Research Article
Synthesis of 3,4-Dihydro-2(1H)-PyrimidonesVia Biginelli Reaction in Organized Media Using Ionic Liquids and Solid Acid Catalysts
Pranab Jyoti DAS*, Jesmin BEGUM
Department of Chemistry, Gauhati University, Guwahati 781014, India
Corresponding author: Pranab Jyoti DAS, Department of Chemistry, Gauhati University, Guwahati 781014, India; E-mail: pranabjdas52@gmail.com
Citation: DAS PJ, BEGUM J. Synthesis of 3,4-Dihydro-2(1H)-Pyrimidones Via Biginelli Reaction in Organized Media Using Ionic Liquids and Solid AcidCatalysts. J Chem Applied Biochem. 2015;2(1): 116.
Copyright © 2015 DAS PJ et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
31/10/2015; Accepted: 26/11/2015; Published: 04/12/2015
Abstract
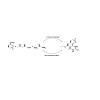
5-Ethoxycarbonyl-4-aryl-6-methyl-3,4-dihydropyrimidin-2(1H)-one have been synthesized in good yield by the condensation of aromatic aldehydes,ethylacetoacetate and urea in the presence of acidic ionic liquids derived from dialkylamines as the catalyst under thermal heating in the absence of VOC.The synthesis of the target molecules revisited using acidic ion exchange resin namely Amberlite IR 120(H+) in a solid phase reaction. The two methodscompared with respect to yields obtained, reaction time, ease of separation of products, cost and recyclability of the catalysts.
Keywords: Aromatic aldehyde; Solvent free reactions; Ionic liquids; Amberlite IR120(H+); Acetoacetic ester; Urea
Introduction
Functionalized dihydropyrimidine (DHMP) scaffold, popularlytermed as “Biginelli compounds” is a heterocyclic system exhibitingremarkable pharmacological efficiency. Dihydropyrimidines displaya broad range of biological effects, including antiviral, antitumor,antibacterial, and anti inflammatory properties. The recognizedpharmacophore is reported to be the partly reduced pyrimidinemoiety [1]. The versatile end use of DHPMs have led to the recentdevelopment of several improved reaction protocols for the synthesisof DHPMs with a variety of substituents in the basic unit, either bymodification of the classical one-pot Biginelli approach [2-4], or bythe development of novel, but more complex multistep strategies[5-7]. Further, several combinatorial approaches towards synthesisof DHPMs have been reported using solid phase techniques [8-10].The use of Cu (NO3)2 deposited on Mont-morillonite K 10 clay in themicrowave-assisted solvent-free condition is a noteworthy example[11]. In general, Lewis acids are used as the catalyst although the useof a variety of other catalysts have been reported. Notable among the catalysts used to drive the reaction are zeolites HZSM-5 and MCM-41 [12], ionic liquids [13], Mg(NO3)2 [14], DDQ [15] and a variety ofTriflates [16,17], Zn( BF4)2 [18], natural acid [19], 12-phosphotungsticacid besides others [20,21-23].
In this investigation, we explored the possibility of synthesisof the functionalized DHPMs namely the 5-Ethoxycarbonyl-4-aryl-6-methyl-3,4-dihydropyrimidin-2(1H)-ones using simpleacidic ionic liquids derived from dialkylamines. Ionic liquids aretermed as neoteric liquids and are recognized as environmentallyharmless media because of their low vapour pressure, high thermaland chemical stability and excellent solubilising characteristics.Their low toxicity, recyclability, air stability and excellent solubilityboth in water as well as in most organic solvents contribute to theirversatility as catalysts. Their unique properties resulted in their wideacceptance as media in many reactions and as catalysts as well. Thedual role played by ionic liquids and their influence on selectivityin reactions have further enhanced their utility [24-32]. Among thewide range of ionic liquids, the salts of imidazolium, ammonium, thiazolium cations are especially popular and simple methods oftheir synthesis and characterization have been reported [33-37].Their applications in organic synthesis have been widely explored.However, the use of some of these IL is less attractive due to theirhigh cost. Thus it is necessary to explore possibilities of preparing lessexpensive IL which may exhibit comparable, if not better, utility ascatalyst and/or solvent. In this work we explored the possibility ofthe use of acidic ionic liquids derived from dialkylamines both as acatalyst and solvent for the three component solvent free reaction ofaromatic aldehydes, ethylacetoacetate and urea for the synthesis of3,4-dihydro-2(1H)-pyrimidones. Further, the reaction was revisitedby using a solid acid catalyst for the condensation and the merits ofthe two environmentally benign processes examined.
Results and Discussions
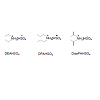
During the course of our study in developing cost effective ionicliquids for the synthesis of 3,4-dihydro-2(1H)-pyrimidones by theBiginelli condensation reaction, we prepared acidic ionic liquids fromeasily available dialkylamines and used them as catalyst in a facile threecomponent reaction of aromatic aldehydes, ethylacetoacetate and ureaas substrates for the synthesis of 3,4-dihydropyrimidin-2(1H) onesin appreciably reduced reaction time as well as in high yield. Threedifferent ionic liquids, namely diethylammoniumhydrogensulphate(DEAHSO4), di-n-propylammoniumhydrogensulphates,(DPAHSO4), diisopropylammoniumhydrogen- sulphate,(DisoPAHSO4) and were prepared by a reported procedure [37].Scheme 1 gives the structures of the IL synthesized.
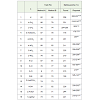
Thermogravimetric analysis showed that all the three ionicliquids are stable and their thermal stability decreases accordingto the sequence [(disopropyl)2NH2]HSO4 [Et2NH2]HSO4>[(n-propyl)2NH2]HSO4 32. DisoPAHSO4 being thermally the moststable, we chose to use this ionic liquid in all the synthesis reportedhere. A trial run was carried out by using 4-methoxybenzaldehyde,ethylacetoacetate and urea in 5mmol of the ionic liquid and theproduct 5-ethoxycarbonyl-4-(4/-methoxyphenyl-6-methyl-3,4-dihydropyrimidin-2(1H)-one was recovered in 88%. Reactionconditions were optimized by varying the mol % of ionic liquid,reaction temperature and reaction time. Results obtained indicatedthat best results were obtained at the reaction temperature of 80 °Cin about 30 mins reaction time using 5 mmol of the ionic liquid. Theresults are summarized in Table 1.
Increase of reaction temperature and time beyond the optimumdid not significantly increase the yields. Consequently, all reactionswere carried out at the optimized reaction conditions mentioned. Alibrary of 5-Ethoxycarbonyl-4-aryl-6-methyl-3,4-dihydropyrimidin-2(1H)-one were synthesized by varying the aromatic aldehydes.
In addition to the use of IL as catalyst for the synthesis of the target molecules, we carried out a solid phase synthesis of targetmolecule using Amberlite IR120 (H+) as the solid acid catalyst in anenvironmentally benign reaction using ethanol as the solvent. Thissolid phase synthesis was also standardized by varying the solidacid catalyst. 5 g of the solid acid was found to give best yield of thetarget molecule. Both the reaction conditions have been comparedwith respect to the yield of the product obtained, reaction conditionused, the method of recovery of products and also the extent ofenvironmental acceptability. Both the procedures conform to thetenets of green techniques in organic synthesis. In case of the use ofacidic ionic liquids, no volatile organic solvent was found necessaryas the acidic ionic liquids performed the dual role of a solvents wellas the catalyst. In case of the reaction condition using the solid acid,organic solvent had to used in order to facilitate reaction at the surfaceof the catalyst. In the absence of a solvent the reaction gave severalpolymeric products which could not be characterized. Use of organicsolvents such as DMF, CH2Cl2, dioxane, methanol and ethanol wereexplored and it was observed that solvents had no effect on the yieldand reaction temperature consequently the benign solvent ethanolwas selected as the convenient solvent. The reaction is shown in
The results obtained on using both the ionic liquid and the acidicion exchange resin are summarized in Table 2.
After optimization of the conditions, the reaction was examinedfor its generality by using a series of aromatic aldehydes under optimalcondition and the results are presented in Table 2.
Work up is simple as the ionic liquid could be easily removedfrom the reaction mixture by washing the products with water. Thereaction however, failed to give good yields with aliphatic aldehydes.
Experimental Section
Melting points were recorded in a VMP-D model Melting point apparatus and are uncorrected. UV-Vis spectra were recorded inShimatzu FT-IR recorded in Perkin Elmar 1600 spectrometer usingKBr pellets, 1H and 13C NMR spectra were recorded in Bruker300MHz spectrometer in CDCl3 and DMSO-d6 and TMS as internalstandard. Aldehydes used were recrystallized by reported proceduresbefore use. Mass spectra were recorded in a Waters Micromass ZQ TM400 mass spectrometer. The thermal decomposition point of the ionicliquids were determined in a TGA-DSC1, Mettler Toledo instrumentheating in a stream of nitrogen atmosphere. All reagents werepurified by standard literature procedure. The dialkylamine basedionic liquids were prepared according to the procedure reportedearlier32.
Thermogravimetric Analysis (TGA) of ionic liquids: Thesamples were weighed and placed in a platinum crucible. Theywere then heated in a stream of nitrogen atmosphere, from roomtemperature to 700 °C with a heating rate usually of 10 °C /min. The thermal stability is seen to vary with the nature of the cation of ILs.The thermal stability increases in the following sequence of the ILs,di-n-propyl ammonium hydrogen sulphate < diethyl ammoniumhydrogen sulphate < di-isopropyl ammonium hydrogensulphate.
Diethylammonium hydrogensulphate [Et2NH2][HSO4]:prepared by reported method 1H NMR (300 MHZ, DMSO, Me4Si) (δH ppm) : 1.148 (6 H, t, J= 7.2 Hz, 2 CH3), 2.890 (4 H, t, J=7.2 Hz, 2CH2), 3.777 (2 H, s, NH2), 8.347 (1 H, s, HSO4) ; 13C NMR (75 MHz,DMSO-d6) ( δ ppm): 11.30, 41.73 ; The thermal decomposition point578K.
Di-n-propylammonium hydrogensulphate [n-Pr2NH2][HSO4]:1H NMR (400 MHZ , DMSO , Me4Si) ( δH ppm): 0.095 ( 6 H, t, J=6.8 Hz, 2 CH3), 1.602 ( 4 H, sextet, J= 7.2 Hz, 2 CH2), 2,841 (4 H, m, 2CH2), 4.447 (2 H, s, NH2), 8.297 (1 H, s, HSO4) ; 13C NMR (100 MHz,DMSO-d6) ( δ ppm) : 11.349, 19.413, 48.816 ; HRMS(ESI) m/z calcdfor C6H17NO4S [M]+ (199.0878), found ( 199.0879).; The thermaldecomposition point 567 K.
Diisopropylammonium hydrogensulphate[iso-Pr2NH2[HSO4]:1H NMR (300 MHZ, DMSO, Me4Si) ( δH ppm) : 1.191 (12 H, d, J=6.3Hz, 4 CH3) 3.305 (2 H, m, 2 CH), 4.975 (2 H, s, NH2), 8.136 (1 H,s, HSO4) ; 13C NMR (75 MHz, DMSO-d6) ( δ ppm): 18.96, 46.56 ;HRMS(ESI) m/z calcd for C6H17NO4S [M]+ (199.0878), found (199.0879). The thermal decomposition point 591K.
General procedure for the synthesis of 5-ethoxycarbonyl-6-methyl-4-substituted-3,4-dihydropyramidin-2(1H)-one usingdiisopropylammoniumhydrogensulphate.
An equimolar mixture of aromatic aldehyde (1.0 mmol),ethylacetoacetate (1.0 mmol) , urea (1.0 mmole) and 5 mmol ofthe acidic diisopropylammoniumhydrogen sulphate was mixedthoroughly in pestle grinder and the homogeneous mixture washeated in an oil bath to 80 °C in the absence of solvent. After 30 minsof heating a distinct change in colour of the reaction mixture to yellowwas observed. The yellow solid mass was washed several times withwater till free of the ionic liquid. The crude product was extractedwith CH2Cl2, dried over anhydrous Na2SO4 and solvent evaporated toobtain the product.
General procedure for the synthesis of 5-ethoxycarbonyl-6-methyl-4-substituted-3,4-dihydropyramidin-2(1H)-one usingsolid acid, Amberlite IR 120( H+) :
An equimolar mixture of aromatic aldehyde (1.0 mmol),ethylacetoacetate (1.0 mmol), and urea (1.0 mmole) was dissolved in50 mL ethanol ( 95% v/v). To this solution 5 g of acidic ion exchangeresin ( Amberlite IR 120H+ ) was added and the heterogeneousmixture was refluxed with constant stirring for about 30 mins. Achange in colour of the reaction mixture to light yellow was observed.The reaction was monitored by using TLC in prepared silica gel platesusing petroleum ether ( 60-80) : ethylacetate : 10:1 as the eluent. Aftercompletion of the reaction, the resin beads were filtered out andevaporation of the ethanol gave the product. The recovered resin waswashed wit dil.HCl and reused. The dihydropyramidin-2(1H)-one An equimolar mixture of aromatic aldehyde (1.0 mmol),ethylacetoacetate (1.0 mmol), and urea (1.0 mmole) was dissolved in50 mL ethanol ( 95% v/v). To this solution 5 g of acidic ion exchangeresin ( Amberlite IR 120H+ ) was added and the heterogeneousmixture was refluxed with constant stirring for about 30 mins. Achange in colour of the reaction mixture to light yellow was observed.The reaction was monitored by using TLC in prepared silica gel platesusing petroleum ether ( 60-80) : ethylacetate : 10:1 as the eluent. Aftercompletion of the reaction, the resin beads were filtered out andevaporation of the ethanol gave the product. The recovered resin waswashed wit dil.HCl and reused. The dihydropyramidin-2(1H)-one were recrystallized from either methanol or ethanol.
Conclusion
In conclusion, an efficient solvent free method is developed forthe synthesis of 5-ethoxy- carbonyl-6-methyl-4-substituted-3,4-dihydropyramidin-2(1H)-one using both the acidic ionic liquidderived from dialkylamines and the solid acid Amberlite IR 120 ( H+).The recovery of product in both the cases is easy. The ionic liquidsused can be obtained from cheap sources, they are easy to handle andenvironmentally benign and recovery of products did not warrantthe use of organic solvents. The use of ionic liquid offers a solventfree reaction and yields are found to be higher as compared to thoseobtained by using the ion exchanger as the catalyst.
Spectroscopic data of selected compounds:
1. 5 - e t h o x y c a r b o n y l - 6 -me t h y l - 4 - p h e n y l - 3 , 4 -dihydropyramidin-2(1H)-one ( entry1): light yellow, UV (in EtOH 95%V/V , λmax nm ): 295, 346, 1H-NMR ( CDCl3) : δ9.18(s,1H), 7.73(s,1H), 7.32-7.23(m. 5H aromatic), 5.13 ( d, J=4.2Hz, 1H), 3.92-3.99 ( q, J=7.1 Hz, 2H), 2.23 (s, 3H), 1.09-1.05(t,3H), 13C(75 MHz): 165.38, 152.18,148.4,144.8,128.43, 127.31,126.28,99.29, 59.24,53.99, 17.81, 4.11; IR ( KBr) : ν cm-1 3244,3116, 1724, 1701.
2. 5-ethoxycarbonyl-6-methyl-4(4-methylphenyl)-3,4-dihydropyramidin-2(1H)-one ( entry2): light yellow ; UV (in EtOH 95%V/V, , λmax nm): 298, 342; 1H-NMR ( DMSO-d6): δ 9.15(s,1H), 7.68(s,1H), 7.1 (m,4Haromatic), 5.09(1H), 3.97-3.92 ( q, J=6.8Hz, 2H), 2.22 ( d, 3H), 2.3 (d, 3H), 1.04-1.11(t,J=7.8Hz, 3 H); 13C(75 MHz);165.37, 152.2, 148.17, 141.96,136.38, 128.9, 126.16, 99.41, 59.17, 53.63, 20.65, 17.77, 14.1;IR ( KBr) : ν cm-1 3354, 3224, 2326, 1695, 1645.
3. 5-ethoxycarbonyl-6-methyl-4-(3,4-dimethoxyphenyl)-3,4-dihydropyrimidin-2(1H)-one ( entry 4) : light yellow ; UV (in EtOH 95%V/V, , λmax nm): 298, 352 ; 1H-NMR ( CDCl3) : δ9.13(s,1H), 7.66(s, 1H), 6.88-6.69(m, 3Haromatic ), 5.084(s,1H),4.01-3.96(t,2H), 3.698(s,6H), 2.226(s,3H), 1.12-1.07(t,3H); 13C(75 MHz): 159.48, 146.29, 142.51, 142.49, 93.42, 53.24,49.58, 49.46, 47.51, 11.81, 8.22 IR ( KBr) : ν cm-1 3251, 3118,1716, 1681.
4. 5-ethoxycarbonyl-6-methyl-4-(4-methoxylphenyl)-3,4-dihydropyrimi- din -2(1H)-one ( entry 5): light yellow , UV( in EtOH 95%V/V, , λmax nm):299, 329; 1H-NMR ( DMSO-d6): δ 9.19 (s,1H), 7.59(s,1H), 7.15-6.81(m,4H), 5.00(s,1H),3.89(q, J=6.7 Hz, 2H), 3.70(s,3H), 2.19(s,3H), 1.07(t,J=6.7,3H); 13C(75 MHz):165.1, 157.9,151.8,148.3,137.5, 126.8,112.7, 99.2, 58.9, 54.9, 17.9; IR ( KBr) : ν cm-1 3400, 3232,1745, 1685.
5. 5-ethoxycarbonyl-6-methyl-4( 4- nitrophenyl)-3,4-dihydropyrimidin-2(1H)-one ( entry6): light yellow ; UV( in EtOH 95%V/V, , λmax nm): 295, 336; 1H-NMR ( CDCl3): δ 8.7(br ,s,1H), 7.3(br,s,1H), 7.31-7.33( t, 2Haromatic ), 7.92-7.95 ( m, 2Haromatic ), 5.22 (d, 2.6Hz, 1H), 3.82-3.89 ( q,2H),2.14(s,3H), 0.98-0.94(t, J=7.68, 3H) ; 13C NMR(75 MHz):165.1, 152.29, 148.4, 146.58, 127.21, 123.22, 59.43, 54.03, 18.04, 13.71. IR ( KBr) : ν cm-1 3390, 3232, 1712, 1633, 1456.
6. 5-ethoxycarbonyl-6-methyl-4(4-chlorophenyl)-3,4-dihydropyrimidin-2(1H)-one ( entry 9): light yellow ; UV( in EtOH 95%V/V, , λmax nm): 298, 335 ; 1H-NMR ( CDCl3) :δ 8.6(s,1H), 7.02 (s,1H), 6.83-6.91(m, 4H), 5.03-5.02(d, 1H),3.80-3.73(q,3H), 2.05(s,3H), 0.97-0.86(q, 3H); 13C ( 75 MHz) :165.1, 152.26, 147.46, 142.68, 132.2,127.7,99.2,77.43,59.03,53.77,17.74,13.54; IR ( KBr) : ν cm-13356, 3240,1701,1543.
7. 5-ethoxycarbonyl-6-methyl-4(3-bromophenyl)-3,4-dihydropyrimidin-2(1H)-one ( entry 12): light yellow ; UV (in EtOH 95%V/V, , λmax nm): 294, 339; 1H-NMR ( DMSO-d6): δ 9.21(s, 1H), 7.79 (s, 1H), 7.45-7.29(m, 4H), 5.13 ( d, J=5.6Hz, 1H), 4.01(t, 2H), 2.48-2.23 (t,3H), 1.16-1.05(t, 3H); 13C(75 MHz) : 165.13, 151.91, 148.97,147.49, 130.83, 130.16,129.18, 125.28, 121.53, 98.6, 59.32, 53.59, 17.83, 14.05; IR (KBr) : ν cm-1 3344, 3213, 1658.7, 1608.63.
8. 5-ethoxycarbonyl-6-methyl-4(2-bromophenyl)-3,4-dihydropyrimidin-2(1H)-one( entry 13): light yellow ; UV (in EtOH 95%V/V, , λmax nm): 298, 338 1H-NMR ( DMSO-d6): δ 9.28(s, 1H), 7.70(s,1H), 7.56-7.14(m, 4Haromatic), 5.60-5.91( d, J=7.1Hz, 2H), 3.91-3.84 ( t, 2H), 2.28 (s, 3H), 1.0-0.95 (t,3H); 13C NMR ( 75 Hz): 164.97, 151.27,149.3, 143.4, 132.6,129.4, 128.79, 128.47, 122.28, 98.26, 59.08, 54.01, 17.67, 14.00;IR ( KBr) : ν cm-1 3344 , 3223, 1697, 1633.
9. 5 - e t h o x y c a r b o n y l - 6 -me t h y l - 4 - n a p t h y l - 3 , 4 -dihydropyrimidin-2(1H)-one ( entry 16) : light yellow ; UV( in EtOH 95%V/V, , λmax nm): 286, 329; 1H-NMR ( CDCl3): δ 9.25(s,1H), 8.26(d,J=7.1Hz,1H), 7.91(s,1H), 7.84(d,J=8.1,1H), 7.75(s,1H), 7.37-7.57(m, 4H aromatic), 6.03(s, 1H), 3.73-3.8(m,2H), 2.48(s,3H), 0.76-0.81(t, 3H) ; 13C NMR( 75 MHz): δ 159.3, 145.75, 142.8, 134.41, 127.5, 124.08, 122.51, 121.98,120.13, 119.78, 119.72, 118.25, 117.67, 93.18, 53.12, 43.76,11.82, 7.78; IR ( KBr) : ν cm-1 3410, 3245, 1726 ,1654.
References
- Kappe CO (2000) Biologically active dihydropyrimidones of the Biginelli-type--a literature survey. Eur J Med Chem 35, 1043- 1052.
- Lu J, Ma H (2000) Iron(III)-Catalyzed Synthesis of Dihydropyrimidinones. Improved Conditions for the Biginelli Reaction. Synlett 2000: 63- 64.
- Hu EH, Sidler DR, Dolling UH (1998) Unprecedented Catalytic Three Component One-Pot Condensation Reaction:  An Efficient Synthesis of 5-Alkoxycarbonyl- 4-aryl-3,4-dihydropyrimidin-2(1H)-ones. J Org Chem 63: 3454-3457.
- Gupta R, Gupta AK, Paul S, Kachroo PL (1995) Ind J Chem 34B: 151-153.
- O'Reilly BC, Atwal KS (1987) Synthesis of Substituted 1,2,3,4-Tetrahydro-6-methyl-2-oxo-5-pyrimidinecarboxylic Acid Esters: The Biginelli Condensation Revisited. Heterocycles 26, 1185-1188.
- Atwal KS, O’ Reilley BC, Gougoutas JZ, Malley MF (1987) Synthesis of Substituted 1,2,3,4-Tetrahydro-6-methyl-2-thioxo-5-pyrimidinecarboxylic Acid Esters. Heterocycles 26: 1189-1192.
- Shutalev AD, Kishko EA, Sivova N, Kuznetsov AY (1998) A New Convenient Synthesis of 5-Acyl-1,2,3,4-tetrahydropyrimidine-2-thiones/ones. Molecules 3: 100-106.
- Wipf P, Cunningham A (1995) Tetrahedron Lett 36: 7819-7822.
- Kappe CO (2000) Highly versatile solid phase synthesis of biofunctional 4-aryl-3,4-dihydropyrimidines using resin-bound isothiourea building blocks and multidirectional resin cleavage. Bioorg Med Chem Lett 10: 49-51.
- Studer A1, Jeger P, Wipf P, Curran DP (1997) Fluorous Synthesis: Fluorous Protocols for the Ugi and Biginelli Multicomponent Condensations. J Org Chem 62: 2917- 2924.
- Diwaker S, Rakshit A, Punjabi PB, Ameta SC (2010) Int J Chem Sci 8: 1973-1982.
- Radha Rani V, Srinivas N, Radha Kishan M, Kulkarni SJ, Raghavan VK (2001) Zeolite-catalyzed cyclocondensation reaction for the selective synthesis of 3,4-dihydropyrimidin-2(1H)-ones. Green Chem 3: 305-306.
- Boumoud T, Boumoud B, Rhouati S, Belfaith A, Deache A, Mosset P (2008) Acta Chim Slov 55: 617-622 .
- Shobha D, Chari MA, Sadanandam P, Mukkanti K (2008) Dichloro dicyano quinone (DDQ) as coupling reagent for high yield synthesis of 3,4-dihydropyrimidin-2(1H)-ones. J Het Chem 45: 1225-1227.
- Ma Y, Qian C, Wang L, Yang M (2000) Lanthanide Triflate Catalyzed Biginelli Reaction. One-Pot Synthesis of Dihydropyrimidinones under Solvent-Free Conditions. J Org Chem 65: 3864-3868.
- Paraskar AS, Dewkar GK, Sudalai A (2003) Cu(OTf)2: a reusable catalyst for high-yield synthesis of 3,4-dihydropyrimidin-2(1H)-ones. Tetrahedron Lett 44, 3305-3308.
- Kundu SK, Majee A, Hazra A (2009) Environmentally benign aqueous zinc tetrafluoroborate-catalyzed one pot Biginelli condensation at room temperature. Indian J Chem 48B: 408-412.
- Patil S, Jadhav SD, Deshmukh MB (2011) Natural Acid Catalyzed Multi-component Reactions as a Green Approach. Arch Appl Sci Res 3: 203-208.
- Mishra BG, Kumar D, Rao VS (2006) H3PW12O40 catalyzed expeditious synthesis of 3,4-dihydropyrimidin-2(1H)-ones under solvent-free conditions. Catal Commun 7: 457-459.
- Kadre T, Jetti SR, Bhatewara A, Paliwal P, Jain S (2012) Green protocol for the synthesis of 3,4-Dihydropyrimidin-2(1H)- ones/thiones using TBAB as a catalyst and solvent free condition under microwave irradiation. Arch Appl Sci Res 4: 988-990.
- Boumoud T, Boudjemaa B, Mosset P, Debache A (2011) Gypsum-Catalyzed One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones Under Solvent-Free Conditions. E-Journal of Chemistry 8: 312-318.
- Dadhania AN, Patel VK, Raval DK (2012) A facile approach for the synthesis of 3,4-dihydropyrimidin-2-(1H)-ones using a microwave promoted Biginelli protocol in ionic liquid. J Chem Sci 124: 921-924.
- Dupont J, De Souza RF, Suarez PAZ (2002) Ionic liquid (molten salt) phase organometallic catalysis. Chem Rev 102: 3667-3692.
- Wilkes JS (2002) A short history of ionic liquids—from molten salts to neoteric solvents. Green Chemistry 4: 73-80.
- Wasserscheid P, Keim W (2002) Angew Chem Int Ed 39: 3772 -3789.
- Welton T (1999) Room-Temperature Ionic Liquids. Solvents for Synthesis and Catalysis. Chem Rev 99: 2971-2083.
- Sheldon RA (2005) Green solvents for sustainable organic synthesis: state of the art. Green Chem 7: 267-278.
- Carlin RT, Wilkes JS (1994) In Advances in non aqueous chemistry, ed Mamantov G, Popov A, Eds; VCH, New York.
- Olivier H (1999) Recent developments in the use of non-aqueous ionic liquids for two-phase catalysis J Mol Cata A: Chem 146: 285-288.
- Sheldon R (2001) Catalytic reactions in ionic liquids. Chem Commun 2399-2407.
- Earle MJ, Seddon KR (2000) Ionic liquids. Green solvents for the future. Pure Appl Chem 72: 1391-1398.
- Burrell AK, Del Sesto RE, Baker SN, McCleskey TM, Baker GA (2007) The large scale synthesis of pure imidazolium and pyrrolidinium ionic liquids. Green Chem 9: 449-454.
- Paulechka YU, Kabo GJ, Blokhin AV, Vydror OA, Magee JW, et al. (2003) Thermodynamic Properties of 1-Butyl-3-methylimidazolium Hexafluorophosphate in the Ideal Gas State. J Chem Eng Data 48: 457-462.
- Kabo GJ, Blokhin AV, Paulechka YU, Kabo AG, Magee JW (2004) Thermodynamic Properties of 1-Butyl-3-methylimidazolium Hexafluorophosphate in the Condensed State. J Chem Eng Data 49: 453-461.
- Weng J, Wang C, Li H, Wang Y (2006) Novel quaternary ammonium ionic liquids and their use as dual solvent-catalysts in the hydrolytic reaction. Green Chem 8: 96-99.
- Varma RS, Namboodri VV (2001) Solvent-free preparation of ionic liquids using a household microwave oven. Pure Appl Chem 73: 1309-1313.
- Das PJ, Das J (2012) Synthesis of aryl/alkyl (2,2′-bis-3-methylindolyl)methanes and aryl(3,3′-bis indolyl)methanes promoted by secondary amine based ionic liquids and microwave irradiation. Tetrahedron Lett 53: 4718- 4720.