDH13and APAF-1 is also implicated in the pathogenesis of CML [3-5]. Only a limited number of studies have been so far conducted in which the role of other possible confounding factors like environment has been studied together with genetic analysis. This hospital based casecontrol study was devised which was aimed to evaluate the promoter region of the irf-4 gene for any nucleotide variation in the CML patients and healthy controls of the Kashmir valley.
Interferon regulatory factors (IRFs) comprise a family oftranscription factors that function within the JAK-STAT pathwayto regulate interferon (IFN) and IFN-inducible gene expression inresponse to viral infection. IRFs play an important role in pathogendefense, autoimmunity, lymphocyte development, cell growth andsusceptibility to transformation.IRF-4 is a lymphoid and myeloidrestricted member of IRF transcriptional family [6,7]. The product of the MUM1/IRF4 gene (also called PIP, LSIRF, ICSAT) [8-11] is a member of the interferon regulatory factor (IRF) family of transcription factors, known to play an important role in the regulation of gene expression in response to signaling by interferons and by other cytokines [12]. Besides Bcr-Abl translocation which is known to be causative genetic aberrationinchronic myeloid leukemia (CML), there is mounting evidence that the deregulation of interferon regulatory factor 4 (IRF-4), is also implicated in the pathogenesis of CML [13]. IRF-4 polymorphisms are associated with susceptibility to chronic lymphoid leukemia (CLL) and non-Hodgkin lymphoma (NHL) [14]. Mutations in the distal and proximal sites of this GC-rich sequence resulted in 62 and 81% reductions in the irf-4 promoter activity, respectively [15].
Till date no study has been done to understand the etiology of CML in Kashmir. Therefore the aim of this hospital based casecontrol study was to screen for variations located in promoter region of irf-4 gene sequence in CML cases and healthy controls in a sample of Kashmiri population.
Materials and Methods
Patient characteristics
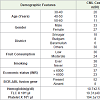
Patients with Chronic Myeloid Leukemia were evaluated at the department of Hematology of Sheri-Kashmir Institute of Medical Sciences (SKIMS), Soura. Clinical details were obtained by going through the medical records of the cases. The diagnoses of CML was based on the standard clinico-hematological criteria and the presence of Philadelphia chromosome (BCR-ABL fusion gene). Controls were taken from healthy individuals of Kashmir valley from Department of Hematology, SKIMS, Soura. Generally, Patients coming for treatment of minor ailments at the SKIMS Hospital, with no evidence of cancer were selected as controls. The patient characteristics are summarized in Table 1. A total of 60 CML cases (males 33 (55 %) and females 27(45 %) with a mean age of 44.7 ± 13.61) were recruited and during the same period 60 volunteer controls (males 42 (70%) and females 18 (30%) with the mean age of 31.83 ± 7.52) were also recruited. Three ml of venous blood was collected in a sterile EDTA coated vials and was stored at -20 °C. All patients and healthy individuals gave their written informed consent to participate in the study. Genomic DNA was isolated from the blood samples by using Phenol-Chloroform method (Sambrook 2001) and the isolated DNA was stored at -20 °C for future use. The study was approved by the Ethics Committee of the Sher-i-Kashmir Institute of Medical Science (SKIMS), India.
PCR amplification
After agarose gel electrophoresis it was found that concentration and purity of genomic DNA is desirable, the desired fragment of DNA i.e., irf-4 promoter, was amplified by polymerase chain reaction (PCR). The technique was standardized for available environmental conditions. After standardizing all the parameters of PCR like varying annealing temperature, dNTP, primer and template concentration, the promoter regionof irf-4 gene was amplified.PCR was performed in total volume of 50μl. The PCR reactions were composed of 100 ng of genomic DNA, 0.2mM dNTPs, 0.4 pmoles/μl of each primer and 0.2 U/μl of Taq polymerase in 1X PCR buffer. The primer pair used for amplification of promoter region of irf-4 was: forward primer: 5´- TTG AGA TGG AGT CTT GCT CTG T -3´; reverse primer: 5´- ATC ACT TCC AGA CTT CAG TTC ACC T -3´which on amplificationproduced a 314-bp product. Initial denaturation at 95 °C for 7 minutes was followed by 35 cycles of 50 second at 95 °C, 1 minute at 67 °C, 1 minute at 72 °C and final extension for10 minutes at 72 °C.
Amplification and specificity of amplicon obtained in the PCR reaction was analyzed by 2% agarose gel electrophoresis (Figure 1). The gel was visualized on UV-illuminator and photographed on theGel Doc System.
Purification and DNA Sequencing
The purification and sequencing was commercially done using the services of Macrogen Inc. Soel, Korea. For purification and sequencing we send 50μl of unpurified PCR product samples along with 50μl of 20μM Forward & Reverse primers were sentto Macrogen, Korea. Alignment of all the sequences pertained to DNA samples of various cases and controls was done with respect to control sequence.
Statistical Analysis
The χ2-test was used to examine the differences in the distribution of genotypes between cases and controls. ORs with 95% CIs were computed using unconditional logistic regression (GraphPad Prism 5) and adjusted for age and gender.
Results
Sequencing of all the samples for any potential variation in the nucleotide sequence of the promoter region of irf-4 gene was donecommercially using the services of Macrogen Inc. Seol, Korea. Alignment of all the sequences pertained to DNA samples of various cases and controls was done with respect to control sequence. Sequencing results for irf-4 promoter for any potential variations for affected and unaffected individuals are respectfully shown by chromatograms in Figures 2 and 3. These chromatograms indicate the presence of single TG polymorphism at -991 position in promoter region of irf-4 gene. Since this T G polymorphism was seen in the promoter region of the gene, which might have an important role in the expression level of irf-4.
In present study we analyzed 60 confirmed CML cases and an equal number of nonmalignant controls belonging to Kashmir division for any potential SNP in the promoter region of irf-4 gene. In CML cases, the allelic frequency for normal allele T was found to be 46.66% (56/120). The allelic frequency observed for T G type was found to be 53.33% (64/120). An equal number of non malignant age and gender matched controls were screened for the any potential SNP in the promoter region of irf-4gene. In controls, the allelic frequency observed for normal allele T was 86.66% (104/120). The allelic frequency in controls observed for TG type was found to be 13.33% (16/120).
Since the frequency observed for the variant allele (TG) was higher in CML cases (53.33%) than in normal controls (13.33%) and was found to be statistically significant (OR= 7.43, 95%CI:(3.9-14.1); p = 0.0001).The frequency of irf-4 promoter alleles in CML patients and controls is summarized in Table 2.
Discussion
Chronic myeloid leukemia (CML) is probably the most extensively studied human malignancy. The definite mechanism leading to the development of this disease is not known yet. The known genetic abnormality associated with the CML is the condition known as Philadelphia chromosome which occurs as a result of reciprocal translocation between chromosome 9 and 22 leading to juxtaposition of BCR-ABL gene [16]. Only a limited number of studies have been so far conducted in which the role of other possible confounding factors like environment has been studied together with genetic analysis. Only causative factor known to be associated withCML is exposure to radioactivity. Individual genotypic differences and also the level of expression of the carcinogen-metabolizing enzymes are crucial in determining the susceptibility of developing the cancer [17]. Interferon-regulatory factor-4 (irf-4) is an IRF family transcription factor important for hematopoietic development andimmune processes. Irf-4 is expressed in lymphoid cells, dendritic cells,and macrophages, where it is associated with regulation of important cellular processes, including cell differentiation, apoptosis, DNArepair, and cytokine production [18]. irf-4 expression is reported to be down-regulated in patients with CML but restored in response to treatment with interferon-α (IFN-α), and higher irf-4 expression is associated with a good response to IFN-α treatment [19,20]. By direct sequencing of the promoter region of irf-4 gene, two base pair changes were found at the position -1081 (TC substitution), and at the position -1068 (AC substitution), which were later found not responsible for absent irf-4-expression. Only few studies have been conducted on CML in Kashmir and the cause of the high incidence rate is yet a mystery. Thus a hospital based case-control study was devised which was aimed to evaluate the promoter region of the irf-4 gene for any nucleotide variation in the CML patients and healthy controls of the Kashmir valley.
The present study consists of sixty confirmed CML cases and also equal number of age, gender and district matched controls. Thirty three percent (20/60) of CML patients were men thus giving the male to female ratio to be 1:1.12. We did not find any significant difference in the mean age of the cases (44.7 years) and controls (43.6 years).We observed a higher representation of CML cases in the age group between 30 and 40. Similar pattern of age at diagnosis was also observed in studies conducted by Mendizabal et al., [21]. The highest number of the CML patients in our study were from central part of Kashmir valley with district Srinagar accounting for 30% of the cases. The wide difference in the incidence rate of the CML across the different districts of the Kashmir valley suggests the role of some environmental exposure of the local population. The influence of such factors on the incidence rate if any needs to be studied by conducting well designed scientific studies. In addition we assessed all the CML patients for the presence of Philadelphia chromosome (BCR-ABL fusion gene) and observed that 52 (87%) of CML cases were positive. Our results are in agreement with other studies which have reported high prevalence of Philadelphia chromosome in CML patients [22,23]. In the present study we analyzed 60 confirmed CML cases and also equal number of gender, age and district matched nonmalignant controls belonging to Kashmir division for any mutation (SNPs) in the promoter region of irf-4 gene by employing direct sequencing method. We found a single T to G substitution in the irf-4 promoter region. In CML cases, the allelic frequency for normal allele (T) was found to be 46.66% (56/120) and the allelic frequency observed for TG type was found to be 53.33% (64/120). An equal number of non malignant age and gender matched controls were screened for the same mutation (SNPs) in the promoter region of irf-4gene. The allelic frequency observed in controls for normal allele (T) was 86.66% (104/120). The frequencies analyzed for TGallele was 13.33% (16/120). Since the frequency observed for T G allele was higher in CML cases (53.33%) than in normal controls (13.33%) and was found to be statistically significant (OR= 7.43, 95%CI:(3.02-18.29); p = 0.0001). It is reported that 62 and 81% reductions in the irf-4 promoter activity occurred due to mutations in the distal and proximal sites of the GC-rich sequence resulted in
respectively [15]. Irf-4 deficiency facilitates BCR/ABL-mediated transformation of B-lymphoid progenitors in vitro and accelerates progression of BCR/ABL-induced B-ALL in mice, and that forcedexpression of irf-4 effectively suppresses lymphoid leukemogenesisby BCR/ABL [18]. Since in our study T G polymorphism was found in the promoter region of the gene, which might have an important role in the expression level of Interferon regulatory factor 4 gene and hence on the risk to CML in Kashmiri population. This information collected by this study lays a foundation for further analysis and understanding the role of various other confounding factors which might be playing a role in the development of CML.
Acknowledgement
This study was financially supported by the Department ofBiochemistry, University of Kashmir, India. The authors wish to thank all the consultants and the other paramedical staff of the Department of Clinical Hematology at SKIMS for their invaluable help during sample collection. We are also highly grateful to all the CML patients and controls who participated in this study.
References
- Ries LAG, Melbert D, Krapcho M, et al. SEER cancer statistics review. 2008; Accessed October 6, 2010 Bethesda, MD National Cancer Institute.
- Sawyers CL (1999) Chronic myeloid leukemia. N Engl J Med 340: 1330-1340.
- Esteller M, Herman JG (2002) Cancer as an epigenetic disease: DNA methylation and chromatin alterations in human tumours. J Pathol 196: 1-7.
- Hermann JG, Civin CI, Issa JP, Collector MI, Sharkis SJ, et al. (1997) Distinct patterns of inactivation of p15INK4B and p16INK4A characterize the major types of hematological malignancies. Cancer Res 57: 837-841.
- Esteller M (2002) CpG island hypermethylation and tumor suppressor genes: a booming present, a brighter future. Oncogene 21: 5427-5440.
- Mamane Y, Heylbroeck C, Génin P, Algarté M, Servant MJ, et al. (1999) Interferon regulatory factors: the next generation. Gene 237: 1-14.
- Mittrücker HW, Matsuyama T, Grossman A, Kündig TM, Potter J, et al. (1997) Requirement for the transcription factor LSIRF/IRF4 for mature B and T lymphocyte function. Science 275: 540-543.
- Taniguchi T, Ogasawara K, Takaoka A, Tanaka N (2001) IRF family of transcription factors as regulators of host defense. Annu Rev Immunol 19: 623-655.
- Grossman A, Mittrucker HW, Nicholl J, Suzuki A, Chung S, et al. (1996) Cloning of human lymphocyte-specific interferon regulatory factor (hLSIRF/hIRF4) and mapping of the gene to 6p23-25. Genomics 37:229-233.
- Eisenbeis CF, Singh H, Storb U (1995) Pip a novel IRF family member, is a lymphoid-specific, PU.1-dependent transcriptional activator. Genes Dev 9: 1377-1387.
- Matsuyama T, Grossman A, Mittrucker HW, Siderovski DP, Kiefer F, et al. (1995) Molecular cloning of LSIRF, a lymphoid-specific member of the interferon regulatory factor family that binds the interferon-stimulated response element ISRE. Nucl Acids Res 23: 21-27.
- Yamagata T, Nishida J, Tanaka T, Sakai R, Mitani K, et al. (1996) A novel interferon regulatory factor family transcription factor, ICSAT/Pip/LSIRF that negatively regulates the activity of interferon-regulated genes. Mol Cell Biol 16: 1283-1294.
- Ortmann CA, Burchert A, Hölzle K, Nitsche A, Wittig B, et al. (2005) Down-regulation of interferon regulatory factor 4 gene expression in leukemic cells due to hypermethylation of CpG motifs in the promoter region. Nucleic Acids Research 33: 6895-6905.
- Do TN, Ucisik-Akkaya E, Davis CF, Morrison BA, Dorak MT (2010) An intronic polymorphism of IRF4 gene influences gene transcription in vitro and shows a risk association with childhood acute lymphoblastic leukemia in males. Biochim Biophys Acta 1802: 292-300.
- Nishiya N, Yamamoto K, Imaizumi Y, Kohno T, Matsuyama T (2004) Identification of a novel GC-rich binding protein that binds to an indispensable element for constitutive IRF-4 promoter activity in B cells. Mol Immunol 41: 855-861.
- Rowley J (1973) A new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and Giemsa staining [letter]. Nature 243: 290-293.
- Kawajiri K, Nakachi K, Imai K, Watanabe J, Hayashi S (1993) The CYP1A1 gene and cancer susceptibility. Crit Rev Oncol Hematol 14: 77-87.
- Acquaviva J, Chen X, Ruibao R (2008) IRF-4 functions as a tumor suppressor in early B-cell development. Blood 112: 3798-3806.
- Schmidt M, Andreas H, Sven A, Merediz K, Cornelia B, et al. (2000) Expression of Interferon regulatory factor 4 in chronic myeloid leukemia: correlation with response to interferon alfa therapy. J Clin Oncol 18: 3331-333.
- Ortmann C, Burchert A, Holzle K, Nitsche A, Wittig B, et al. (2005). Down-regulation of interferon regulatory factor 4 gene expression in leukemic cells due to hypermethylation of CpG motifs in the promoter region. Nucleic Acids Res 33: 6895-6905.
- Mendizabal AM, Anderson WF, Garcia-Gonzalez P, Levine PH (2010) Differing age patterns in chronic myeloid leukemia by racial ethnic group. J Clin Oncol 28: 1577.
- Kurzrock R, Gutterman JU, Talpaz M (1988) The molecular genetics of Philadelphia chromosome-positive leukemias. N Engl J Med 319: 990-998.
- Goldman JM (2007) How I treat chronic myeloid leukemia in the imatinib era. Blood 110: 2828-2837.