Research Article
Study of Milk Adulteration in Hyderabad, Telangana State
Madhuri A*, Bai BK, Anisha A, Ankitha E, Sushma C, Poojitha M and Sushma MS
Department of Microbiology, Government Degree College for Women, Begumpet, Hyderabad, Telangana, India
*Corresponding author: Madhuri A, Department of Microbiology, Government Degree College for Women, Begumpet,
Hyderabad, Telangana, India; E-mail: a.madhuri108@gmail.com
Article Information: Submission: 07/05/2022; Accepted: 28/05/2022; Published: 31/05/2022
Copyright: © 2022 Madhuri A, et al. This is an open access article distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Food adulteration is age old practice and is an issue of concern throughout world. The threat of adulteration in food is seen more in developing countries
due to lack of proper monitoring facilities. However, this can be one foremost common phenomenon that has been overlooked in many countries. Unfortunately,
milk adulterants can pose serious health hazards resulting in fatal diseases. This paper showed the results for presence of milk adulterants in 20 milk samples
collected from various places from Hyderabad. We have checked for adulterants like urea, formalin, detergent, salt, maltodextrin etc. and we observed that
majority of the samples are positive for formalin, maltodextrin and salt. These adulterants can result in health problems like liver damage, allergic reactions,
weight gain etc. Other adulterants are found in less number of samples.
Keywords
Milk adulteration; Food adulteration; Viable count; Antibiotic sensitivity
Introduction
Milk is a very important source of nutrients required for growth
in infants and kids and for maintenance of health in adults. Milk is
the lacteal secretion produced from the mammary glands of the
mammals. Milk in its natural form has very high nutrient value. It
gives nutrients like carbohydrates, vitamins, fat, protein and minerals
in moderate amount which can be easily digestible [1].
Normally, the adulteration in food is done either to gain
benefits in monetary form or due lack of proper hygienic
conditions of processing, storing, transportation and marketing.
This ultimately ends in cheating of consumers or become victim of
diseases. Such adulteration is quite common in developing countries.
Milk is produced throughout the year. However, milk production is
decreased to great extent due to the stress of heat and fodder shortage
in summer. Milk is transported from point of production to cities
mainly through middlemen. Such milk is added with adulterants
to make more profits by adding materials like starch, flour, urea,
cane sugar and edible fat as adulterants. Milk dealers may either
dilute the milk or extract valuable components and there after
add cheap substances to maintain its compositional parameters. These cheap substances include starch, urea, and preservatives like
formalin, hydrogen peroxide, boric acid and various antibiotics [1].
An editorial of esteemed e-paper “Economic Times”, in September
2018 stated that around 68.7% of milk and milk products sold within
the country is not as per the standards laid down by the Food Safety
and Standards Authority of India (FSSAI) [2].
Many types of adulterants are added into milk to gain financial
benefit but the effects they cause may be dangerous [3]. The external
addition of water in milk though contains no health hazards
associated with it but the water used is contamination free but dilutes
the quality of nutrients in milk. One of the important parameters in
estimating the quality of milk is total SNF (Solid-Not-Fat) content.
Some adulterants added to milk can enhance the SNF content of the
milk however alters the sanctity and purity of milk [4]. Sometimes in
order to increase the natural protein content in the milk, melamine
is added [5]. Some amount of Melamine can be present in milk
samples acquired during milk packaging and use of nitrogen rich
fertilizers. According to The US Food and Drug Administration
(FDA), maximum permissible limit of melamine in milk is 50 ppb,
whereas The Food Safety and Standards Authority of India (FSSAI)
have set melamine limits from 0.5 ppm to 2.5 ppm [6]. Urea is also added sometimes to increase non-protein nitrogen content of milk.
Permissible limit given for urea by FSSAI is 70 mg / 100 mL [7]. In
some areas, Formalin is added to milk as preservative but it can result
in renal problems.
There are reports of milk adulteration with other preservatives
such as potassium dichromate, benzoic acid, hydrogen peroxide,
salicylic acid etc. putting the common people’s health at stake [3].
Another component that is found frequently in milk, to maintain the
required density is ammonium sulphate [4]. The addition of detergents
gives frothy appearance to milk [8]. Whey is added to milk because
it promotes an increase in the volume of milk without significantly
changing the percentage of proteins or effecting noticeable sensory
changes for most people [9]. The other adulterants added to milk
are starch, to increase the SNF content and Formalin, Salicylic
acid, Benzoic acid and Hydrogen peroxide act as preservatives and
increase the shelf life of the milk [3,4]. Most of the above mentioned
compounds decrease the quality of milk but simultaneously cause
serious health risk to consumers. Melamine if consumed over a
prolonged duration may cause kidney failure and can sometimes
prove to be fatal, especially to infants [4].
Methodology
Detection of urea in milk: To 1 ml of milk in a test tube, 1 ml of
1.6% (w/v) DMAB reagent was added and mixed well. Positive result
is indicated by yellow colour [10].
Detection of formalin in milk: This detection was done by
leach test. In this 5ml milk is taken in test tube. Then it is added with
concentrated HCl and ferric chloride. Then the tube is kept in water
bath for 3-4 min. Positive result is indicated by brownish pink colour
and negative result by white colour [10].
Detection of salt in milk: Salt was detected in milk by silver
nitrate test. In this reagents like silver nitrate and potassium
dichromate were used. To 5 ml of milk in test tube, 1 ml of 0.1 n silver
nitrate solutions were added and mixed thoroughly. To this again 0.5
ml of 10% potassium dichromate solution was added. Positive result
is indicated by yellow colour and negative result by red colour [10].
Detection of detergent in milk: Detergent in milk sample was
tested by using methylene blue dye and chloroform. In this test, 1ml
of milk sample is taken in 15 ml test tube. To the milk sample 1 ml
of dye solution was added followed by addition of 2 ml chloroform.
Then the contents were vortexed for 15 seconds and centrifuged at
1100 rpm for 3 min. In this case intensity of blue colour in the upper
and lower layer was checked. Positive result is indicated by presence
of more intense blue colour in the lower layer and negative result in
indicated by blue colour in the upper layer [10].
Detection of maltodextrin in milk: Maltodextrin in the milk
sample was tested by Iodine reagent. In this test 5 ml milk sample
is added with 2ml Iodine reagent. The contents were mixed well
and observed for colour change. The positive result was observed by
chocolate red colour and negative result by slight yellow colour [10].
Detection of cane sugar in milk: Cane sugar in milk sample
was tested by resorcinol test. In this method, reagents resorcinol
and conc. HCl were used. 5 ml of milk is taken in test tube, to that 1 ml of concentrated HCl and 0.1g resorcinol were added and mixed.
The tube is kept in boiling water bath for 5 min and then observed
for colour change. Positive result was indicated by red colour and
negative test by no colour change [10].
Detection of hydrogen peroxide in milk: Hydrogen peroxide in
milk samples was tested by using reagents like potassium iodine and
starch. In this method first 1 ml of milk is taken in test tube, and then
to this, 1ml of a mixture of potassium iodine-starch reagent is added
and mixed well. The tubes are observed for colour change. Positive
colour is indicated by blue colour and negative colour is indicated by
white colour [10].
Detection of sucrose in milk: Sucrose presence in milk was tested
by molisch test. In this test, molisch reagent and Conc. H2SO4 were
used. In this test 1ml of milk sample is taken in the test tube and to
these 2-3 drops of Molisch reagent was added and then 1 ml of Conc.
H2SO4 was added through walls of the test tube. Positive result was
indicated by the violet ring formation at the junction of two layers
[10].
Kirby-Bauer method: Antibiotic sensitivity test is done by Kirby-
Bauer method. In this method, 1:10 diluted milk samples are spread
on nutrient agar plates and antibiotics discs (Penicillin, Amoxicillin,
Tetracyclin, Gentamycin) were placed on nutrient agar and incubated
at 37ºC for 24 hours. After allowing the bacteria to grow overnight,
areas of clear zone surrounding the discs indicate that the antibiotic
inhibited bacterial growth. The concentration of antibiotic that
diffuses into the media decreases with increasing distance from the
source [11].
Results & Discussion
We have collected 20 milk samples from different areas
of Hyderabad. For these samples we have checked for various
adulterants like urea, formalin, salt, detergent, maltodextrin, cane
sugar, hydrogen peroxide and sucrose. The results are given in the
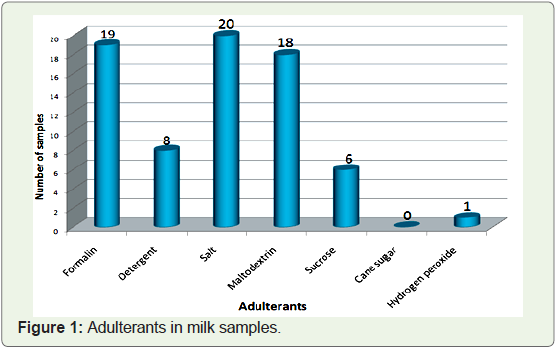
following Table 1 (Figure 1).
Out of 20 samples, Formalin, maltodextrin and salt showed
positive result for 19, 18 and 20 samples respectively. The presence
of these adulterants in majority of the samples may result in
health diseases for example formalin may cause liver damage and
maltodextrin may result in side effects like allergic reactions, weight gain, gas, flatulence and bloating. It may also cause allergic reactions,
asthma, cramping and difficulty in breathing. Detergent found to be
positive in 8 samples, and sucrose is positive in 6 samples whereas urea
and hydrogen peroxide were positive in 3 and 1 samples respectively.
These results show that adulteration is present in majority of the milk
samples and they can also cause considerable health issues.
The present study reported the presence of salt in all milk samples
tested. This is similar to the findings of the study by Hemanth and
Sukumaran (2014) [12], Riya and Gurmeet (2022) and Arun Kumar
et al (2015) where the presence of salt was found in 82, 82 and 80%
of samples, respectively [13,14]. However, Brindha et al (2017)
reported fewer samples were added with salt, i.e. 13-16% [1]. This
study also showed that the presence of formalin was more prevalent
in milk samples; i.e., 95% of samples in the present study tested
positive for formalin. The presence of formalin in studies conducted
by Hemanth and Sukumaran (2014) [12], Riya and Gurmeet (2022)
and Arun Kumar et al (2015) was found to be 32%, 32% and 30%
respectively [13,14]. Next adulterant found in the majority of samples
was maltodextrin , i.e., 90% samples showed positive result for
maltodextrin presence, whereas Amita et al (2021) found it to be 50%
[15]. Detergent was found to be present in 40% of samples in our
study. This result is in accordance with Hemanth and Sukumaran
(2014) [12]; Riya and Gurmeet (2022) and Arun Kumar et al (2015)
[13,14], where detergent was found to be present in 44%, 32% and
44% respectively. Sucrose was found to be present in 30% of our
samples; where as Hemanth and Sukumaran (2014) found 22% of
samples were positive for sucrose [12]. The presence of hydrogen
peroxide was found in 5% of samples in our study. However, Brindha
et al (2017) did not find Hydrogen peroxide in any sample [1], and
Hemanth and Sukumaran (2014) [12], Riya and Gurmeet (2022) and
Arun kumar et al (2015) observed its presence in 32%, 44% and 36%
of samples, respectively [13,14].
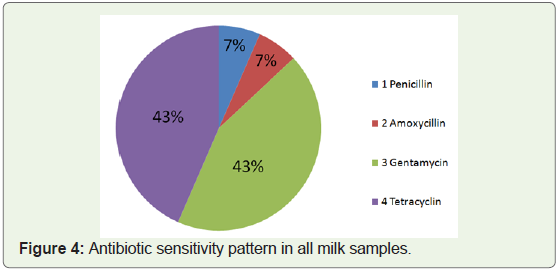
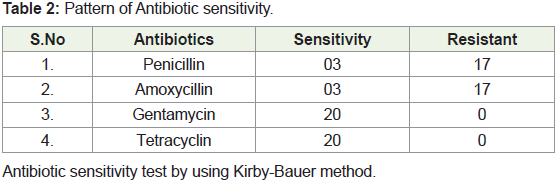
Antibiotic sensitivity:
For all the 20 samples we have conducted Antibiotic sensitivity
test by Kirby-Bauer method. In this we have used penicillin,
amoxicillin, gentamycin and tetracycline (Table 2). In this method
we have taken 1:10 dilution of milk sample and spread on the nutrient
agar plates (Figure 2). Then different antibiotic discs were placed on
them and incubated at 37ºC for 24 hours. In this test, it was observed
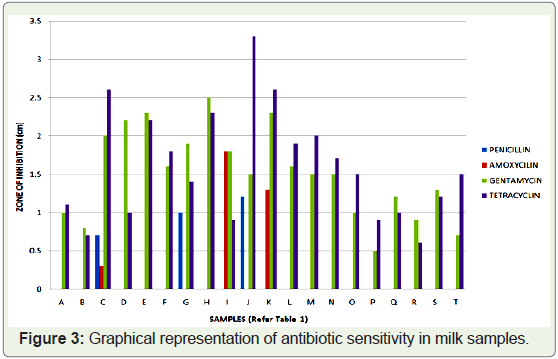
that for penicillin and amoxycillin antibiotics, out of 20 samples, only
3 samples showed some sensitivity, whereas bacteria present in all 17
samples showed resistance towards these antibiotics. For tetracyclin
and gentamycin, zone of inhibition was seen in all the 20 samples
and the diameter of zone of inhibition is in the range of 0.5 to 3.3
cm. Similar experiments were conducted by Reta et al (2016) [16],
where they observed that Staphylococcus aureus isolated from milk
samples showed resistance rate of 93.1 towards penicillin followed by
tetracycline (69 %), and very low level of resistance to vancomycin
(6.9 %) and rifampicin (3.4 %) (Figure 3 & 4).Conclusion
It is observed that most of the milk samples taken by us are found
to be added with some adulterants. We have checked for viable count
and antibiotic sensitivity also. In these we have observed that there is
considerable viable count in most of the samples and for antibiotic
sensitivity most of the samples showed resistance towards penicillin and amoxycillin and they were sensitive towards gentamycin
and tetracyclin. Milk is one of the best nutrition sources so, while
choosing the source, we have to be careful and select a good sample
for consumption so that we cannot get affected by possible health
problems.
To stop milk adulteration, the regulatory bodies, public administration, scientific communities should work together to
bring an end to all such unethical malpractices at large. Giving right
information to the consumers and making detections of adulterations
easy and exercising awareness campaigns can drastically reduce this
malpractice.
References
Citation
Madhuri A, Bai BK, Anisha A, Ankitha E, Sushma C, et al. Study of Milk Adulteration in Hyderabad, Telangana State. Indian J Nutri. 2022;9(2): 258.