Research Article
Milk Consumption Patterns among Persons with Type 1 Diabetes Mellitus from Mumbai city – an Exploratory Study
Goyal A1, Mehta A2, Salis S3, Desai S4, Udipi SA5, Vaidya RA6 and Vaidya ADB7
1Junior Research Fellow, Kasturba Health Society’s Medical Research Centre, Mumbai, India
2Consulting Diabetologist, Kasturba Health Society’s Medical Research Centre, Mumbai, India
3Director, Nurture Health Solutions, Mumbai, India
4Post-doctoral Fellow, Kasturba Health Society’s Medical Research Centre, Mumbai, India
5Research Director, Kasturba Health Society’s Medical Research Centre, Mumbai, India
6Honorable Director, Div. of Endocrine and Metabolic Disorders, Kasturba Health Society’s Medical Research Centre, Mumbai, India
7Professor Emeritus, Kasturba Health Society’s Medical Research Centre, Mumbai, India
*Corresponding author: Goyal A, Junior Research Fellow, Kasturba Health Society’s Medical Research Centre, Mumbai, India;
Phone: +91-9029287575; E-mail: goyal.aditi157@gmail.com
Article Information: Submission: 13/06/2021; Accepted: 20/07/2021; Published: 24/07/2021
Copyright: © 2021 Goyal A, et al. This is an open access article distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Reports of epidemiological studies in the Western world, strongly suggest that consumption of A1 milk is associated with increased risk of Type 1 Diabetes
Mellitus, Autism Spectrum Disorders, etc. However, very few studies in India have addressed this issue. Therefore, an exploratory study was undertaken to
examine the milk consumption patterns of persons with T1DM attending clinics in Mumbai city and their awareness about A1 and A2 milk. Ninety six people
with T1DM were approached, of which seventy five agreed to participate. The average age of participants was 22.4 years (3 - 52 years) (Median = 21 years).
The mean age at diagnosis of type 1 diabetes was 9.5 years (6 months – 29 years) (Median = 9). Among the 75 participants, 64 consumed milk, but 11 did
not consume milk. Almost half the participants (n = 30) had heard about A2 milk. Only ten participants (14.7%) had begun consuming A2 milk when it became
commercially available, for reasons of their own or family members’ health. Knowledge about A1/A2 milk and the possible role of A1 milk in the onset of
diabetes was relatively poor and only twelve participants had some knowledge about the possible health implications.
Keywords
Milk; BCM7; Type 1 Diabetes mellitus, A1/ A2 milk
Abbreviations
T1DM -Type 1 Diabetes mellitus; BCM7 - β-casomorphin 7; SD -
Standard deviation; NIN - National Institute of Nutrition; NOD mice
- Non-obese diabetic mice; ASDs - Autism Spectrum Disorders; SIDS
- Sudden Infant Death Syndrome; MPO - Myeloperoxidase activity;
MCP-1 - Monocyte chemotactic protein; IL4 - Interleukin 4; IgE -
Immunoglobulin E; IgG - Immunoglobulin G
Introduction
According to the 9th edition of the International Diabetes Federation Diabetes Atlas (2019) [1], one in six adults with diabetes
is from India. As of 2019, India is home to the second largest number
of children and adolescents with Type 1 diabetes mellitus (T1DM)
globally, with 171,281 patients in the age group 0-19 years. Due to
its large population, India adds15,900 children, the highest number
of children with T1DM, per year in the age group of 0 to 14 years
[1]. Das (2015) reported a T1DM prevalence of 3.2 cases/ 100,000
children in Chennai [2], 10.2 cases/ 100,000 children in Karnal
and17.93 cases/ 100,000 children in Karnataka. Dhingra and Kalra
(2018) reported that in India the prevalence of T1DM is more than 10/100,000 population [3], but in some urban pockets the prevalence
is > 30/100,000 population. It is estimated that the increase in
incidence is about 3 to 5 percent per year.
One of the risk factors identified in the multifactorial pathology
of T1DM that is receiving attention since about two decades, is the
consumption of A1 milk. The A1/A2 hypothesis states that exposure
to the peptide β-casomorphin-7 (BCM-7), formed after the digestion
of A1 milk, may act as an immunosuppressant, impair tolerance to
dietary antigens in the gastrointestinal tract and thus contribute to
the onset of T1DM [4,5]. This has led to growing concern about A1
milk in the world and in this country as well [6].
India is the largest producer of milk in the world and traditionally
Indian cattle breeds produce A2 milk that does not lead to the
formation of the BCM-7 peptide. However, in order to augment milk
production, cross breeding of bovines with breeds such as Holstein
was done [6]. At this time, the A1 allele entered in the genome,
resulting in production of milk that would lead to the formation of
BCM-7. We, in our laboratory analyzed milk produced by pure bred
Gir cows as well as Holstein Frisian and Jersey cows crossbred with
other indigenous breeds. We found that pure bred Gir cows produce
A2A2 milk whereas Holstein Frisian and Jersey cows crossbred with
other indigenous breeds produce A1A2, A1A1 as well as A2A2 milk
(Unpublished Data).
Given that milk is widely recommended globally for its health
benefits throughout the life cycle, particularly in India where it is
consumed in almost every household, there is cause for concern,
when health properties of A1 milk are questioned.
In this context, we undertook a survey to assess the milk
consumption practices in persons with Type 1 diabetes and their
knowledge regarding A2 milk.
Materials and Methods
Ethics Approval:
The study was approved by the Intersystem Biomedica Ethics
Committee (ISBEC) (Approval No. ISBEC/NR-30/KM-MN/2019)
(July 22, 2019).Sample Selection:
Subjects were persons with T1DM attending two clinics that
specialised in diabetes care and were located in a Western suburb in
Mumbai city. Participants were recruited after obtaining informed
consent.The study was conducted between July 2019 and April 2020.
Ninety six participants were contacted, among whom seventy five
consented to participate in the study.
Data Collection:
Information about age at diagnosis, medical history, family
medical history was collected from the participants by a trained
dietician using an interview schedule. Present age and age at
diagnosis was calculated from date of birth (received from 74 out of
75 participants). Information regarding milk consumption patterns
included current practice for consumption of milk and milk products, type and amount of milk consumed, frequency of milk consumed
per day, presence of symptoms of lactose intolerance and knowledge
about A1/A2 milk.Data Analysis:
Results were tabulated in MS Excel (version 10) and data was
analysed using SPSS version 20. The 75 participants were categorized
into two groups based on the type of milk consumed: Group
1consisted of those who consumed cow’s milk or cow and buffalo
milk but were not aware of the type of milk i.e. A1 or A2, and Group
2 included those who consumed A2 cow or buffalo milk. Milk nonconsumers
were placed in a third group, Group 3. One way Analysis
for Variance was applied for statistical comparison.Results
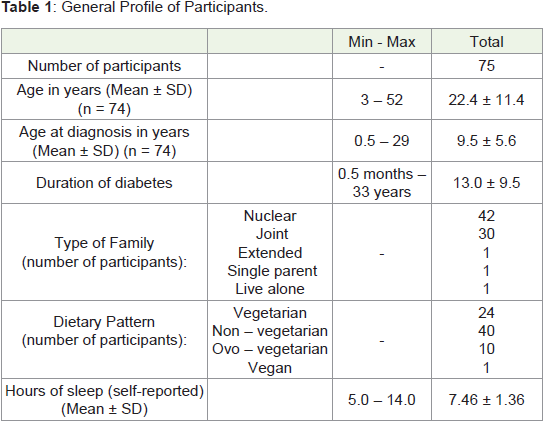
Profile of Participants:
Among the 75 participants, 30 participants (40%) were males and
45 (60%) were females. Mean age of the participants was 22.4 years ±
11.4 years (Median = 21 years). Age at diagnosis varied from 0.5 years
to 29 years, with the mean age at diagnosis being 9.5 years (Median
= 9). Average duration of being diabetic was 13.0 years. The general
profile of participants is given in (Table 1).Milk Consumption:
Sixty four of the 75 participants (85.3%) consumed milk. Among
these 64, 36 participants (48%) were consuming cow milk or cow +
buffalo milk (Group 1), and 28 participants (37.3%) reported that
they were consuming A2 cow or buffalo milk (Group 2); of which,
11 consumed A2 cow’s milk and 17 consumed buffalo milk. Eleven
participants (14.7%) were not consuming milk at all (Group 3). A
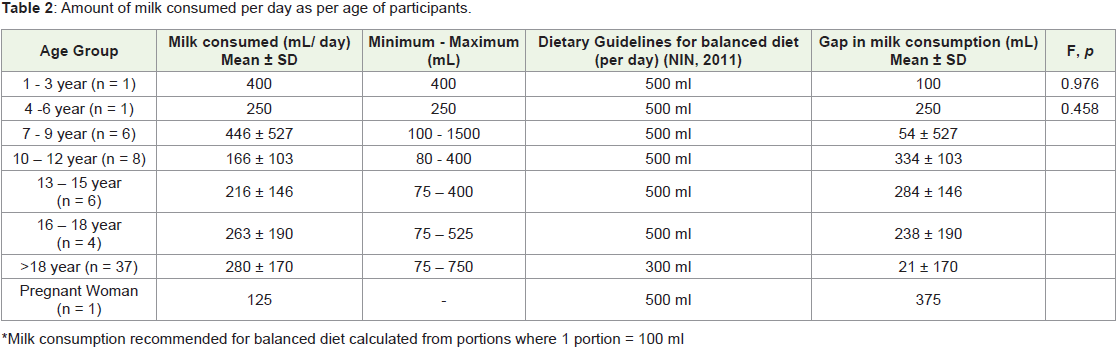
total of 19 different brands of milk were identified in our study group.One-third of the participants (33.3%, n = 25) consumed milk
twice per day, and one - fourth consumed milk once in a day (25.3%,
n = 19). Average milk intake of participants per day was calculated by
age group. The intake was compared to the amount recommended
by the National Institute of Nutrition (2011) to be consumed as part
of a balanced diet. In the age-groups of 1-3 years [7], 7-9 years and
>18 years, on average about 80% of the recommended milk per day
was consumed. In contrast, among the 4- to 6-year-old participants,
only 50% of the recommended intake was met. Participants in the age
groups 10-12 years (29.4%), 13-15 years (37%), 16-18 years (42%) and
pregnant woman (25%) all had less than 50% of the recommended
milk consumption (Table 2).
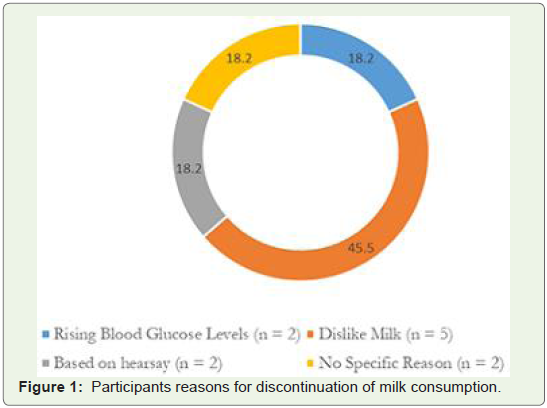
Eleven participants (14.7%) did not consume milk. Among
these 11 participants, 10 had stopped consuming milk and/or milk
products soon after they were diagnosed with diabetes. Only two
(18.2%) participants stopped consuming milk because it caused their
blood glucose levels to rise. Five participants had stopped consuming
milk because they disliked it (45.5%). Two participants (18.2%)
excluded milk and its products from their diet based on hear say
information available online and two participants (18.2%) did not
provide any reason for discontinuing milk consumption (Figure 1).
All 11 participants took the decision on their own, without consulting
their supervising diabetologist or dietician.
Consumption of milk products:
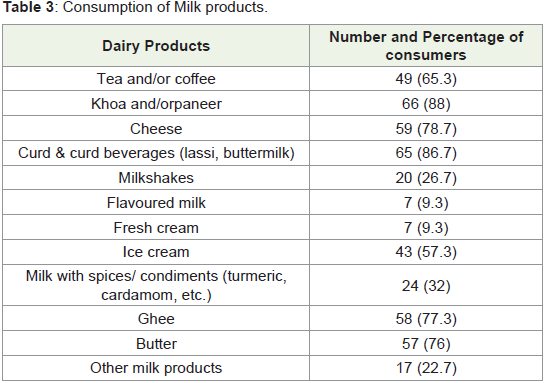
Besides milk, participants were asked about consumption of
milk products. The most commonly consumed dairy products were
paneer and/or khoa (88%, n = 66), followed by curd and curd- based
beverages (86.7%, n = 65) (Table 3). Flavoured milks and fresh cream
were not commonly consumed (9.3%, n = 7 for each product).Knowledge about A1/ A2 milk:
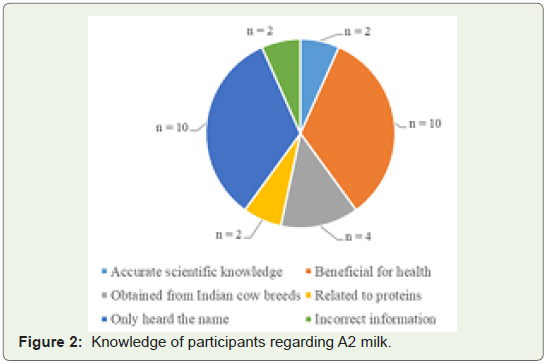
Of the 75 participants, 30 (40%) were aware about A1 and A2
milk, whereas 45 (60%), were not. Of the 30 participants who were
aware, only 2 participants (6.7%) knew about the scientific debate
pertaining to A1/A2 milk. One-third of the participants (33.3%,n =
10) opined that A2 milk is beneficial for health, four (13.3%) were
aware that it is obtained from Indian cow breeds, and two (6.7%) were
aware that the topic was related to proteins. Ten participants (33.3%)
did not know anything concerning A1/ A2 milk, and the remaining (n
= 2, 6.7%) gave incorrect information about A1 and A2 milks Figure 2.Shift to A2 milk:
Twenty participants (26.7%) reported that they had recently
changed the milk that they used to habitually consume. Three
participants had shifted to soy milk, four were using cows and buffalo
milk both, and two were using cow’s milk of a different brand. The
remaining 11 (14.7%) participants had stopped using the milk they
had used previously, although they did not know the type of milk
they were using and switched to using either A2 cow’s milk or buffalo
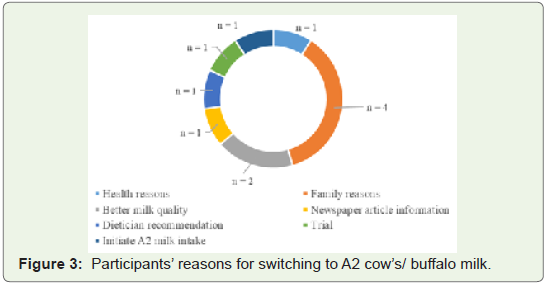
milk.Various reasons were given for switching to A2 milk/ buffalo milk
(Figure 3). The reasons were:
• For health reasons (n = 1, 9.1%)
• Because another member of the family switched to A2 milk
(Family Reasons) (n = 4)
• For better quality of milk (n = 2, 18.2%)
• Based on a newspaper article (n = 1, 9.1%)
• On the recommendation of a dietician (n = 1, 9.1%)
• To try A2 milk (n = 1, 9.1%), and
• To initiate A2 milk consumption (n = 1, 9.1%).
Discussion
One of the first studies to show a correlation between consumption
of A1 containing milk in T1DM was reported by Elliot et al., (1999)
[8]. Since this report, evidence comes largely from in vitro and
animal studies. The diabetogenicity of bovine milk is based on
observations and apparent incidence figures, and evidence regarding
this association in humans comes largely from ecological studies [6].
The Diabetes Autoimmunity Study in the Young (DAISY) (2014)
[9], found that cow’s milk increased the risk of autoimmunity in
children at a low genetic risk for T1DM and there were higher chances
of progression to T1DM [9].
Animal models have also been used to study this correlation.
China and co-workers (2018) conducted an intergenerational study
with adult NOD mice [10], where in they compared A1 or A2
β-casein supplemented diets. There was no difference observed in the
incidence of diabetes in the first two generations. However, in the F3
generation, the incidence was doubled in the mice fed the A1 β-casein
supplemented diet and in the F4 mice, they found subclinical insulitis and altered glucose handling was evident as early as 10 weeks of age
in the A1 group. They concluded that possibly the adverse effects of
dietary A1 β-casein on glucose homeostasis and development of type
1 diabetes may manifest in succeeding generations.
Epidemiological and animal studies have thus shown that
consumption of A1 β-casein and other such variants with histidine
at the 67th position could be potential risk factors in the onset of
diabetes [8,10,11].
However, the evidence regarding this association is fairly
equivocal. Savilahti & Saarinen (2009) earlier found no association
between early exposure to cow’s milk and development of T1DM
[12]. A study conducted with a murine model showed that different
casein hydrolysates did not significantly alter biochemical parameters
in healthy and diabetic rats [13].
While the negative effects of A1 milk and BCM7 have received
great attention, the possible benefits of this in terms of mucus
secretion [14,15], increased activity of superoxide dismutase and
catalase [16], increased levels of prolactin and its analgesic role, the
development of innate immunity, lymphocyte proliferation and
cellular immunity have apparently been downplayed [17]. There are
reports from animal studies that BCM-7 has a protective role against
hyperglycaemia and free radical- mediated oxidative stress [18].
In contrast, variants of β-casein have also been implicated in
diseases such as ischemic heart disease, ASDs, schizophrenia, SIDS,
gastric discomfort, etc. [19,20]. A study conducted to evaluate the
inflammatory response of β-casein showed that A1A1 and A1A2
case in variants caused a significantly higher rise in myeloperoxidase
activity (MPO), monocyte chemotactic protein (MCP-1) level,
interleukin 4 (IL4), as well as IgE and IgG levels compared to A2A2
β-casein [21]. The paradoxical responses of various experiments with
regard to the physiological implications of A1 β-casein consumption,
therefore, warrant further investigation before deciding on whether
A2 milk should be preferred over A1 milk.
The debate about A1 /A2 milk and several brands marketing A2
milk motivated us to determine whether individuals with T1DM
associated it with milk consumption or whether they had changed
their milk consumption practices; and their knowledge about A1/2
milk, after diagnosis.
In the present study, we found that none of the participants
associated consumption of milk as a cause of T1DM. Another notable
observation in this study was that some participants had chosen to
consume A2 milk. It is noteworthy, that this was a self-made decision
on the part of all the 11 participants. Only one participant made
the change upon receiving the recommendation from a dietician,
whereas the other 10 participants chose A2 milk without consulting
their diabetologist and/or dietician.
We also found that knowledge of A1/A2 milk was quite poor.
Among the 75 participants, only 30 participants were aware about A1
and A2 milks. Only two participants were aware about the details of
the A1/A2 controversy, suggesting the level of knowledge among our
participants was relatively poor. These findings show that there is still
much to be understood about people’s perception regarding milk and
its role in diabetes mellitus, particularly in T1DM.
Among the 75 participants, 64 consumed milk with the amounts
varying from 75 mL to 1500 mL per day (273 ± 220 mL). On average,
adults in urban India consume 120.7 g of milk and milk products,
whereas mean consumption among rural adults is 117.9 g [22].
In a Nestle Nutrition workshop in 2011 [4], it was pointed out
that there are several, infact there are 13 different variants of β-casein
in dairy cattle; and A1, A2 and B, which are variants in dairy cattle
are also present in human milk. The amino acid sequences of these
β-casomorphins bear considerable similarity regardless of whether
the source is bovine or human milk. In this workshop it was noted that
epidemiological studies and animal models do not provide supportive
evidence of association of these milk proteins even in breast milk and
the development of T1DM. The European Food Safety Authority
concluded that “a cause and effect relationship is not established
between the dietary intake of BCM7 (beta-casomorphin-7), related
peptides or their possible protein precursors and non-communicable
diseases” [23].
Globally and locally within India, while the debate continues over
the potential health implications of A1/A2 milk, the perception that
A1 milk is harmful to human health, seems to be regarded seriously
by some segments of the scientific community [5,6,14-17]. While the
A1/A2 hypothesis is intriguing and if it is conclusively proven that A1
milk does increase the risk of T1DM and cardiovascular diseases, it
will have important public health connotations. However, presently
available data is not sufficient enough to be able to draw a conclusion
about a cause-and-effect relationship. More studies in vivo and with
animals; and generation of data with humans are required.
This study is a part of a larger study where we intend to explore
the knowledge of A1/A2 milk and their health implications in multiple
cohorts including bio-medical professionals. Our study can be
considered preliminary, where none of our 75 participants indicates
that majority of our participants did not associate milk consumption
with negative implications for health. Our study had limitations
in terms of lack of information whether the commercially available
popular brands of cow’s milk are either A1 or a mixture of A1 and A2
milk. Also, some brands did not clearly state whether the source of milk
was cow or buffalo. Since the study was restricted to Mumbai city, it
may be worthwhile to undertake a well-designed epidemiological study
on a larger representative population to investigate the physiological
effects of A1 milk or A2 milk or buffalo milk using available diagnostic
techniques to determine whether people with diabetes have antibodies
to the BCM peptide. This is particularly important considering the
rising incidence of T1DM in India.
Acknowledgement
We acknowledge and thank the Bombay Gow Rakshak Trust
for funding this work. We also extend our sincere gratitude to Dr.
Prajakta Paradkar for her help during the writing of this manuscript.
References
3. Kalra S, Dhingra M (2018) Childhood diabetes in India. Ann Pediatri Endocrinol Metab 23: 126-130.
Citation
Goyal A, Mehta A, Salis S, Desai S, Udipi SA, et al. Milk Consumption Patterns among Persons with Type 1 Diabetes Mellitus from Mumbai
city – an Exploratory Study. Indian J Nutri. 2021;8(3): 237.