Review Article
Cirrhosis, Hepatic Encephalopathy, and Liver Transplantation - Special Focus on Nutrition
Lawate PS1*and Sharma M2
1Consultant Gastroenterologist & Hepatologist and Gastrointestinal Endoscopist, Clinic & Endoscopy Rooms, 1204/15, Vitthal Niwas,
Ghole road, Shivajinagar, Pune, Maharashtra, India
2Hepatology and Regenerative Medicine, Consultant GI, Hepatology and Liver Transplant, AIG Hospitals, Hyderabad, Telangana,
India
*Corresponding author: Lawate PS, Consultant Gastroenterologist & Hepatologist and Gastrointestinal Endoscopist, Clinic &
Endoscopy Rooms, 1204/15, Vitthal Niwas, Ghole road, Shivajinagar, Pune, Maharashtra, India; Phone:+91 9822028559; Email:
parimallawate@gmail.com
Article Information: Submission: 03/11/2022; Accepted: 29/11/2022; Published: 01/12/2022
Copyright: © 2022 Lawate PS, et al. This is an open access article distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Malnutrition and sarcopenia are common in patients with chronic liver disease (CLD) and are associated with increased risk of decompensation
progressing to hepatic encephalopathy (HE), and poorer outcomes after liver transplantation (LT). Prevalence of malnutrition in cirrhosis ranges from 65-90%
and is seen in 20% and >80% of patients with compensated and decompensated cirrhosis, respectively. Malnutrition and CLD increase healthcare burden by
increasing incidence and severity of complications with associated poor survival. Specific issues that merit consideration in the assessment of nutritional status
and management of malnutrition in the Indian context are reviewed here. Assessing and treating malnutrition are essential in the management of patients with
CLD. Several screening tools to identify patients at risk and/or to confirm the presence and severity of malnutrition have been validated in cirrhotic patients.
Complete nutritional care design with timely nutritional modification by a dietician is advisable. Most nutritional intervention studies and guidelines in cirrhosis/
CLD recommend 30-35 kcal/kg dry body weight/day and protein intake of 1.2-1.5 g of proteins/kg/day. Adoption of a breakfast containing proteins and a late
evening snack to shorten the period of nocturnal fasting are recommended in cirrhotic patients to achieve improvement in metabolic profile. Supplementation
with branched chain amino acids (BCAAs) is known to improve abnormal glucose tolerance and serum albumin concentration in patients with CLD. Late
evening snacks with BCAAs can be useful in improving protein metabolism and lipolysis in cirrhotic patients. Several clinical trials have suggested that BCAA
supplementation improves nutritional status, reduces complications/arrests disease progression, and improves prognosis of cirrhotic patients regardless of
patient age or disease stage.
Keywords
Malnutrition; Cirrhosis; Chronic liver disease; Hepatic encephalopathy, Liver transplantation; Nutrition; Branched chain amino acids
Nutrition in Cirrhosis and End Stage Liver Disease
Prevalence of malnutrition cirrhosis
Malnutrition is highly prevalent yet often neglected in patients
with chronic liver disease (CLD) and is found in 65-90% of patients
with advanced liver disease and in almost all candidates undergoing
liver transplantation (LT). Malnutrition has been reported in 20% of
patients with compensated cirrhosis and up to 80% of patients with
decompensated liver disease [1].
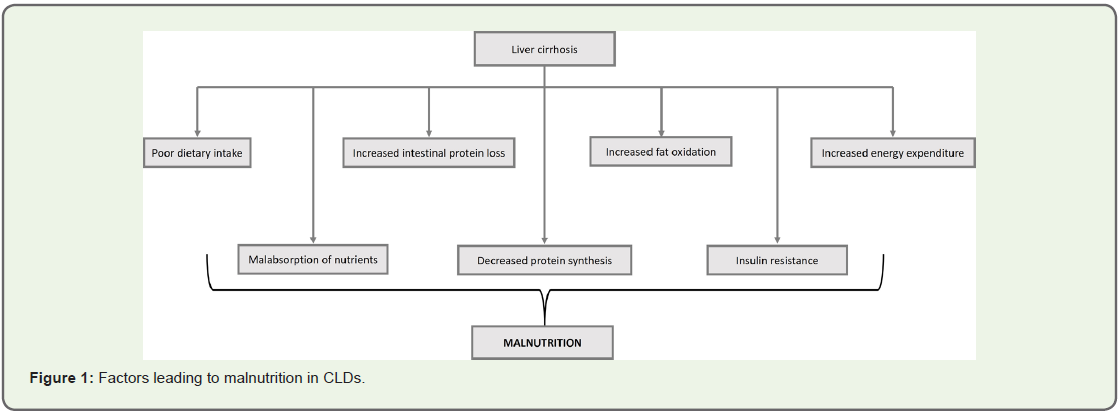
Mechanism of malnutrition in CLD:
Cirrhosis is a state of accelerated starvation with decrease in
protein synthesis and increase in gluconeogenesis from amino acids,
demanding proteolysis, with resultant sarcopenia. This sarcopenia is
further aggravated by reduced dietary intake due to a variety of factors
including dysgeusia, anorexia of chronic disease, inappropriate
dietary protein restriction, salt restricted food that is not tasty, portal
hypertension, impaired gut motility, decreased nutrient absorption
and protein losing enteropathy, hospitalization with periods of
fasting for diagnostic and therapeutic procedures, encephalopathy
and gastrointestinal bleeding (Figure 1) [1,2].Clinical outcomes related to malnutrition in CLD:
Malnutrition and sarcopenia are well recognized as predictors of morbidity and mortality in patients with advanced liver disease.
Compared with the global population, a high prevalence of
malnutrition and sarcopenia in cirrhosis has been reported in India
varying between 47% and 84% [3]. Prevalence of sarcopenia in
Indians is higher (61%) because of factors like lower muscle mass,
high carbohydrate and low protein diet and lack of physical activity
[4].Sarcopenia is associated with a high rate of infection and long
duration of hospital stay, recurrent HE, poor quality of life, increased
treatment-related mortality, increased healthcare cost and tumor
recurrence with poor survival in patients with hepatocellular
carcinoma (HCC) [5-10]. Sarcopenia also contributes to the model
for end-stage liver disease (MELD) score by 10 add-on points [11].
Optimizing nutritional status in this patient population is therefore
of critical importance [12].
Nutritional assessment in patients with CLD:
Literature documents wide variety of diagnostic tools and
different cut off values/ criteria giving wide range of malnutrition
prevalence in different studies. Commonly used are subjective
global assessment (SGA), mid arm muscle circumference (MAMC),
triceps skinfold thickness (TSF), and handgrip strength (HGS), and
less commonly, imaging devices (bioelectrical impedance analysis,
computed tomography [CT], and magnetic resonance imaging
[MRI]) [13].Although nutritional assessments for cirrhotic patients are
challenges faced in clinical current practice, regular nutritional
assessment should be done as soon as the patient is diagnosed for
cirrhosis and thereafter, it should be done yearly if it is a compensated
cirrhosis and monthly - every 3 months - every 6 months in case of
decompensated cirrhosis.
Frailty test are quicker and easier methods to perform in routine
clinical practice in Indian clinics. Often body mass index (BMI) and
mid-arm circumference/skin fold thickness are used as tools though
they are less reliable especially in presence of ascites.
In clinical setups, if possible, best way to assess for sarcopenia is
non-contrast CT at L3 and Dexa scan.
There is a need for development of an easy and efficient tool
for nutritional assessment of patients with CLD in routine clinical
practice.
Nutritional management principles in CLD:
As per European Society for Clinical Nutrition and Metabolism
(ESPEN) guidelines, hemodynamically stable cirrhosis patients with
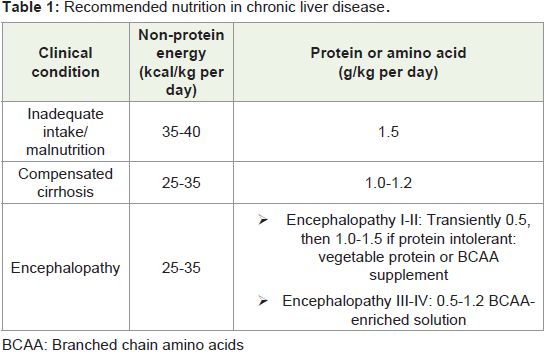
sedentary lifestyle require 30-35 kcal/kg dry body weight/day [14].
The protein intake should be 1.2 g of proteins/kg/day in the absence
of malnutrition or 1.5 g/kg/day in the presence of malnutrition [2].
Fat being a dense energy food, it provides more calories than proteins.
As cirrhotic patients need energy, fats do help on to achieve patients’
nutritional goals.Meal pattern during the day (multiple, small, frequent 4-6
hourly meals) should prevent prolonged periods of fasting: early
morning breakfast [high protein content] and late evening snacks
[energy-dense e.g. comprising of complex polysaccharide (50 g)]
have both been found to be beneficial and can avoid an early onset,
gluconeogenic starvation like state [3]. Nocturnal branched chain
amino acids (BCAAs) administration as a late evening snack (LES)
has reported to improve serum albumin level and glucose tolerance
in cirrhosis patients (Table 1) [15,16]. Fluid restriction to less
than 1.2-1.5 liters is advised in cirrhotic patients having dilutional hyponatremia [3]. Physical exercise being an anabolic stimulus is also
very helpful in regaining the loss of muscle mass [1].
Correction of sarcopenia by nutritional therapy reflects in
improvement of deranged parameters and clinical outcomes. For
patients with sarcopenic obesity, hypocaloric and high protein diet
i.e. approximately 800 kcal/day, but 1.2 to 1.5 g proteins/day should
be given to the patients. Goal should be to reduce the weight by 5%
to 10% [3].
Nutritional intervention with BCAA in CLD:
Deficiency of the essential BCAAs valine, leucine, and isoleucine
is involved in the development of liver diseases. Administration of
BCAAs in patients with CLD stimulates hepatic protein synthesis
and subsequently results in an improvement in their nutritional
status, thus resulting in a better quality of life. A decrease in the
BCAA: aromatic amino acid [AAA] ratio due to alterations in amino
acid metabolism in patients with liver disease may be responsible
for some of the complications associated with liver disease, such as
HE. Normalization of this ratio via BCAA supplementation has the
potential to prevent these complications. BCAAs may be the preferred
nutrients for cirrhotic patients due to their higher energy efficiency
than glucose or fatty acids. A late evening snack containing BCAAs
improves protein metabolism and lipolysis in patients with cirrhosis
and thereby can improve respiratory quotient (RQ), nutritional state,
and glucose intolerance [17].Mechanism of action of BCAAs in CLD:
BCAAs are involved in various biological actions, and their
supplementation is a promising therapeutic option for patients with
CLD [15].In liver cirrhosis, BCAAs induce mitochondrial biogenesis and
inhibit reactive oxygen species (ROS) production; stimulate albumin
and glycogen synthesis; inhibit hepatocyte apoptosis, promote
liver regeneration, and stimulate Hepatocyte growth factor (HGF)
production; and improve insulin resistance. In patients with HCC,
BCAAs inhibit proliferation of HCC cells and hepatocarcinogenesis.
Several clinical trials have suggested that BCAA supplementation
improves the prognosis of cirrhotic patients, regardless of patient age
or disease stage. In patients with advanced cirrhosis, they prevented
progressive hepatic failure and improved surrogate markers with
improved perceived health status. In patients with advanced
cirrhosis, they are effective in improving nutritional status and may
also improve abnormal glucose tolerance in addition to improving
serum albumin concentration [15].
Across India, 111 experts in gastroenterology and hepatology
participated in 10 virtual, focused-group meetings. Evidence-based
discussions were conducted about nutritional assessment and
management principles in patients suffering from CLDs. Expert
opinions were formulated and finalized after collating the discussion
from all meetings and developed into a consensus document.
Expert Opinion on Nutritional Recommendations for Liver Cirrhosis:
Eggs are a good source of proteins, with egg yolk providing 2.5 grams of proteins. Protein restriction is to be avoided; however, in
patients with renal dysfunction up to 1 g/kg is advised. Lemon and
black salt may be used to improve palatability for patients restricted
to low salt diet. Fat intake can be increased by incorporating ghee in
the meals.Dietary source of BCAAs include chicken, salmon, turkey, tuna,
milk and milk products. To meet their protein requirements, 12-15
grams of BCAA per day can be given to patients.
It is important to restrict sodium intake and not fluids in
patients with ascites. Screening of micronutrients and appropriate
supplementation with vitamin A, D, magnesium, and zinc is equally
important.
Physical exercise is also very helpful in regaining the lost muscle
mass. Weight bearing/training exercises and simple resistance
exercises can help in regaining the muscle mass e.g., running
stationary cycle at home and upper limb weight training exercises
with 1-1.5 kg dumbbells.
Nutrition in Hepatic Encephalopathy:
HE is a frequent and serious complication of cirrhosis that is
associated with poor prognosis and quality of life [15]. HE is associated
with prolonged hospitalizations, repeated re-admissions, and high
rates of mortality, irrespective of the severity of the underlying liver
disease [18]. Overt and clinically evident forms of HE occur in 30%–
45% of patients with cirrhosis and 10%-50% of patients who undergo
transjugular intrahepatic portosystemic shunt (TIPS) placement
[18,19]. Estimates of the prevalence of minimal HE (mHE) in patients
with cirrhosis range from 20% to 80%, often asymptomatic. Patients
with a previous episode of overt HE have a 42% risk of recurrence at
1 year, and those with recurrent overt HE have a 46% risk of another
episode within 6 months, despite receiving standard care [18]. Large
bleed, spontaneous peritonitis, and infection are common reasons for
recurrence of HE episodes.Pathophysiology of HE:
Pathogenesis of HE is multifactorial. Elevated blood ammonia
is one of the main pathogenic factors for the development of HE.
However, monitoring of serum (arterial) ammonia has no linear
correlation to HE status and prognosis and is possible only in
hospitalized patients as a simple and quick method, but it is not yet
available for outpatient clinics. Other factors include systemic and
neuroinflammation, oxidative stress and cellular senescence and
blood-brain barrier (BBB) permeability/altered integrity with altered
neurotransmission causing deleterious effects [18].Biochemical derangements in HE:
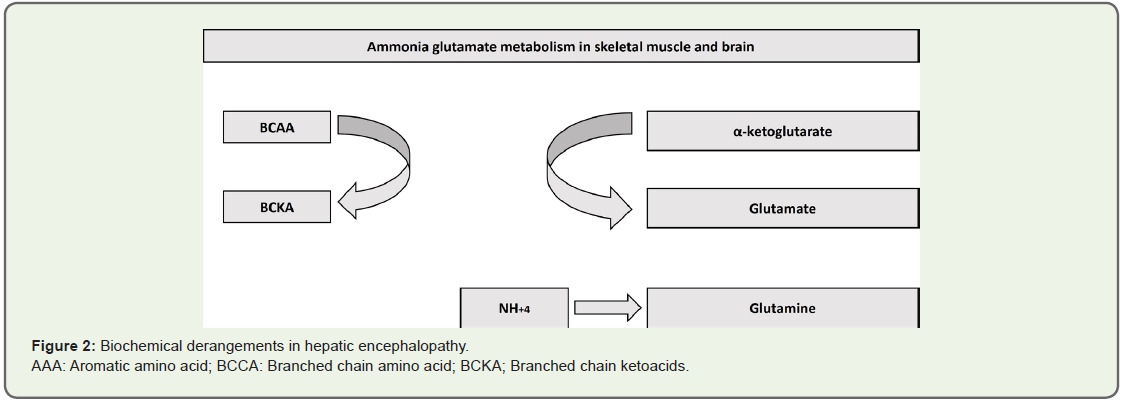
Physiologically normal Fischer’s ratio (i.e. BCAA/AAA ratio) is
3.0-3.5:1. This ratio is decreased in patients suffering from cirrhosis.
BCAAs decrease as they are rapidly consumed to form glutamate from
α ketoglutarate [α-KG] as a pivotal step in ammonia detoxification to
glutamine (GLN) in muscles and brain (Figure 2) [20].
Figure 2: Biochemical derangements in hepatic encephalopathy.
AAA: Aromatic amino acid; BCCA: Branched chain amino acid; BCKA; Branched chain ketoacids.
Accelerated consumption of α-KG may disturb the function of
tricarboxylic acid (TCA). AAA increase is due to decreased ability
of the diseased liver to metabolize these amino acids. Therefore, decreased Fischer’s ratio leads to augmented uptake of AAAs by
the brain causing an imbalance in the synthesis of nor-epinephrine
and serotonin and increased synthesis of false neurotransmitters e.g.
octopamine, phenyl ethanolamine, and tyramine [18].
Impact of HE:
HE manifests as a wide spectrum of neurological or psychiatric
abnormalities, from subclinical alterations (mild cognitive
impairment detectable only by neurophysiological/neuropsychological
assessment) to marked disorientation, confusion and coma [18,21].
Neuroinflammation and neuronal cell death are features of HE and
episodes of overt HE can lead to irreversibility.Treatment of HE:
Therapeutic approach aims to manage bouts of overt HE efficiently
with respect to duration and its consequences and further preventing
recurrence and hospital readmissions. Most of the therapies for HE
targets ammonia.Lactulose remains the first line of therapy in the management
of HE patients. Rifaximin can be given if patients are refractory to
lactulose monotherapy or to prevent episodes of HE recurrence [17].
Around 0.25 g of BCAA per kg body weight can be given orally in
patients who are protein intolerant or in patients having recurrent
HE episodes to prevent recurrences [14].
Nutritional treatment principles and evidence-based therapies for HE:
Energy requirements in patients with cirrhosis per se and cirrhosis
with HE are considered to be similar, with focus on prevention and/
or delaying progression of the disease [1]. Type and amount of food
for each HE patient is advised to be adjusted to maintain a total
energy intake of at least 1.3 times the basal energy expenditure (BEE)
(e.g., a protein intake of 1.2 to 1.5 g/kg including BCAAs increased
according to degree of hepatic decompensation), in accordance with
ESPEN guidelines [14].In patients with HE, the protein intake should not be restricted
but preferably be enriched by high biological value proteins
(vegetable and dairy proteins preferred over animal proteins). BCAA can be used in case of protein intolerance as well [14]. Additionally,
micronutrient and vitamin [both fat and water soluble] deficiency, if
identified should be corrected.
Salt restriction should be avoided to avoid poor palatability /poor
intake because strict salt restriction can be associated with higher
incidence of hyponatremia and diuretic-induced renal impairment.
Therefore, intake of 5-6 g/day of salt is advisable.
Oral dietary intake is preferred in patients with early HE and
in those who can tolerate recommended intake. In patients with
advanced HE and in those who are not able to take orally, nasogastric
tube feeding or parenteral nutrition should be considered. Critically
ill patients with cirrhosis have higher nutritional requirements due to
a net catabolic state and should be replenished as a priority [14,18].
Role and rationale of BCAAs in HE patients:
BCAAs may not prevent HE but may prevent the progression
of hepatic failure and may improve minimal HE and muscle mass
[15]. BCAA therapy was found to improve the manifestation of overt
HE in a systematic review, while it was found to have no effect on
mortality, quality of life, or nutritional parameters [22]. BCAAs have
been documented to promote muscle protein synthesis and improve
muscle mass loss; however, additional multicenter clinical trials to
determine the effect of BCAAs versus active interventions are needed
[17,22].Expert Opinion on Nutritional Recommendations for Bcaas In He:
Protein restriction should only be done in an inpatient department
(IPD) setting for patients suffering from recurrent and severe HE.Dairy protein can be better tolerated than protein from mixed
sources and that vegetable protein is better tolerated than meat
protein; as they have low levels of ammonia-genic amino acids.
BCAAs can be used as supplements for achieving the total protein
requirements of HE patients. For patients who are protein intolerant
or are having recurrent HE episodes, BCAA supplementation can
help meet protein requirements or reduce the number of HE episodes.
International Society of Hepatic Encephalopathy and Nitrogen Metabolism (ISHEN) guidelines recommend BCAA supplements
occasionally for patients intolerant of dietary proteins.
Small meals or liquid nutritional supplements evenly distributed
throughout the day and as late-night snacks should be offered.
Parenteral BCAAs can be given in cases of HE.
Around 0.25 g BCAA per kg body weight can be given orally in
patients who are protein intolerant or in patients having recurrent
HE episodes. Experts opined that BCAAs function more like a drug
than a supplement in such patients.
Nutrition in Patients Undergoing Liver Transplantation:
Alcoholic liver disease, followed by non-alcoholic fatty liver
disease (NAFLD), is the most common cause of liver transplantation
in the Indian population. Pre-transplant malnutrition and sarcopenia
are associated with increased risk of decompensation, infections
and increased waitlist mortality, while post-transplant these
complications predict poor outcomes with longer time to extubation
after transplantation, increased post-operative infections, prolonged
intensive care unit (ICU)stay and hospitalization, and decreased
survival [23].Principles of nutritional management in patients with liver disease and transplant candidates:
Assessment of nutritional status of all patients being prepared for
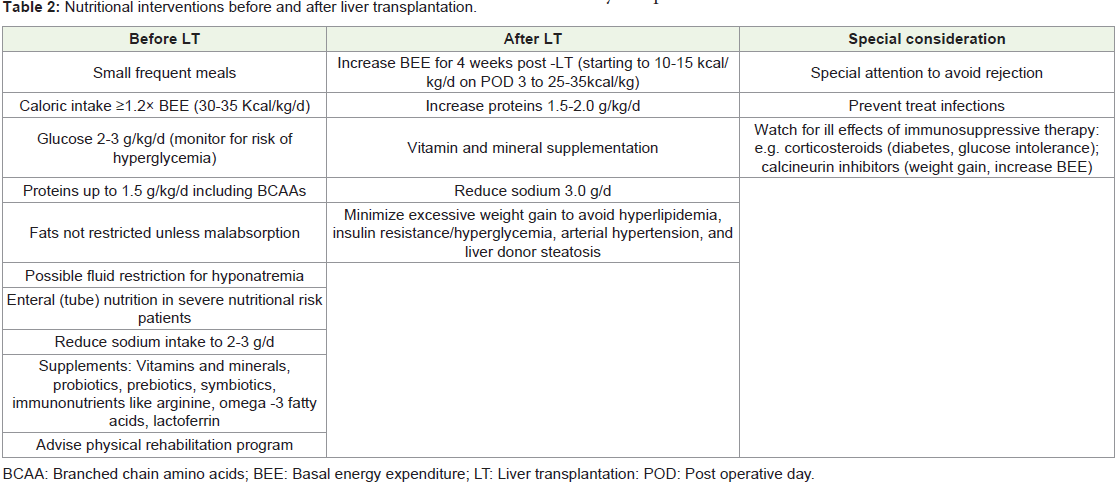
LT should undergo thorough nutritional evaluation.Before Liver Transplantation:
Pre-transplant nutritional improvement is very important
and should aim to prevent further nutrient deficiency and muscle
depletion and to correct vitamin and mineral deficiencies to improve
the post-operative outcomes specifically to minimize risk of infection,
reduced hospital ICU stay, and debility [23].ESPN guidance recommends enteral nutrition (EN) with a gastric
or jejunal small-bore feeding tube for patients incapable of ingestion and parenteral nutrition (PN) for patients with fulminant hepatic
failure and those in coma, for patients who are moderately or severely
malnourished and who cannot achieve adequate energy intake, either
orally or via EN due to gastrointestinal dysfunction [14].
Carbohydrate intake should exclusively be provided by
glucose infusion [2-3 g/kg body weight per day] without causing
hyperglycemia. Dietary fat should not be restricted unless true fat
malabsorption has been diagnosed. Whole-protein formulas or
BCAA-enriched formulas are advisable in patients who develop HE
during re-feeding. Protein intake should be at least 1 g/kg/day initially
with progressive increment to up to 1.8-2.0 g/kg/day as tolerated.
Recovery of all weight loss happens in the 1st post-transplant year, fat
mass progressively increases, but muscle mass recovery is subtle and
non-significant by the end of the first year [23].
Patients with ESLD are susceptible to vitamin and mineral
deficiencies, which need to corrected appropriately. Probiotic
supplements can alter gut microbiota, prevent bacterial translocation,
decrease endotoxin levels, and restore neutrophil phagocytic capacity
[23].
Nutritional Support after LT:
After LT surgery, energy and protein requirements are increased
for weeks. In the immediate phase after the surgery, protein catabolism
is markedly increased, and patients should receive about 1.5-2.0 g/
kg/day of proteins. Resuming EN within 12 h of LT has been shown
to reduce postoperative infections and to produce better nitrogen
retention, bile duct complications, length of ICU stay, and time on
ventilator. Patients should be advanced from nutritional support
to an oral diet using smaller and more frequent feedings as soon as
tolerated after LT [23] (Table 2).With dietitian consultation, patients are instructed to be on a
calorie-sufficient diet comprised of low-fat content, with adequate
amounts of lean protein foods to promote muscle gain and healthy
body composition.
BCAA treatment in liver transplantation:
There is a lack of substantial data for efficacy of BCAAs in pre
and post transplantation patients. Absence of preoperative BCAA
treatment was found to be an independent risk factor for postoperative
severe infection and in-hospital death in subjects undergoing livingdonor
liver transplantation (LDLT).On the contrary, pre-treatment
with BCAA before LDLT may reduce the incidence of post transplant
bacteremia and sepsis. Data reports that early interventional oral
BCAAs might prolong the liver transplant waiting period by
preserving hepatic reserve in patients with cirrhosis [15]. A pre-
LT BCAA-enriched formula has been reported to lower ammonia,
thereby improving Fischer’s ratio and albumin, prealbumin, total
lymphocyte count, BCAAs/tyrosine ratio [BTR], glucose intolerance,
liver regeneration, immune system function, maturation of dendritic
cells and the ability of peripheral blood mononuclear cells and
preventing postoperative sepsis. Oral BCAAs can be of importance
to improve post-LT mortality by preserving the hepatic reserve of
scheduled LT recipients [15,20].Expert Opinion on Nutritional recommendations For Bcaas in Patients Undergoing Liver Transplantation:
Frequent feeding with concentrated meals is advised. Focus on
fortifying habitual nutritional intake e.g., changing the composition
of chapati flour, adding seeds, nuts and millets to meals, kneading
pulses and millets into the chapati flour, whey water (paneer water
and curd water) and lentils, pulses, beans, soya, quinoa, or amaranth,
are recommended.Supplemental sip feeding, e.g., 2 × 200 mL drinks of a standard
polymeric formula containing 300 kcal each,could be given in
malnourished cirrhotic patients who are unable to reach their
nutritional requirements.
Hypomagnesia and hypoglycemia are common metabolic
disturbances seen in post-transplant patients, and these should be
timely addressed.
Conclusion
Nutritional assessment and management of malnutrition are
crucial in patients with CLD, and these can be improved by close
cooperation between attending physicians, hepatologists, nursing
staff, dieticians, and family members. ESPEN guidelines recommend
energy intake of 30-35 kcal/kg/day and protein intake of 1.2-1.5 g/
kg/day as standard advice. Frequent meals with late evening snacks
are important to practice. Patients with CLD have been successfully
treated clinically with BCAA supplements. Potential benefits of
BCAAs include positive effects on ammonia detoxification with
decreased ratio of BCAAs to AAAs, liver regeneration, albumin
synthesis, immune and hepatic function, glucose metabolism, and
thus improved outcome in CLD.
Acknowledgment
The authors would like to thank Dr. Swati Lad Naik MBBS,
MD Pharmacology, Ms. Deepal Mistry, B. Pharm, D.B.M., and HP
INFINEX for medical writing support.
References
Citation
Lawate PS, Sharma M. Cirrhosis, Hepatic Encephalopathy, and Liver Transplantation - Special Focus on Nutrition. Indian J Nutri. 2022;9(3): 270.