Research Article
Study of Acoustical and Physico-Chemical Properties on the Binary Mixture of Cyclopentyl Methyl Ether (Cpme) and Methyl Acrylateat 298.15 K Temperature
Dwivedi S*
Department of Chemistry, DBS (PG) College, Dehradun, Uttarakhand, India
*Corresponding author: Dwivedi S, Department of Chemistry, DBS (PG) College, Dehradun, Uttarakhand, India; E-mail:
somdwivedi5@gmail.com
Copyright: © 2022 Dwivedi S. This is an open access article distributed under the Creative Commons Attribution License,
which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 07/04/2022; Accepted: 08/05/2022; Published: 10/06/2022
Abstract
Acoustical and Physico-chemical properties of liquid mixtures and solutions are playing very important role in understanding the nature of intermolecular
interactions. Excess function; have been used as a qualitative and quantitative guide to predict the extent of complex formation in binary liquid mixtures. In
this paper evaluated experimental densities (ρ) and ultrasonic velocities (U) for the pure liquids and liquid mixture of Cyclopentyl methyl ether and Methyl
acrylate using bicapillary pycnometer and single frequency variable path over the different concentration range at 298.15 K Temprature. The observed data
had been utilized to calculate various acoustical parameters like isentropic compressibility (KS), Intermolecular free length (Lf) and acoustic impedance (Z).
Excess ultrasonic velocities (UE), Excess Isentropic compressibility (KS
E), Excess Intermolecular free length (Lf
E) and Excess acoustic impedance (ZE) were
calculated using the measured values and correlated with the Redlich-Kister polynomial equation. The observed variations of the properties for the above
mixture conclude that the interactions between unlike molecules predominate over the dissociation effects in the individual components. It is also evident that
the presence of strong interactions between unlike molecules is predominant and characterized by the deviations of Excess acoustical parameters.
Keywords
Acoustic impedance; Density; Inter molecular free-Length; Isentropic compressibility; Ultrasonic velocity
Introduction
Due to recent developments made in the theories of liquid
mixtures and experimental techniques, the study of binary liquid
mixtures, has attracted several researchers in the field [1]. The
prediction of the physicochemical parameters of liquid mixtures is a
goal of long standing, with both theoretical and practical importance.
A truly fundamental theory would predict the parameters along with
other thermodynamic and transport properties from the knowledge
of the intermolecular forces and radial distribution function alone.
Such a programme has had appreciable success in application to
pure simple liquids such as the liquefied rare gases [2], for solutions
however although the general theory has been formulated, It has
not been reduced successfully to numerical results continuing a
study of the effect of molecular structure on refractive index-density
relationships [3], mixtures of the three possible combinations of the
aromatics compounds were investigated The effect of composition
and temperature on refractive index dispersion and density
measurement were presented for the mixture at 20, 30 and 40oC.
Density measurement provided a satisfactory means for analysing for
this system.
In chemical process industries, the materials are normally
handled in fluid form and as a consequence, the physical chemical,
and transport properties of fluids assume importance. Thus data
on some of the properties associated with the liquids and liquid
mixtures like density, viscosity and ultrasonic velocity, to find
extensive application in solution theory and molecular dynamics [4]. Such results are necessary for interpretation of data obtained from
thermo chemical, electrochemical, biochemical and kinetic studies
[5]. During the last two decades, ultrasonic study of liquid mixtures
has gained much importance in assessing the nature of molecular
interactions through the study of the physico chemical properties of
such systems. Ultrasonic velocity and related data of liquid mixtures
are found to be the most powerful tool in testing the theories of
liquid state. In addition, ultrasonic velocity data can be utilized to
deduce some useful properties of liquid mixtures which are not easily
accessible by other means [6]. The measurement of ultrasonic velocity
has been adequately employed as a versatile tool for investigating
the physical properties of matter-solid, liquid and gas. These studies
are very important because of their extensive use in textile industry,
leather industry, and pharmaceutical industry and in many others.
Ultrasonic velocity measurement has proved useful in dealing with
the problems of liquid structure and molecular interactions in liquid
mixtures [7].
The practical importance of liquid mixtures rather than single
component liquid systems has gained much importance during the
last two decades in assessing the nature of molecular interaction
sand investigating the physico-chemical behavior of such systems
[8]. Thermodynamic investigation of liquid mixtures consisting of
polar and non-polar components are of considerable importance in
understanding inter molecular interactions between the component
molecules and they find applications in several industrial and
technological processes [9]. These studies are very important because
of their extensive use in textile industry, leather industry, and
pharmaceutical industry and in many others. Acoustical parameters
have proved useful in dealing with the problems of liquid structure
and molecular interactions in liquid mixtures.
Methyl acrylate is a very important industrial chemical and is
widely used commercially for the production of technically important
high polymeric and latex compounds. It is polar (dipole moment, μ =
1.77 D at 298.15 K) and strongly associated aprotic solvent due to the
presence of polar carbonyl group in the molecule and it is a versatile
liquid which finds use as a monomer in the preparation of poly
(Methyl acrylate) which has innumerable industrial applications [10].
Cyclopentyl methyl ether (CPME) also known as methoxy
cyclopentane, is a totally new hydrophobic ether solvent. It have
high boiling point of 106 °C (223 °F) and preferable characteristics
such as low formation of peroxides, relative stability under acidic and
basic conditions, formation of azeotropes with water coupled with a
narrow explosion range render CPME an alternative to other ethereal
solvents such as tetrahydrofuran (THF), 2-methyltetrahydrofuran
(2-MeTHF), dioxane, and 1,2-dimethoxyethane (DME). Cyclopentyl
methyl ether is used in organic synthesis, mainly as a solvent. However
it is also useful in extraction, polymerization, crystallization and
surface coating. The utilization of CPME in several organic reactions
pertaining to organometallic-, organo-, and biocatalysis. This solvent
have favourable physical and chemical characteristics, such as low
toxicity and high chemical and thermal stabilities, which make them
promising media for a large breadth of synthetic chemistry and
triggers development of reactions with green chemistry protocol [11-12]. The objective underlying the present work is to obtain information regarding molecular interactions in mixtures of a highly polar liquid
with non-polar or weakly polar liquids [13]. CPME (Cyclopentyl
methyl ether) is an important green solvent. Thus, a study of physical
properties data on the binary mixture containing CPME has attracted
considerable interest in the literature. Thus, Methyl acrylate in
CPME mixed solvent would enable us to have a large number of
solvents with appropriate physico-chemical properties, which can be
used for a particular chemical process. Moreover, literature survey
indicates that no study on this binary system has been reported at
298.15 K. Therefore, present study was undertaken in order to have
deeper understanding of the intermolecular interaction between the
components of the above binary liquid mixture.
Material & Methods
Cyclopentyl methyl etherand Methyl acrylate were purchased
from Sd fine chemicals India. Mixture was prepared by mixing
weighed amounts of the pure liquids adopting the method of closed
system by using Mettler balance with the precision of ± 0.1 mg.
Mixture was allowed to stand for some time before every measurement
so as to avoid air bubbles. The purities of the liquids were checked
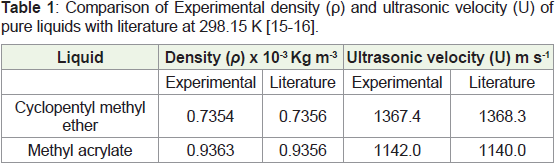
by comparing the values of densities and ultrasonic velocities with
literature data and are given in Table 1 & 2 . The measurements were
made with proper care in an AC room to avoid evaporation loss.
The densities (ρ) of liquids and their mixture were measured using
bicapillary pycnometer having a capillary diameter of 0.85 mm, which
was calibrated using double distilled water. The necessary buoyancy
corrections were applied. The density values were reproducible within
± 0.2 Kg m-3. The ultrasonic velocity (U) measurements were made
by a single frequency (2 MHz) variable path.
Table 1: Comparison of Experimental density (ρ) and ultrasonic velocity (U) of
pure liquids with literature at 298.15 K [15-16].
Results & Discussion
From the measured densities (ρ) and ultrasonic velocities
(U) the various acoustical parameters such as KS, Z and Lf were
calculated using the following equations 1, 2 & 3 respectively and are
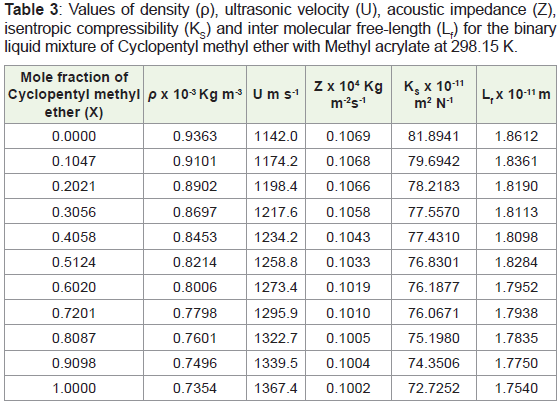
incorporated in Table 3, for the binary system under study [14-17].
Table 3: Values of density (ρ), ultrasonic velocity (U), acoustic impedance (Z),
isentropic compressibility (KS) and inter molecular free-length (Lf) for the binary
liquid mixture of Cyclopentyl methyl ether with Methyl acrylate at 298.15 K.
Where ‘KT’ is Jacobson’s constant. It is temperature-dependent
empirical constant, proposed by Jacobson in 1952 and given as
KT= (93.87 5 + 0.375 × T) × 10−8 at temperature T [18]. The excess
functions YE are calculated using the relation:
Where Y denotes U, Z, KS and Lf respectively, X is the mole
fraction and suffixes 1 & 2 denote the components 1 & 2 in binary
liquid mixture and the values are given in Table 4. The dependence of
UE, ZE, KS
E and LS
E on the mole fraction of Cyclopentyl methyl ether
for liquid mixture was fitted to the following Redlich-Kister equation
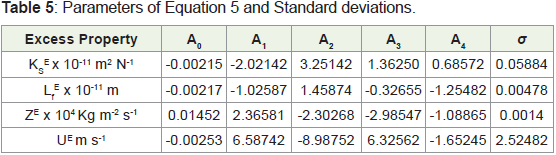
by the least-squares method and the values are given in Table 5 [19].
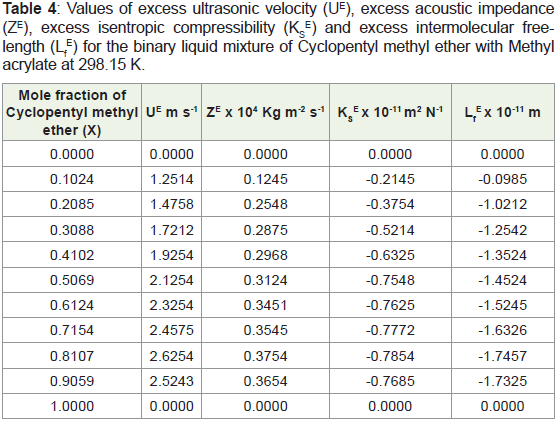
Table 4: Values of excess ultrasonic velocity (UE), excess acoustic impedance
(ZE), excess isentropic compressibility (KS
E) and excess intermolecular free length
(Lf
E) for the binary liquid mixture of Cyclopentyl methyl ether with Methyl
acrylate at 298.15 K.
Where YE is UE, ZE, KS
E and Lf
E parameters. The parameters Ai,
obtained by a nonlinear least squares polynomial fitting procedure,
are also given in Table 5 together with the standard deviations (σ)
values. From Table 3, it is observed that the values of U, Z, Ks and Lf
varied linearly with the mole fraction of Cyclopentyl methyl ether.
This indicates the presence of interactions between the components
in this binary liquid mixture.
With this view in mind, the variations in excess acoustical
parameters, like the excess ultrasonic velocity (UE), excess Acoustic
impedance (ZE), excess isentropic compressibility (KS
E) and excess
intermolecular free-length (Lf
E) with the mole fraction of Cyclopentyl
methyl ether is examined from the Table 4. It is observed that
the positive deviations in UE and ZE for in the system standby are
observed over the entire range of composition due to orientation
of liquids. The negative deviations observed of KS
E and Lf
E due to
little dipolar association between their molecules anticipated dipole
moment of Cyclopentyl methyl ether indicate significant interactions
between Methyl acrylate and Cyclopentyl methyl ether molecules
forming dipole dipole interaction. In contrast, there is possibility
of the electron donor acceptor (charge-transfer) type interactions
[20,21]. The mole fraction of Cyclopentyl methyl ether is increased
with decreased density due to size and shape of molecules more
hindered. The interactions are minor intensity of Cyclopentyl methyl
ether and Methyl acrylate molecules; they can lead to in the term of
short range forces exist while the long range forces are negligible.
Cyclopentyl methylether and Methyl acrylate liquid mixture is more
compressible due to structural effects and dispersive type interaction.
This study helps to understand the effect of the molecular interaction on behavior of liquid mixtures of polar and nonpolar liquids clearly
and concisely.
Conclusion
In the present research workcalculated various excess
parameters like Ultrasonic velocity (UE), Isentropic compressibility
(KS
E), Intermolecular free length (Lf
E), acoustic impedance (ZE) of
Cyclopentyl methyl ether and Methyl acrylate liquid mixture and
investigation on the acoustical and physico-chemical Properties. It is
observed from the measured data that the values of UE, ZE are positive
deviations and KS
E, Lf
E, are negative deviations in the mixture and
the variation of the properties of the mixture studied supports the
view that the strong interactions between molecules predominate
over the dissociation effects in the individual components and nature,
molecular geometry, concentration of mixture.
Future Aspects
The purpose of the present work should focus on the future.
Theoretical values of acoustical and physico-chemical parameters of
this binary liquid mixture has been compared with experimental
data to verify the applicability studied.
Acknowledgment
The author is thankful to the authorities of D. B. S. (P. G.) College,
Dehradun, Uttarakhand for providing research facilities to carry out
this work. This research did not receive any specific grant from any
funding agencies.
References
Citation
Dwivedi S. Study of Acoustical and Physico-Chemical Properties on the Binary Mixture of Cyclopentyl Methyl Ether (Cpme) and Methyl
Acrylateat 298.15 K Temperature. J Chem Applied Biochem. 2022;6(1): 124.