Case Report
Cyclosporine Induced Mesial Temporal Sclerosis - A Case Report
Sachin SJ1*, Shalaka S2, Goutham KJ2, Smruthy AM2, Anjali M1, Priyank T2, Hrishi V2, Amey CP1, Nishit1 and Shivakumar S3
1Deparment of Haematology and Stem cell transplant, HCG Cancer Hospital, India
2Deparment of Clinical Pharmacology, HCG Cancer Hospital, India
3Deparment of Radiology, HCG Cancer Hospital, India
*Corresponding author: Dr. Sachin SJ, Group Head, Haematology and Stem Cell Transplant, HCG Cancer Hospital,
Bangalore India, Tel: 9741351357; Email: drsachin.jadhav@hcgel.com
Article Information: Submission: 13/10/2021; Accepted: 18/11/2021; Published: 20/11/2021
Copyright: © 2021 Sachin SJ, et al. This is an open access article distributed under the Creative Commons Attribution
License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is
properly cited.
Abstract
Cyclosporine is an imperative part of GvHD prophylaxis post-transplant. It acts as an immunosuppressive agent by targeting T-cells and inhibiting the
activation cascade for lymphokine production. Among much toxicity, Cyclosporine induced neurotoxicity remains slightly daunting owing to inadequate
number of studies elucidating the mechanism and implication behind it. The toxicities present with varying symptoms such as visual disturbances, ataxia,
seizure episodes and signs of encephalopathy. Herein We report a case of AML, FLT3-ITD mutated with low allelic ratio, C-KIT mutated and AML 1-ETO
positive, Post Allogenic stem cell transplant on cyclosporine for immunosuppression which induced Mesial temporal sclerosis. Cyclosporine induced Mesial
Temporal Sclerosis is inadequately reported and therefore briefly understood. The availability of case reports with similar clinical picture encourages further
study to understand the risk factors and consequent management of the toxicity.
Keywords
Cyclosporine; Mesial Temporal Sclerosis
Introduction
Cyclosporine (CSA)is an imperative part of Graft vs Host Disease
(GvHD) prophylaxis post-transplant. It acts as an immunosuppressive
agent by targeting T-cells and inhibiting the activation cascade for
lymphokine production. Among many toxicities, CSA induced
neurotoxicity remains slightly daunting owing to inadequate number
of studies elucidating the mechanism and implication behind it. The
toxicities present with varying symptoms such as visual disturbances,
ataxia, seizure episodes and signs of encephalopathy [1-3].
Among the several manifestations of neurotoxicity, Mesial
Temporal Sclerosis (MTS) also known as hippocampal sclerosis
is very rare and scarcely reported. It is considered to be one of the
causes of drug resistant epilepsy among adults. It is characterized by structural and functional alterations and brain lesions in the
hippocampus. It is also considered to be a secondary consequence
of seizure activity as it is vulnerable to prolonged seizures, traumatic
brain injuries and other inflammation [4].
This case report describes a patient a known case of Acute
Myeloid Leukemia, treated as per guidelines, post haploidentical
stem cell transplant who developed prominent hippocampal and Para
hippocampal changes in MRI indicating Mesial Temporal Sclerosis,
after the use of CSA. Due consent was taken before writing the report.
Case Details
Herein reported is the case of a 39-year-old female. An evaluation
following complaints of generalized weakness, bleeding per
vaginal and passing clots, revealed 52% blasts in peripheral smear. Consequently, bone marrow aspiration biopsy with flow cytometry
and Fluorescence in situ hybridization (FISH) reports led to the
diagnosis of Acute Myeloid Leukaemia (AML), FLT3-ITD mutated
with low allelic ratio, C-KIT mutated and AML 1-ETO positive. Her
induction therapy included 7+3 regimen with Daunorubicin and
Cytarabine, during which she developed an episode of hyponatremia
induced seizures(Na+: 116 mg/dL). She was prescribed Levetiracetam
thereafter for appropriate epilepsy control.
Her bone marrow post one cycle of consolidation with
intermediate dose Cytarabine, showed remission and FLT3-ITD was
negative. She was counselled and initiated on conditioning therapy for
Haploidentical Stem Cell Transplant in CR1. Conditioning regimen
comprised of Fludarabine, Treosulfan, Anti-thymocyte globulin
(ATG) and Total Body Irradiation (TBI). Post stem cell transplant
she had one fever spike and XCyton revealed Aspergillus in the blood.
She was treated with Voriconazole for the same. She presented with
Grade 2 mucositis and radiation induced dermatitis which resolved
eventually. Patient was on CSA for GvHD prophylaxis.On D+35
she was admitted for Cytomegalovirus (CMV) colitis and CMV
pneumonitis (Viral load: 1,69,100 cp/mL). She was treated with
Ganciclovir (28 days) and IV-Ig for the same. Subsequent CMVPCRs
were negative.
OnD +104, she was admitted to the hospital with complaints of
febrile episodes, with a recorded temperature of 100.4 °F. Her vitals
were stable and an unhealthy mucosa in the oral cavity was noted,
indicating resolving acute oral GvHD. Her initial blood biochemistry
revealed low potassium and magnesium levels (K+: 2.63 mg/dL;
Mg2+: 0.8 mg/dL) and CSA trough levels were 27.85 ng/mL. Required
microbiology profiles were sent for evaluation and she was treated
with appropriate antibiotics and electrolyte corrections.
On D+106, the patient complained of generalized weakness which
worsened on standing. The following day, the patient complained of
blurring of vision and loss in the center of field of vision. There was no
history of falls, blackouts or seizures. On examination she presented
right sided ataxia and scotoma of the left eye. There was non-specific
imbalance in gait. Her motor score was 4/5 for both upper limbs
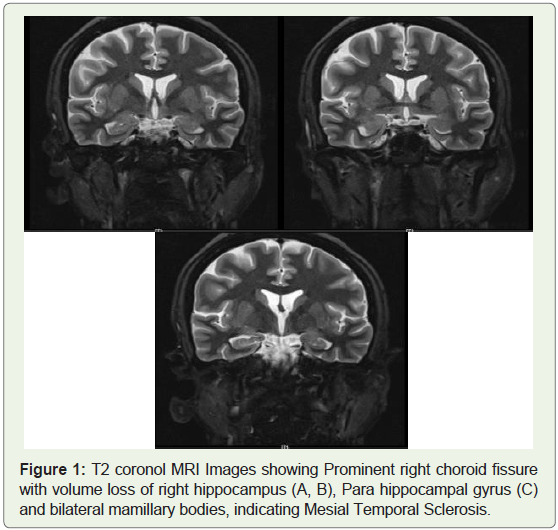
and lower limbs. An MRI done, revealed Prominent right choroid
fissure with volume loss of right hippocampus, Para hippocampal
gyrus and bilateral mamillary bodies. These findings raised a concern
for and indicated Mesial Temporal Sclerosis (Figure 1). Repeated
Cyclosporin trough levels were 152.29 ng/mL. Given there was no
history of trauma or recent episodes of seizures, drug toxicity were
suspected. CSA in known to cause neurological adverse reactions
such as PRES, visual disturbances and cerebellar ataxia. Using the
WHO-UMC Adverse Drug Reaction (ADR) causality assessment
scale, the reaction was graded as Certain [5].
Figure 1: T2 coronol MRI Images showing Prominent right choroid fissure
with volume loss of right hippocampus (A, B), Para hippocampal gyrus (C)
and bilateral mamillary bodies, indicating Mesial Temporal Sclerosis.
Based on the literature available and the patient’s condition,
a decision was taken to discontinue CSA and replace it with
Mycophenolate Mofetil. Following the change in medication, the
patient’s vision improved and her gait stabilized. There were no
episodes of seizures or falls in the coming days. She recovered from
the adverse effect with no residual symptoms.
Discussion
Cyclosporin, a lipophilic cyclic oligopeptide, is a potent immunosuppressant which is widely used in transplant setting.
CSA exerts its immunosuppressant action via reversibly inhibiting
the activation of primary T-helper cells and the consequent release
of lymphokines such as interleukine-2 (IL-2). CSA binds to various
blood components such as lipoproteins, erythrocytes and leukocytes,
leaving less than 5% free drug concentration. Thus, lipoprotein
structures serve as drug reservoir. It is extensively metabolized
in the liver by the CYP450 family, its metabolites having inferior
immunosuppressive action. However, the metabolites tend to be
more toxic [1, 6].
CSA requires stringent therapeutic drug monitoring owing to its
narrow therapeutic index and variable absorption. While there is a
risk of subclinical dose, there is also a risk of adverse events if the
plasma concentrations are above the desired range [6]. Hypertension
and renal damage have been established toxicities among others.
Despite a fair incidence of neurotoxicity induced by CSA, it remains
an inadequately studied adverse effect of the drug [7].
CSA induced neurotoxicity presents with a spectrum of
symptoms ranging from mild visual disturbance and ataxia to severe
encephalopathy, seizures and related changes in Magnetic Resonance
Image. Many of the lower grade symptoms and damages appear to be
reversible. However, the risk of irreversible changes induced by the
symptoms of neurotoxicity such as seizures and encephalopathy also
remain [7, 8].
The mechanism of CSA induced neurotoxicity is poorly
understood however a few experimental studies point towards
interference in mitochondrial function and induction of neuronal
apoptosis. Studies also suggest the neurotoxicity to be a result of
direct neuronal synaptic hyperexcitability, or indirect damage
to brain vasculopathy by the release of vasoconstrictors such as
endothelin or thromboxane which cause vaso spasm. Additionally,
studies suggest, lower plasma levels of cholesterol may contribute to higher toxicity rates, given the drug is highly lipoprotein bound. In
situations where, blood levels of CSA are higher than the therapeutic
range, neurotoxicity symptoms can be expected, however, studies
suggest that the milder symptoms can occur at concentrations well
within the therapeutic range as well [1, 8-10].
A case series by Anna Noè et al, presented the clinical picture of
six patients who developed varied symptoms of neurotoxicity due to
CSA such as seizures, signs of endocranial hypertension and MRI
images implying reversible leukoencephalopathy. The drug levels were
within the desired therapeutic range, indicating higher blood levels
are not necessarily a risk factor for neurotoxicity. Interestingly, the
occurrence of evident neurotoxicity in these patients was preceded by
symptoms of arterial hypertension, headache, visual disturbances and
vomiting. These patients were then prescribed Tacrolimus following
which the patients remained symptom-free at median survival of
882 days [11]. Our patient experienced a similar pattern, where she
presented with visual disturbance and gait imbalance, before the
MRI investigation. Three days following the withdrawal of CSA, the
patient symptoms improved.
While Mesial Temporal sclerosis, a highly epileptogenic lesion
marked by changes in hippocampus, and amygdala, can be considered
as a manifestation of neurotoxicity, reports of CSA induced MTS are
scarce. A case serious published by M Faraci et al, elucidated similar
findings as our patient in pediatric patients post BMT. Three out of
four of the patients developed seizures induced by toxic levels of CSA.
The symptoms recurred even on rechallenging the drug making it
certain the neurotoxicity was caused by CSA. The authors theorized
that factors such as neurotoxic therapy in first line and conditioning
regimens along with history of febrile seizures and other CNS injuries
may have induced irreversible vascular damage causing hippocampal
shrinkage and subsequent MTS. [10]. Some recent studies done, have
shed light on the possible risk factors for CSA induced neurotoxicity.
A retrospective observational study done by Alberto Lue et al, among
patients who underwent liver transplant elucidated the risk factors
of CSA induced neurotoxicity. Although, the transplant setting is
different from our patient, some non-specific risk factors such as
previous history of encephalopathy, and pre-transplant Sodium
levels can be recognized. A retrospective case series study by Yong
Wang et al, among children who underwent haploidentical stem
cell transplant, reported hypertension to be higher among the group
with CSA induced neurotoxicity than the non-neurotoxicity group.
Transient headache was another common prodrome among these
patients [12, 13].
CSA induced mesial temporal sclerosis although rare, needs to
be studied elaborately to understand its occurrence and implications.
Contrary to previous reports we observed that MTS is not always preceded by elevated plasma CSA levels or seizure episodes. Therefore,
it is advisable to monitor symptoms of neurotoxicity among patients
receiving CSA.
Conclusion
CSA induced Mesial Temporal Sclerosis is inadequately reported
and therefore briefly understood. The availability of case reports with
similar clinical picture encourages further study to understand the
risk factors and consequent management of the toxicity. Studies
have indirectly pointed towards certain predictors such as changes
in blood pressure and occurrence of headaches; however, dedicated
studies will help us understand the event in more detail.
References
Citation
Sachin SJ, Shalaka S, Goutham KJ, Smruthy AM, Anjali M, et al. Cyclosporine Induced Mesial Temporal Sclerosis - A Case Report. Indian J Neurol. 2021;2(1): 104.