Research Article
Efficacy and Tolerability of a Fixed-Dose Combination of Pregabalin and Nortriptyline in the Management of Diabetic Peripheral Neuropathy among Indian Population
Richa Giri1, Girish Gokuldas Bhatia2, Gopal Rao A3, Poorna Prasad S4, Manjula S5* and Krishna Kumar M5
1GSVM Medical College, Kanpur, Uttar Pradesh, India-208002.
2Medipoint Hospitals, Pune, Maharashtra, India-411007.
3Rajiv Gandhi Institute of Medical Sciences and RIMS Government General Hospital, Srikakulam, Andhra Pradesh, India-532001.
4Shettys Hospital, Bangalore, Karnataka, India-560068.
5 Micro Labs Limited, Bangalore, Karnataka, India-56001
2Medipoint Hospitals, Pune, Maharashtra, India-411007.
3Rajiv Gandhi Institute of Medical Sciences and RIMS Government General Hospital, Srikakulam, Andhra Pradesh, India-532001.
4Shettys Hospital, Bangalore, Karnataka, India-560068.
5 Micro Labs Limited, Bangalore, Karnataka, India-56001
*Corresponding author: Manjula S, Department of Medical Services, Micro Labs Limited, Race Course Road, Bangalore, Karnataka, India.Email Id: drmanjulas@gmail.com
Article Information: Submission: 28/11/2023; Accepted: 23/12/2023; Published: 25/12/2023
Copyright: © 2023 Giri R, et al. This is an open-access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Treatment of Diabetic Peripheral Neuropathy (DPN) is challenging, and effective therapies are not available for many patients; therefore, developing improved pharmacotherapy is essential. A combination therapy of pregabalin with nortriptyline could provide enhancing and complementary
effects through different mechanism of action, thereby reducing the symptoms. However, there are only limited studies available to support this combination.
Objective: To assess the safety and tolerability of a fixed dose combination of pregabalin and nortriptyline in the treatment of diabetic peripheral neuropathy among Indian population.
Methods: This was a multi-centric, open label, observational, phase IV (post-marketing surveillance) study conducted among type 2 diabetes mellitus patients with the evidence of painful diabetic neuropathy over last 1 month with the mean pain intensity of more than 50% by patient assessment by NRS
(numerical rating scale). The enrolled subjects received the fixed dose combination of Pregabalin 75 mg and Nortriptyline 10 mg once daily in the morning.
Efficacy was evaluated by measuring the change in the mean pain score (on the 11-point NRS) derived from entries on patient visits on visit 1 (baseline visit; day 1), to visit 3 (day 28). Efficacy was further evaluating using the Patient Global Impression of Change (PGIC) and Daily Sleep Interference Rating
Scale (DSIRS). Safety was assessed by evaluating the nature, duration, severity, and causal relationship of all adverse events to the study drug which were
documented at each clinic visit.
Results: Out of 224 subjects screened, 210 subjects were enrolled who had at least one visit and were administered at least one dose of the study
medication. The mean pain score measured on the Numerical Rating Scale (NRS), Daily Sleep Interference Rating Scale (DSIRS) and Patient Global Impression of Change (PGIC) scale during visit 1/ baseline visit was 7.94, 7.00 and 4.00 which was reduced on visit 2 and further decreased into 2.46, 3.48
and 1.39 on visit 3 respectively.A total of 12 subjects (6%) reported adverse events and were mild in severity and completely resolved within 24 hours of administering symptomatic/ appropriate medications.
Conclusion: The fixed dose combination of Pregabalin and Nortriptyline was efficacious and well-tolerated in the therapeutic management of patients with diabetic peripheral neuropathy.
Keywords: Diabetic peripheral neuropathy; Pregabalin; Nortriptyline; Diabetes mellitus; Neuropathic pain
Introduction
Diabetes mellitus (DM) is a chronic metabolic condition
characterised by hyperglycemia as a primary symptom whereas
chronic hyperglycemia leads to long-term damage and dysfunction
to multiple tissues and organs (such as the eyes, kidneys, nerves,
heart, and blood vessels) [1]. Diabetic peripheral neuropathy (DPN)
is described as “a symmetrical, length-dependent sensorimotor
polyneuropathy attributable to metabolic and micro vascular
alterations as a result of prolonged hyperglycemia and cardiovascular
comorbidities” by the Toronto Consensus Panel on Diabetic
Neuropathy. Sensory complaints typically manifest initially in the
toes and progress to the upper limbs in a “stocking and glove” pattern.
In the first phases of DPN, motor participation is not frequently
observed. Patients describe a variety of sensory symptoms, including
allodynia (painful sensation to innocuous stimuli) or hyperalgesia
(increased sensitivity to painful stimuli), loss of pain sensation or
“Novocain-like” insensitivity, tingling, “pins and needles” sensation,
burning, and “electric shocks” [2].
The development of more effective therapy is crucial since treating
DPN is difficult and many patients do not have access to appropriate
therapies. There is a dearth of information on pharmaceutical and
combination therapies that can stop or reverse DPN alterations or
partially relieve pain and thus, the therapeutic management of painful
DPN has a variety of unmet requirements [3].
Currently, Pregabalin, a GABAergic drug that is primarily used as
a first-line agent to treat painful DPN. As well as providing adequate
pain relief, it was well tolerated and the co-morbidities of DPN, such
as anxiety and sleep issues, can also be effectively treated with it [4-6].
Another molecule, nortriptyline, a tricyclic antidepressant and the
primary active metabolite of amitriptyline has modest antimuscarinic
effects and has been demonstrated to be useful in treating diabetic
peripheral neuropathy by inhibiting the reuptake of norepinephrine
and serotonin[7-9] . A combination therapy of pregabalin with
nortriptyline could provide enhancing and complementary effects
through different mechanism, thereby reducing the symptoms [10,11].
However, there are only limited studies available characterizing
the efficacy and safety of combination therapy of pregabalin and
nortriptyline, particularly in the management of diabetic peripheral
neuropathy on the Indian population. Hence, this study was designed
to assess the efficacyand tolerability of pregabalin with nortriptyline
as treatment for diabetic peripheral neuropathy.
Methods
A multi-centric, open label, observational, phase IV (postmarketing
surveillance) study conducted to assess the safety and
efficacy of combination of Pregabalin and Nortriptyline in diabetic
peripheral neuropathy. This study included patients who were
either on an inpatient or outpatient basis with the presence of type
2 diabetes mellitus and the evidence of painful diabetic neuropathy
over last 1 month with the mean pain intensity of more than 50%
by patient assessment by NRS (numerical rating scale). Subjects
having intolerance, hypersensitivity or any other contraindication
to either Pregabalin or Nortriptyline, evidence of other causes for
neuropathy and painful conditions, pregnant and lactating subjects
were excluded from the study. The enrolled subjects received the fixed
dose combination of Pregabalin 75 mg and Nortriptyline 10 mg once
daily in the morning.
Efficacy was evaluated by measuring the change in the mean pain
score (on the 11-point NRS) derived from entries on patient visits
on Visit 1 (Baseline Visit; Day 1), Visit 2 (Day 14) and Visit 3 (Day
28) where the numeric rating scale ranged from 0 being no pain to
10 being the worst possible pain. A rating of 1-3 was considered as
mild pain, 4-6 as moderate pain, and 7-10 as severe pain. Efficacy
was further evaluated using the Patient Global Impression of Change
(PGIC) and Daily Sleep Interference Rating Scale (DSIRS). Safety
was assessed by evaluating the nature, duration, severity, and causal
relationship of all adverse events to the study drug which were
documented at each clinic visit.
Statistical analysis was done by using SPSS (Statistical Package for
Social Sciences) of version 27 and R software. To test the hypotheses
of the study, descriptive statistics (mean and SD) and inferential
statistics, Friedman’s ANOVA (analysis of variance) test were used.
Value of P<0.001 were considered significant.
Results
A total of 224 subjects were screened from all participating study
centers on visit 1 (day 1), which included physical examination, vital
signs including height, weight and blood pressure, and assessment
of pain on the Numerical Rating Scale (NRS).Out of 224 subjects,
210 subjects were enrolled after meeting the inclusion and exclusion
criteria during the screening and 14 subjects were found not eligible
for enrolment and were deemed to be screen failures. A total of 7
subjects dropped out or were lost to follow up on visit 2 and/or visit
3. The total number of subjects who completed all visits and were
analysed for efficacy was 203. The total number of subjects who had
at least one visit and were administered at least one dose of the study
medication were analysed for safety was 210.
The mean score of pain on the Numerical Rating Scale (NRS)
during visit 1/ baseline visit was 7.94 ± 0.62. The mean pain score
subsequently decreased to 4.95 ± 0.78 on visit 2. The mean score
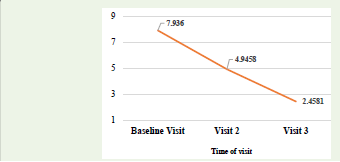
further decreased to 2.46 ± 0.80 on visit 3 as depicted in [Figure 1].
The mean score observed on the Daily Sleep Interference Rating Scale
(DSIRS) during visit 1/ baseline visit was 7.00 ± 1.05. The mean pain
score subsequently decreased to 3.96 ± 1.11 on visit 2. The mean score
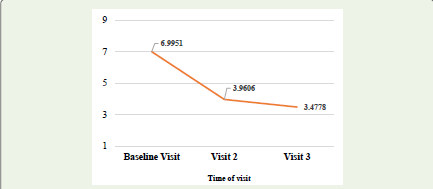
further decreased to 3.48 ± 1.22 on visit 3 as shown in [Figure 2].
The mean score noted on the Patient Global Impression of Change
(PGIC) scale during visit 1/ baseline visit was 4.00 ± 0.00. The mean
pain score subsequently decreased to 1.63 ± 0.61 on visit 2. The mean
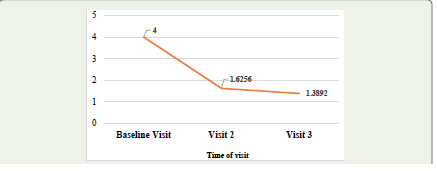
score further decreased to 1.39 ± 0.58 on visit 3 [Figure 3]. These
scores were found to be highly significant (p<0.001) on Friedman’s
ANOVA (analysis of variance) test with confidence interval of 95%.
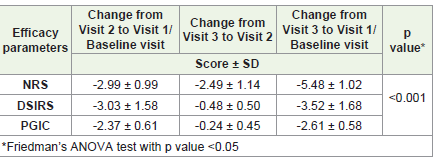
The change in the mean pain score on the Numerical Rating
Scale (NRS) from visit 2 to visit 1/ baseline visit was -2.99 ± 0.99. The
change in the mean pain score from visit 3 to visit 2 was -2.49 ± 1.14.
The change in the mean pain score on the Numerical Rating Scale
(NRS) from visit 3 to visit 1/baseline visit was -5.48 ± 1.02.The change
in the mean pain score on the Daily Sleep Interference Rating Scale
(DSIRS) from visit 2 to visit 1/ baseline visit was -3.03 ± 1.58. The
change in the mean pain score from visit 3 to visit 2 was -0.48 ± 0.50.
The change in the mean pain score on the Daily Sleep Interference
Rating Scale (DSIRS) from visit 3 to visit 1/ baseline visit was -3.52
± 1.68. The change in the mean pain score on the Patient Global
Impression of Change (PGIC) from visit 2 to visit 1/ baseline visit was
-2.37 ± 0.61. The change in the mean pain score from visit 3 to visit
2 was -0.24 ± 0.45. The change in the mean pain score on the Patient
Global Impression of Change (PGIC) from visit 3 to visit 1/ baseline
visit was -2.61 ± 0.58. There was no worsening of pain as assessed by
the Patient Global Impression of Change (PGIC) in any of the study
subjects on visit 2 or visit 3. The change in scores were found to be
significant (p<0.001) on Wilcoxon signed-rank test with confidence
interval of 95% [Table 1].
A total of 12 subjects (6%) reported adverse events, of these 5
subjects each were from RIMS, Srikakulam and Shetty’s Hospital,
Bangalore; 3 subjects were from Medipoint Hospitals, Pune, and 2
subjects from GSVM, Kanpur. All 12 adverse events were unrelated
to the investigational product and were mild in nature. All events
resolved completely after the administration of appropriate
medications, including over-the-counter medications. The most
common adverse events reported were body pain with 3 subjects
(1.5%) and headache and drowsiness with 3 subjects (1.5%). A total of
2 subjects each (0.5%) reported fever and pain, hyperacidity and fever
respectively. All adverse events were mild in severity and completely
resolved within 24 hours of administering symptomatic/ appropriate
medications for the same.
Discussion
More than one-third of diabetic patients have diabetic neuropathy,
a disorder that can lower their quality of life [12].Neuropathic pain,
which affects 8 to 26% of diabetic patients, is also regarded as one of
the most challenging pain syndromes to manage. Despite standard
treatment approaches, fewer than 50% of patients experience relief,
and the majority of patients still experience pain [13]. A few studies
comparing the medications used to treat neuropathic pain have been
carried out. This is the first study that directly compares pregabalin
and nortriptyline.
The present study showed that the mean pain scores of the
Numerical Rating Scale (NRS) decreased from 7.9360 ± 0.62207 on
visit 1 to 4.9458 ± 0.78477 on visit 2 and to 2.4581 ± 0.79725 on visit 3,
which was found to be statistically significant with p <0.05 with 95%
confidence interval on Friedrich’s ANOVA test. Comparison of the
scores from visit 3 (end of the study) to the baseline visit also showed
that the decrease in the pain score on the Numerical Rating Scales
were statistically significant on the Wilcoxon Singed-Rank Test, Z=
-12.572, p <0.05.In line with this, Khajuria et al. pointed out that there
was a statistically significant decrease in post drug mean NRS scores
with both pregabalin and nortriptyline group from baseline during
entire 8 week’s trial (p<0.0001) [14].
The findings of this study were similar to Shahid W et al. who
reported a decrease in mean VAS score from 6.99 ± 1.12 to 4.91 ± 0.82
with 12 weeks of therapy with pregabalin (p <0.0001) [4]. Mahmood
R et al. reported the mean pain score fell from 6.17 ± 0.14 to 3.50 ±
0.15 from day 0 to day 90 (p = 0.001) with pregabalin [15]. Bansal D
et al. reported good, moderate and mild pain relief were noted in 21
(48%), 6 (13%) and 7 (15%) patients on pregabalin [16].Lesser H et
al. highlighted that patients in the 300- and 600-mg/day pregabalin
groups showed improvements in mean pain score (primary efficacy
measure) compared to placebo (p = 0.0001). Improvements were
also seen in weekly pain score, sleep interference score, patient
global impression of change, clinical global impression of change,
SF-McGill Pain Questionnaire, and multiple domains of the SF-36
Health Survey. Improvements in pain and sleep were seen as early
as week 1 and were sustained throughout the 5 weeks [17]. Tölle T
et al. reported that pregabalin 600 mg/day was significantly superior
to placebo in improving pain-related sleep-interference scores (p =
0.003), PGIC (p = 0.021), and CGIC (p = 0.009) and all pregabalin
dosages were superior to placebo in improving EQ-5D utility scores
(all p >/= 0.0263 vs placebo) [18].Gomez-Perez FJ et al. in two
separate studies demonstrated the efficacy of nortriptyline along with
fluphenazine brought significant relief of both pain and paresthesia in
subjects with painful diabetic neuropathy [19,20].
The mean scores of the Daily Sleep Interference Rating Scale
(DSIRS) decreased from 6.9951 ± 1.05068 on visit 1 to 3.9606 ±
1.10732 on visit 2 and to 3.4778 ± 1.21999 on visit 3, which was found
to be statistically significant with p<0.05 with 95% confidence interval
on Friedrich’s ANOVA test. Comparison of the scores from visit 3
(end of the study) to the baseline visit also showed that the decrease
in the pain scores on the Numerical Rating Scales were statistically
significant on the Wilcoxon Singed-Rank Test, Z= -12.191, p<0.05.
The mean scores of the Patient Global Impression of Change
(PGIC) decreased from 4.00 ± 0.00 on visit 1 to 1.6256 ± 0.61155
on visit 2 and to 1.3892 ± 0.58129 on visit 3, which was found to be
statistically significant with p<0.05 with 95% confidence interval on
Friedrich’s ANOVA test. Comparison of the scores from the end of
the study to the baseline visit also showed that the decrease in the pain
scores on the Numerical Rating Scales were statistically significant on
the Wilcoxon Singed-Rank Test, Z= -12.807, p<0.05.
Safety and tolerability analysis revealed that a total of 12 subjects
(6%) reported adverse events and the most common adverse events
reported were body pain with 3 subjects (1.5%) and headache and
drowsiness with 3 subjects (1.5%). Two subject each (0.5%) reported
fever and pain, hyperacidity and fever respectively. Shahid W et al.
reported that lethargy/ somnolence (8.1%) and peripheral edema
(3.4%) were the commonly reported adverse events in the pregabalin
group and that pregabalin was efficacious in the relief of neuropathic
pain with good safety profile and was well tolerated [4]. Azmi S et al.
also reported that the most frequent adverse events with pregabalin
were somnolence and dizziness, which can lead to withdrawal in ~30%
of long-term use [21]. In a study by Bansal D, it was observed that
pregabalin caused adverse events in 18 (25%), of which drowsiness
was the most common in nine (20%) patients [16]. Another study by
Lesser H also reported that the most common adverse events were
dizziness and somnolence with pregabalin [17]. Gomez-Perez FJ et
al. reported that the adverse effects were not severe enough to lead to
cessation of nortriptyline [19].
To the best of the authors knowledge, this was the first study
assessed the fixed dose combination of Pregabalin with Nortriptyline
among diabetic peripheral neuropathy patients in various centres.
Although this study was conducted using robust methodology and
stringent inclusion and exclusion criteria, certain limitations need to
be considered while interpreting the results. The study had a small
sample size and included sample of patients with a similar race
and cultural background, and smaller duration of follow-up; thus,
the generalizability of the results should be made with caution. So,
it should be proved in a larger sample size with different race and
ethnicity to provide more specific results.
Conclusion
The fixed-dose combination of Pregabalin 75 mg and Nortriptyline
10 mg was efficacious and well-tolerated in the therapeutic
management of patients with diabetic peripheral neuropathy.
Funding: Research grant from Micro Labs Limited, Bangalore
Conflict of interest: Manjula S and Krishna Kumar M are employees of Micro Labs Limited, India.
Ethical approval: This study was approved by Institutional Ethics Committee Government Medical college, Government General Hospital, Srikakilam; Life Care Hospital Institutional Review Board Life Care hospital and Research centre, Sahakarnagar, Bangalore; Institutional Ethics Committee of GSVM and Shetty’s Hospital Ethics committee.
Conflict of interest: Manjula S and Krishna Kumar M are employees of Micro Labs Limited, India.
Ethical approval: This study was approved by Institutional Ethics Committee Government Medical college, Government General Hospital, Srikakilam; Life Care Hospital Institutional Review Board Life Care hospital and Research centre, Sahakarnagar, Bangalore; Institutional Ethics Committee of GSVM and Shetty’s Hospital Ethics committee.
References
Citation
Giri R, Bhatia GG, Gopal Rao A, Poorna Prasad S, Manjula S, et al. Efficacy and Tolerability of a Fixed-Dose Combination of Pregabalin and Nortriptyline in the Management of Diabetic Peripheral Neuropathy among Indian Population. Indian J Neurol. 2023;4(1): 127.