Review Article
Probiotics in Pregnancy
Tania SG*
Head, Department of Obstetrics and Gynecology, GNS Multispeciality Hospital, Chattarpur, New Delhi, India
*Corresponding author: Tania SG, Head, Department of Obstetrics and Gynecology, GNS Multispeciality Hospital, Chattarpur,
New Delhi, India; Tel: 9625930047; Email: taniasingh.ts@gmail.com
Article Information: Submission: 22/04/2021; Accepted: 28/05/2021; Published 31/05/2021
Copyright: © 2021 Tania SG. This is an open access article distributed under the Creative Commons Attribution License, which
permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Probiotics are live microorganisms (in most cases bacteria) that are similar to beneficial microorganisms found naturally in the human gut. They are
available mainly in the form of dietary supplements and foods, and when administered in adequate amounts confer a health benefit on the host. They are a safe
and effective way of enhancing the diversity and health of the microbiome in pregnant women. Probiotics given prenatally are an important way for mothers to
safeguard their health during pregnancy as well as the health of their babies.
Colonization of the neonatal gut by beneficial bacteria is important in protecting the neonate from enteric pathogens and local as well as systemic
inflammation. Maternal microbiome in pregnancy together with infant diet impacts neonatal microbiome.
Keywords
Microbiota; Dysbiosis; Synbiotics; Bifidobacterium; FODMAP; Lactobacillus; Prebiotics; Pregnancy; Urogenital infections; Infant colic
Introduction
Gut microbiota is a heterogeneous microbial community that
includes 1014 [1,2] microorganisms comprising predominantly
bacteria, along with viruses, archaeans, and protozoa. The
gastrointestinal microbiota can be considered as an organ within an
organ contributing to host nutrition, developmental regulation of
intestinal angiogenesis, protection from pathogens and development
of the immune response [3]. Three dominant phyla comprise almost
80% of gut flora [4]: Bacteroidetes, Firmicutes, and Actinobacteria
[5].
The neonatal microbiota is highly different compared to the adult
one, since the former is characterized by rapid changes especially in
the first year of life [6]. Colonization of the neonatal gut by beneficial
bacteria is important in protecting the neonate from enteric pathogens
and local as well as systemic inflammation. Maternal microbiome in
pregnancy together with infant diet impacts neonatal microbiome.
Dysbiosis in pregnancy increases the risk of preeclampsia, diabetes,
infection, preterm labour, and later childhood atopy. It can also lead
to postnatal maternal depression and infant colic. It also plays an
important role in necrotizing enterocolitis and sepsis, both of which
can occur as a result of prematurity affecting the long term outcomes
in neonates. Administration of enteral prebiotics, probiotics, and
synbiotics during pregnancy, lactation, and postnatal life appears to be a safe and feasible method to alter the maternal and neonatal
microbiome, thus improving pregnancy and neonatal outcomes [7].
Synbiotics refers to a product which has an appropriate
combination of both probiotic and prebiotic components. Synbiotics
ensures that it has a superior effect when compared to the activity of
probiotic or prebiotic alone. It was developed in order to overcome
some possible difficulties in the survival of probiotics in the
gastrointestinal tract.
Prebiotics:
Prebiotics generally refers to a substrate (non-digestible food
ingredient) which, when added to the dietary intake, is selectively
used by host microorganisms conferring a health benefit [8] by
selectively stimulating the growth and/or activity of one or a limited
number of bacteria in the colon, especially Bifidobacterium species
[9]. Prebiotics evade digestion in the small intestine and must be
selectively fermented in the colon.Prebiotics are typically comprised of nondigestible carbohydrates
but can also include non-carbohydrates such as fatty acids, phenolics,
and phytochemicals [8].
The most widely accepted prebiotics are together referred
to as FODMAPs (Fermentable Oligosaccharides Disaccharides
Monosaccharides and Polyols). The types of FODMAPs which hold prebiotic effects are mainly fructans, galacto-oligosaccharides (GOS),
and inulin. Foods high in fructans include wheat products, rye
products, onion and garlic. Foods high in GOS include legumes, such
as chickpeas and baked beans. Inulin is found in some yoghurts (even
some lactose-free), asparagus, garlic/onion, and some confectionary.
The reason fructans and GOS are able to act like prebiotics is because
our gut doesn’t have enzymes to break them down, so they remain
undigested like prebiotics [10,11].The down side to a low FODMAP
diet is that it often limits the intake of prebiotics (Table 1).
Table 1: Foods High in Fermentable Oligosaccharides, Disaccharides, Monosaccharides, and Polyols (FODMAPs).
Prebiotic carbohydrates are a major substrate for bacterial
growth, selectively stimulating the growth and/or activity of beneficial
members of the gut microbiota, particularly bifidobacteria [12,13].
Short-chain prebiotics are mainly fermented in the cecum and
colon ascendens. Long chain oligosaccharides are fermented along
the entire colon [14].Thus, prebiotics could, at least in theory, have
more global effects on colonization than adding a single probiotic
strain. A second, more direct immune effect appears to be mediated
by the fermentation products of prebiotics. Gut microorganisms
ferment prebiotics to produce short-chain fatty acids (SCFAs) that
have direct anti-inflammatory effects [15]. SCFAs also promote
intestinal integrity through effects on epithelial cell proliferation
and differentiation [16]. Animal and human studies suggest that
prebiotics may directly affect both mucosal and systemic immunity
[17-19]. However, more studies are needed to confirm that these are
clinically relevant effects.
The gastrointestinal microbiota of breastfed babies differ
from classic standard formula fed infants. While mother’s milk
is rich in prebiotic oligosaccharides and contains small amounts
of probiotics, standard infant formula doesn’t [14]. Different
prebiotic oligosaccharides are added to infant formula: galactooligosaccharides,
fructo-oligosaccharide, polydextrose, and mixtures
of these which brings infant formula one step closer to breastmilk.
Prebiotics are present as supplements also, and they’re sometimes
added to probiotic supplements or yoghurts.
Probiotics:
The United Nations Food and Agriculture Organization (FAO)
and World Health Organization (WHO) have defined probiotics
as “live micro-organisms, which when administered in adequate
amounts confer a health benefit on the host” with a wide and varying
range of clinical and immunologic capacities [20].The term probiotic
means “for life” and it is currently used to name bacteria associated
with beneficial effects for humans and animals [21].The concept of probiotics was introduced in the early 20thcentury
by Elie Metschnikoff, the Russian born Nobel Prize winner working
at the Pasteur Institute, who suggested that “The dependence of the
intestinal microbes on food makes it possible to adopt measures to
modify the flora in our bodies and to replace the harmful microbes
by useful microbes” [22]. At the same time, Henry Tissier, a French
paediatrician, observed that children with diarrhea had in their
stools a low number of bacteria characterized by a peculiar, Yshaped
morphology. These “bifid” bacteria were, on the contrary, abundant
in healthy children [23]. He suggested that these bacteria could be
administered to patients with diarrhea to help restore a healthy gut
flora.
Probiotics, however, have gained momentum in recent past after
almost a century, with considerable growth in functional food market.
The concept of probiotic food came from the fact that bacteria
are normal inhabitants of humans (as well as the bodies of upper
animals and insects) including the gastrointestinal tract, where more
than 400 bacterial species are found [24]: half of the wet weight of
colonic material is due to bacterial cells whose numbers exceed by
10-fold the number of tissue cells forming the human body. Normally
the stomach contains few bacteria (103colony forming units per
mL of gastric juice) whereas the bacterial concentration increases
throughout the gut resulting in a final concentration in the colon of
1012 bacteria/g. Bacterial colonization of the gut begins at birth, as
newborns are maintained in a sterile status until the delivery begins,
and continues throughout life, with notable age-specific changes [25].
Bacteria, forming the so-called resident intestinal microflora, do not
normally have any acute adverse effects and some of them have been
shown to be necessary for maintaining the wellbeing of their host.
The term probiotic was introduced in 1953 by the German
bacteriologist Werner Kollath to mean “active substances essential for
a healthy life” [26]. In the last 30 years or so, however, research in the
probiotic area has progressed considerably and significant advances
have been made in the selection and characterization of specific
probiotic cultures and substantiation of health claims relating to their
consumption. For use in foods, the probiotic microorganisms should
not only be capable of surviving passage through the digestive tract
but also have the capability to proliferate in the gut. This means they
must be resistant to gastric juices and be able to grow in the presence
of bile under conditions in the intestines, or be consumed in a food
vehicle that allows them to survive passage through the stomach and
exposure to bile. The most commonly used probiotics are lactobacilli and bifido bacteria strains [27-28], but not exclusively, as other
micro-organisms have also been used as probiotics, including the
yeast Saccharomyces boulardii.
Probiotic microorganisms are generally LAB belonging to
the species Lactobacillus acidophilus, L. gasseri, L. helveticus, L.
johnsonii, L. (para)casei, L. reuteri, L. plantarum, L. rhamnosus,
and L. fermentum, while members of the genus Bifidobacterium are
also used, e.g., Bifidobacterium bifidum, B. longum, B. animalis, and
B. breve [29-31]. On the basis of the currently available literature,
probiotics can balance intestinal microbiota, and thereby regulate
proper intestinal function and be effective in the prevention or
treatment of several gastrointestinal disorders such as infectious
diarrhea, antibiotic-related diarrhea, irritable bowel syndrome or
Crohn’s disease [32]. Other examples of health benefits promoted
by probiotics supplied via dairy products are immunomodulatory
effects (L. casei CRL431), reduction of serum cholesterol level (L.
reuteri NCIMB 30242) and antihypertensive effects (L. plantarum
TENSIATM) [33-35]. Of late, probiotics seems to be quite helpful in
the management of rheumatoid arthritis.
Some foods naturally contain probiotics, while others have
probiotics added during preparation. Foods containing probiotics
include:
Live yoghurt
Live yoghurt drinks
Fermented and unfermented milk
Miso and tempeh, which are made from fermented soya beans
Some juices and soya drinks
Probiotics are available as supplements also.
Importance of Probiotics in pregnancy and otherwise:
Bifidobacteria are the most important constituent of the dominant
active flora [36]. Lactobacilli are part of the sub-dominant flora and
are under control by the dominant flora. Dietary and environmental
changes constitute the transient flora, which is exogenous and does
not colonize the GI tract.Since probiotics normally exist in our digestive system, their
intake is generally considered safe. But effectiveness in treating
specific symptoms or conditions is strain specific. Before starting a
regimen of probiotics, it is wise to look for the right strain in that
particular health condition.
GastroIntestinal Symptoms:
Constipation:
Constipation is one of the most common conditions that can
cause discomfort in pregnancy. Hormones of pregnancy often result
in relaxation of smooth muscle in the gastrointestinal (GI) tract,
therefore, dietary manipulation that includes increasing fibre and
fluids can help reduce constipation [37]. Over years, several studies
have found that yogurt can treat constipation, of these few studies
have been done in pregnant population. In one such randomized
controlled trial of 60 women, researchers found that 300 g of probiotic-enriched yogurt (Bifidobacterium and Lactobacillus 4.8 ×
1010 [CFU]) per day alleviated constipation better than conventional
yogurt among pregnant women [38].Few researchers have recommended including probiotic foods
such as fermented dairy products like yogurt and kefir every day over
probiotic supplements. They also mention the need for prebiotics in
the form of fibre, recommending at least 30 g of fibre each day from
whole foods such as whole grains, fruits and vegetables (including
legumes), and nuts and seeds in the diet during pregnancy.
The proposed mechanisms by which probiotics can help in
constipation are:
First, probiotics modify the gastrointestinal microbiota, which is
known to be altered in constipation [39-40].
Second, probiotic metabolites may alter gut function, including
sensation [41-42] and motility [43-44].
Third, some probiotics increase the production of lactate and
short-chain fatty acids, reducing luminal pH, which some researchers
have proposed will enhance colonic peristalsis and shorten whole gut
transit time (GTT) [45-46].
A meta-analysis by Dimidi et al [47] indicated that overall
probiotics positively affected and significantly improved the cardinal
symptoms of constipation such as bloating, sensation of incomplete
evacuation, occurrence of hard stools, ease of stool expulsion. There
was a decrease in GTT by half a day. This latter finding was consistent
with a meta-analysis by Miller et al [48] who had earlier shown that
gut transit time is significantly decreased with probiotics.
B.lactis plays a significant role in increasing the stool frequency
and improving stool consistency [49]. Normal stool frequency ranges
from 3 to 21 bowel movements per week [50-51] and an increase of
1.3 bowel movements per week through probiotic consumption could
normalize bowel frequency in adults with functional constipation.
Acute diarrhea:
Probiotics can potentially provide an important means to reduce
the problems associated with acute diarrhea, which is a major health
problem globally, especially among children. The strongest evidence of
a beneficial effect of defined strains of probiotics had been established
using Lactobacillus rhamnosus GG and Bifidobacterium lactis BB-
12 for prevention [52-53] and treatment [54-58] of acute diarrhea
mainly caused by rotaviruses in children. A systematic review that
included 12 randomized, controlled trials in the Cochrane database
(the majority from affluent countries) concluded that probiotics
reduced the mean duration of acute diarrhea in children by 29.2
hours in a fixed-effects model and by 30.48 hours in a random-effects
model [59]. Two meta-analyses that evaluated similar studies found
statistically significant but modest reductions of diarrhea duration
[60]. Two RCTs evaluated probiotics for children with persistent
diarrhea and reported dramatic reductions in diarrhea duration—4.8
and 3.9 days in Argentina [61] and India [62] respectively. Two
trials evaluated probiotics for diarrhea prevention; children in Peru
had 13% fewer diarrheal episodes after 15 months of Lactobacillus
rhamnosus [63], whereas diarrhea frequency was reduced by 14% among children in India who received daily doses of Lactobacillus
casei for 12 weeks, with a 12-week follow-up period [64].In the prevention of antibiotic-associated diarrhea (AAD),
meta-analyses of published results of RCTs provide evidence for
efficacy of a number of probiotic strains, such as S. boulardii [65-69].
Approximately one in seven cases of AAD was prevented by the use
of a probiotic [65]. According to another review, administration of
lactobacilli reduces AAD in adults, but not in children [66].
Diarrhea occurs in up to 34% of pregnant women, and its’ causes
in pregnancy mirror those of the non pregnant state, with the most
common being infectious agents (e.g, Salmonella, Shigella, and
Campylobacter species; Escherichia coli; protozoans; viruses). Food
poisoning, medications, and irritable bowel syndrome are other
common causes. Exacerbations of inflammatory bowel disease can
also occur in pregnancy. Gastroenteritis symptoms, when severe, can
cause dehydration and even preterm labour in cases that go untreated.
Therefore, addition of probiotics would definitely make a difference.
Activity against Helicobacterpylori:
Another development for probiotic applications is activity against
Helicobacterpylori (H. pylori), a Gram negative pathogen responsible
for type B gastritis, peptic ulcers and gastric cancer. Epidemiological
studies show a close association between the prevalence of H.
pylori and dyspeptic symptoms. Data from various studies suggests
that most of the Lactobacillus and Bifidobacterium strains possess
properties of acid tolerance and antimicrobial activity [70-72].A study by Chen et al reported that probiotics, L. rhamnosus
(GMNL-74) and L. acidophilus (GMNL-185), possess potent activity
to inhibit H. pylori adhesion to epithelia, thereby inhibiting the release
of inflammatory cytokines, and alleviating gastric inflammation [73].
The results are consistent with study done by Martinez et al [74].
A persistent stomach infection with H. pylori induces secretion
of proinflammatory cytokines, including IL-1β, IL-6, IL-8, and
TNF-α, which are closely linked to MALT-lymphoma and gastric
adenocarcinoma [75]. In addition, H. pylori infection alters gastric
microbiota, leading to dysbiosis that favours H. pylori colonization
and development of gastric cancer [76].
It has been further reported that probiotics exhibit anti-obesity
effects by lowering serum cholesterol [77-78]. High serum cholesterol
levels in humans leads to an increased risk of being infected with H.
pylori and induction of pathogenesis [79-80].
Regulation of blood sugar levels in pregnancy:
Gestational diabetes mellitus (GDM) is defined as carbohydrate
intolerance first diagnosed during pregnancy [81]. Maternal
pregnancy complications in GDM include preeclampsia and
instrumental or operative delivery. Fetal complications include
macrosomia, polyhydramnios, preterm birth, shoulder dystocia and
neonatal complications of admission to neonatal ICU, respiratory
distress, hypoglycaemia, and jaundice. Both women with GDM and
their infants are at increased risk of diabetes mellitus and metabolic
dysfunction later in life [82-83]. A study examining probiotics in
pregnancy suggested a benefit in reducing the incidence of gestational diabetes [84]. Supplementation with probiotics has been shown to
improve glycaemic control in men and women with type 2 diabetes
[85-86]. The gut microbiome is thought to influence obesity and type
2 diabetes through modification of energy extraction, inflammation,
hunger and satiety, as well as lipid and glucose metabolism [87-89].
Probiotics may regulate glucose metabolism and metabolic syndrome
[90-92], and the regulation of glucose metabolism is associated
with improvement in type 2 diabetes and hyperglycemia. Probiotic
supplementation during pregnancy may help maintain the density
of the intestinal flora, thereby reducing the metabolic imbalance in
pregnant women [93-94]. They are helpful in preventing worsening
of insulin resistance in late pregnancy [95].A recent Cochrane review studying the role of probiotics in GDM
involving 256 women showed a 60% decrease in the rate of diagnosis
of gestational diabetes mellitus in women taking probiotics from
early pregnancy [96]. A systematic review and meta‐analysis looking
at the effect of treatment of GDM on pregnancy outcomes showed
that treatment significantly reduced the risks of fetal macrosomia,
large‐for‐gestational‐age births, shoulder dystocia and gestational
hypertension, as well as a tendency to reduction of perinatal/neonatal
mortality and birth trauma [97].
In a randomized controlled trial, published in January 2017,
subjects either took Lactobacillus rhamnosus HN001 or a placebo.
Those who took the probiotic had a significantly lower incidence
of GDM than those who didn’t (2.1% vs 6.5%) [98]. A previously
published randomized controlled trial found no protection against
gestational diabetes with the use of Lactobacillus salivarius UCC118,
perhaps indicating the difference in efficacy of the strain [99]. Other
factors could be dosage, duration of treatment, timing of delivery
(early vs later in pregnancy), or other genetic or environmental
differences between the study groups.
A very recent study by Callaway et al [100] studied the role of
probiotics in the prevention of GDM in overweight and obese
women, in a total of 411 participants fulfilling the criteria. Probiotics
containing strains Lactobacillus rhamnosus and Bifidobacterium
animalis subspecies lactis were administered from the second
trimester in these women. Assessment by OGTT (oral glucose
tolerance test) was done at 28 weeks’ gestation. The probiotics used
in this study did not prevent GDM in overweight and obese pregnant
women.
The diet therapy (including specific fat and fibre intake
recommendations) was given to all patients enrolled. Diet together
with probiotic group had a significantly reduced rate of gestational
diabetes mellitus when compared to controls in a study conducted
by Baral M including 256 women [101]. Probiotic treatment included
Lactobacillus rhamnosus GG and Bifidobacterium lactis. Each
probiotic was taken at 10 billion CFU per day. Specific bacterial ratios
will either encourage or discourage obesity later in life and even predict
obesity development [102]. High numbers of bifidobacteria and low
numbers of Staphylococcus aureus in infancy can help protect against
overweight and obesity in children, possibly revealing a key reason
that breastfed infants enjoy a healthier metabolic outcome.
Probiotics help modulate the immune system and therefore inflammation [103]. Obesity can lead to a state of low-grade systemic
inflammation, possibly explaining the increased incidence of asthma
in obese patients [104]. Since obesity and inflammation are related, it
can be postulated that the probiotic control of inflammation plays a
role in obesity prevention.
Probiotics use in preventing urogenital infections:
The microbial species that inhabit the vaginal tract play an
important role in the maintenance of health, and prevention of
infection. Over 50 microbial species have been recovered from the
vaginal tract [105-107]. These species do not exist independently,
and studies in vitro and in humans have shown that a multispecies
microbiota, usually associated with bacterial vaginosis (BV), are
present in dense biofilms [108-111], while a lactobacilli dominant
microbiota can be sparsely distributed on the epithelium [108,109,112]. Despite the close proximity of the vagina to the anus, the
diversity of microbes present in the vagina is much lower than in the
gut. The reason for this lower diversity is still unclear. Some species
found in the gut, such as E. coli and Streptococcus, can also be found
in the vagina, indicating that proper receptors, nutrients, and oxygen
tension, are present for them to grow.Factors such as hormonal changes (particularly estrogen), vaginal
pH, and glycogen content can all affect the ability of lactobacilli to
adhere to epithelial cells and colonize the vagina [113]. The menstrual
cycle can also cause changes in the vaginal microbiota, with high
concentrations of estrogen increasing adherence of lactobacilli to
vaginal epithelial cells [114]. With the decrease in estrogen levels,
there is also a decrease in lactobacilli present in the vaginal tract as
seen in postmenopausal women [109,115-117].
While a vaginal tract dominated by lactobacilli appears to protect
the host against some vaginal infections, it does not fully prevent
colonization by other species. Pathogens are still able to coexist with
these commensal organisms, as shown by Burton and Reid [118],
where G. vaginalis, a pathogen associated with BV, was detected in
a vaginal sample which also contained a species of Lactobacillus.
Interestingly, G.vaginalis was displaced beyond detectable limits
for 21 days, following a single intravaginal instillation of probiotic
lactobacilli [115].
Pathogenic organisms are able to infect the vagina, with BV, yeast
vaginitis, and UTI (urinary tract infections) causing an estimated
one billion or more cases per year [119-122]. Lactobacilli are often
found in patients with yeast vaginitis, therefore, the induction of
infection does not appear to require the yeast displacing or killing
off the lactobacilli. Urinary tract infections occur when pathogenic
bacteria ascend from the vagina and replicate on, and sometimes
within, the bladder urothelium [119,123.124]. In women with no
history of UTI, vagina and perineum are most commonly colonized
by lactobacilli [125], while in women with recurrent UTI there is an
inverse association between lactobacilli and E. coli [126], suggesting
that lactobacilli play a role in preventing infection.
The most common urogenital disorder in women of reproductive
age is BV. Aerobic vaginitis has also been described in which the
vagina is colonized by organisms such as E. coli and enterococci [127]. During pregnancy, BV can increase the risk of preterm
labour and low birth weight [128-129]. Other problems associated
with BV include pelvic inflammatory disease, UTI, and increased
susceptibility to sexually transmitted diseases, including HIV [130-133].The organisms associated with BV form dense biofilms on the
vaginal epithelium, and these are associated with increased resistance
to lactobacilli-produced lactic acid and hydrogen peroxide (H2O2)
which are normally antagonistic to planktonic organisms [126].
The bio films are also able to induce host expression of certain
inflammatory factors, such as IL-1 and IL-8 [135]. It is not currently
known whether the production of H2O2 by lactobacilli has a clinically
protective role against BV.
As antimicrobial treatment of urogenital infections is not always
effective, and problems remain due to bacterial and yeast resistance,
recurrent infections [136-137], as well as side effects, it is no surprise
that alternative remedies are of interest to patients and their caregivers.
It is assumed that recurrences are due to antimicrobials failing to
eradicate the pathogens, perhaps because of biofilm resistance, or that
the virulent organisms come back from their source (the person’s gut,
or a sex partner) and attack a host whose defenses are suboptimal.
Young girls who suffer from UTI are more likely to have repeated
episodes in adulthood, and overall many UTI, BV, and yeast vaginitis
patients will have a recurrence [138-139].
Recurrent infection may also be due to the elimination of the
commensal organisms in the vagina by the antimicrobial, thereby
increasing susceptibility to recolonization by pathogens [140-141]. This is one of the main reasons for considering the use of
probiotics, to replenish the commensal microbes as a way to lower
the risk of reinfection. The concept of delivering lactobacilli orally
to repopulate the vagina was first reported in 2001 [142], and based
upon the question “If urogenital pathogens can do this, why cannot
lactobacilli”? The organisms were delivered in a milk base and shown
to be recovered from the rectum [143]; therefore supporting the
concept that ingested strains could pass through the intestine, reach
the rectum, and potentially ascend to the vagina. This was confirmed
independently by others [144].
The mechanisms whereby lactobacilli function as anti infective
defenses are still not fully understood. As discussed above, this may
involve production of antimicrobial factors [145], and maintenance
of a vaginal pH of ≤4.5. It could also be due to bio surfactants which
alter the surrounding surface tension and reduce the ability of a
wide range of pathogens to adhere [146-147]. This might explain the
relatively sparse coverage of epithelial cells noted in healthy women
[112]. In addition, lactobacilli have been shown to bind (coaggregate)
some pathogens and this may be a means to block their adhesion,
kill them through production of antimicrobials, and prevent their
spread to other areas of the vagina and bladder [108]. Among 10
strains of lactobacilli being evaluated for use in a probiotics tablet,
Mastromarino et al. [148] found, in vitro, that Lactobacillus gasseri
335 and Lactobacillus salivarius FV2 were able to coaggregate with
G.vaginalis. When these strains of lactobacilli were combined with
Lactobacillus brevis CD2 in a vaginal tablet, adhesion of G. vaginalis
was reduced by 57.7%, and 60.8% of adherent cells were displaced.
Boris et al. found that the adherent properties of G. vaginalis were similarly affected by Lactobacillus acidophilus [149]. It has been
known for some time that Lactobacillus produce bacteriocins that can
inhibit the growth of pathogens, including some associated with BV,
such as G. vaginalis [150]. Only relatively recently has a study shown
in animals that bacteriocin production might have an effect in vivo.
Neri et al. [140-151] studied 84 women in the first trimester of pregnancy
to observe the effects of probiotic-containing yoghurt on BV. The
subjects were randomized to one of three treatment groups: inserting
a tampon containing 5% acetic acid, a 10 to 15 mL vaginal douche
containing > 1.0 × 108 colony-forming units/mL of L. acidophilus, or
no treatment. Both active treatments were administered twice a day
for one week. Amsel criteria were absent in 88%, 38%, and 15% of
subjects who received intra vaginal lactobacilli, acetic acid tampons,
and placebo, respectively, after 30 days. There was a significant
difference in the cure rate between probiotic and control groups, and
lactobacilli and acetic acid groups.
The efficacy of combining probiotics or placebo with oral
metronidazole was assessed in 125 women aged 18 to 44 [152]. Oral
metronidazole was administered at 500 mg twice daily to all patients
for 7 days, and they were randomized to receive twice-daily oral
capsules containing either a placebo or L. rhamnosus GR-1 and L.
reuteri RC-14 at 1.0×109 colony-forming units for a total treatment
duration of 30 days. At the end of 30 days, BV was considered absent
if the patient had a negative sialidase test and a Nugent score of <3.
This was the case in 40% of placebo and 88% of probiotic subjects.
If an intermediate Nugent score was regarded as “cure of BV”, the
cure rate was 100% with metronidazole and probiotics versus 70%
with metronidazole and placebo. This study is important as it implies
that probiotics can augment the effects of antibiotics in treatment of
disease.
Group B Streptococcus (GBS) is the leading cause of neonatal
morbidity and mortality [153]. It may be passed from the mother
colonized in the genital tract by GBS to their baby during vaginal
birth, or by being spread to the amniotic fluid. This vertical
transmission can lead to early-onset GBS disease of the newborn
(EOGBSD), which manifests in the first 7 days of life, and can be
fatal. While approximately 10 to 30% of pregnant people harbour
GBS in the vagina or rectum, the incidence of neonatal GBS disease
is 1 to 2 infants per 1000 births. The use of intrapartum antibiotics
to treat colonized individuals with or without risk factors has led to
a 70% decline in the incidence of early-onset GBS sepsis in the past
decade [154]. Despite this impressive decline, antibiotic resistance
has become a major public health concern. Association between
intrapartum antibiotic use and ampicillin resistance in E. coli isolated
from neonates has previously been documented [155]. Furthermore,
while GBS has remained sensitive to penicillin, 20% are resistant to
erythromycin and clindamycin, which are alternate drugs for patients
allergic to penicillin. Alternative approaches are therefore needed to
reduce the risk of GBS infection.
Earlier researches have been done which include the use of
Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14
(Lactobacillus fermentum RC-14) in the colonization status of
GBS in pregnant people [152]. Lactobacilli are part of normal gut
and vaginal flora and have been widely used as probiotics to treat various conditions. In particular, these two strains have shown to be
beneficial in the treatment of urinary tract infections and bacterial
vaginosis [156]. As lactobacilli are part of the human gut flora and
have low pathogenicity, no adverse reaction to lactobacilli is usually
anticipated, though some patients may experience flatulence.
Bacteria of the lactobacillus sp. are the most common in probiotic
capsule preparations. The natural vaginal flora play an important
role in dislodging and inhibiting pathogens. The rationale for the use
of probiotics then, is the return of the vaginal flora to their healthy,
natural state [157]. Indeed, probiotics have been shown to alter the
vaginal flora being an effective supplement in the treatment and cure
of bacterial vaginosis, and vulvovaginal yeast infections. Probiotic
capsules are considered safe for use in pregnancy [158].
The ongoing researches are using 2 capsules (together) of oral
Lactobacillus GR-1 and RC-14 once daily for 12 weeks from 24
weeks of pregnancy until their GBS swab test is taken at 35-37 weeks
of pregnancy. The studies tend to provide evidence that probiotic
prophylaxis is an effective, low-risk strategy that can be offered to
patients to reduce their risk of GBS colonization and thus, the need for
intrapartum antibiotic prophylaxis and the risks and inconvenience
associated with it.
The raised estrogen and growth hormone during pregnancy may
increase the activity of HPV molecule and human papilloma virus
(HPV) infection. Short-time HPV persistence has been associated
with higher risk for cervical intra-epithelial neoplasia and a higher
risk of High-Grade Squamous Intraepithelial Lesion (HSIL). Clinical
data showed GR-1 and RC-14 can improve the cervical malignancy
diagnostics quality for non-pregnant women. The influence of oral
probiotics on postpartum diagnosis of cervical pathology remains
unknown.
In a study by Hanson et al [159], participants in the probiotic
group reported no adverse events or minor side effects; one half
reported improved gastrointestinal symptoms. Although two women
in each group had positive qualitative prenatal GBS cultures at 36
weeks, the probiotic group participants had lower quantitative GBS
colony counts. The eight GBS negative averaged 90% probiotic
adherence compared with two GBS positive women who averaged
68%. Yogurt ingestion was inversely related to GBS colonization.
It was concluded that prenatal probiotic therapy has the potential
to reduce GBS colonization which appears to be linked to its daily
adherence.
Reduction in the incidence of Preeclampsia and Preterm delivery:
Timely intake of probiotics during pregnancy might help lower
the risks of preeclampsia and premature birth, suggests observational
research. Probiotics may have an anti-inflammatory effect on lipopoly
saccharide inflammatory response in human placental trophoblast
cells [160,161]. The anti-inflammatory effect of orally ingested
probiotics has also been shown in vivo [162-163].In a study by Nordqvist et al [164], it was revealed that probiotic
milk intake during late pregnancy (but not before or in early
pregnancy) was associated with reduced risk of preeclampsia, and that intake during early pregnancy (but not before or in late pregnancy)
was associated with reduced risk of preterm delivery.
Two previous studies in the Norwegian Mother and Child Cohort
Study (MoBa) showed associations between intake of milk containing
probiotics during the first half of pregnancy and reduced risk of
preeclampsia and spontaneous preterm delivery [165-166].
The gastrointestinal tract represents the largest immune
interface with the environment, probiotics are known to modulate
gastrointestinal health through suppression of pathogenic bacteria as
stated earlier. Steinborn et al showed that preeclampsia and preterm
delivery are characterised by changes in the composition of regulatory
T cell, decreasing their suppressive activity [167]. In vitro studies
have shown that probiotics (Lactobacillus rhamnosus GR-1 and
LGG) may have an anti-inflammatory effect on LPS inflammatory
response in human placental trophoblast cells [160,161], potentially
a key cell type in preeclampsia. In another large observational study
by Brantsaeter et al, it was concluded that there is an independent
protective association between intake of probiotic milk products
and preeclampsia, especially severe preeclampsia, suggesting
that probiotics might specifically “target” and modify the type of
inflammation underlying severe preeclampsia [165].
In a randomised, double-blind, placebo-controlled trial which
was completed by 29 mother-infant pairs, it was shown that bacterial
DNA was detected in all placental samples. Microbial DNA in
amniotic fluid and placenta was associated with changes in TLRrelated
gene expression in the fetal intestine. Maternal probiotic
supplementation (109 Bifidobacterium lactis alone or in combination
with 109Lactobacillus rhamnosus GG) significantly modulated the
expression of TLR-related genes both in the placenta and in the fetal
gut. These findings suggest a link between the maternal gut and that of
the developing fetus and that microbial contact at the feto-placental
interface may be considered a physiological phenomenon [168].
Probiotics in allergic disease (Controlling inflammation, Stabilizing immune systems):
Probiotics appear not only to modulate T-cells and cytokine
profile, but also to help accelerate recovery of barrier function. There
is increasing evidence that disturbances in gut microbial composition
play a role in the pathophysiology of immune-mediated disorders,
such as allergic disease [169]. Gut microbiota are key players in the
early development of both local immune maturation and systemic
immune programming.Gut microbiota confer specific immune-protective effects that are
probably mediated through complex pathways within (and potentially
even beyond) the gut-associated lymphoid tissue (GALT), the largest immune “organ” in humans. These effects include altered local
immunoglobulin A (IgA) production and induction of tolerogenic
dendritic cells and regulatory T cell populations, with production
of immunomodulatory cytokines, such as interleukin (IL) 10 and
transforming growth factor (TGF) beta [170]. These mechanisms
appear to collectively inhibit local inflammation, improve gut barrier
mechanisms, and consequently reduce the risk of inappropriate
systemic immune responses.
Early studies reported that low levels of Bifidobacterium and
early colonization with potentially pathogenic bacteria, such as
Clostridioides (formerly Clostridium) difficile [171,172] and
Staphylococcus aureus [171], were more prevalent in children who
subsequently developed allergy. Consequently, it has been suggested
that a high gut microbial diversity might be more important than
the absence or presence of specific genera or species in the context
of immune system maturation and subsequent development of
immune-mediated disorders. This view is supported by prospective
studies that demonstrated reduced gut microbial diversity early in life
in infants who later developed allergic manifestations [173-175].
In a double-blind, randomized, placebo-controlled trial, L.
rhamnosus GG was given to pregnant women for four weeks prior to
delivery, then to newborns at high risk of allergy for six months with
the result that there was a significant reduction in early atopic disease
[176].The precise mechanisms have not been elucidated, but the
premise is based upon the ability of lactobacilli to reverse increased
intestinal permeability, enhance gut-specific IgA responses, promote
gut barrier function through restoration of normal microbes,
and enhance transforming growth factor beta and interleukin 10
production as well as cytokines that promote production of IgE
antibodies [176,177].
Production of folate by intestinal bacteria, especially the Bifidobacteria:
Folates represent an essential nutrition component in the
human diet, being involved in many metabolic pathways. The daily
recommended intake of folate is 400 μg/day for adults [178,179]
(Table 2).The gut microbiota has been recognized as a source of vitamins.
The microbiota of the human colon is known to produce vitamin K
(menaquinones) and most of the water-soluble vitamins of group
B, including biotin, nicotinic acid, folates, riboflavin, thiamine,
pyridoxine, panthotenic acid, and cobalamin [184]. Unlike dietary
vitamins, which are mainly absorbed in the proximal part of the small
intestine, the uptake of microbial vitamins predominantly occurs
in the colon [185]. Colonocytes appear to be able to absorb biotin,
thiamin, folates, riboflavin, panthotenic acid, and menaquinones, indicating that the microbiota-produced vitamins may contribute to
the systemic vitamin levels and especially to the homeostasis of the
vitamins in the localized epithelial cells [185,186].
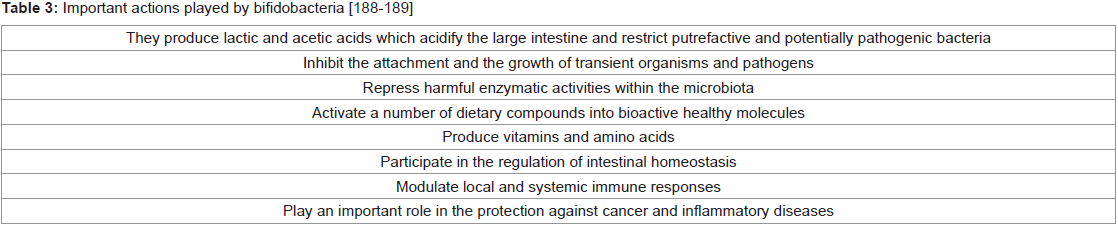
Bifidobacteria - How important they are!:
Bifidobacterium are one of the most important health-promoting
groups of the colonic microbiota and one of the most important
microorganisms to be used as probiotics [187]. Bifidobacterium
is a genus of high G + C Gram-positive eubacteria within the
phylum of Actinobacteria (Table 3). Among nearly fifty species of
bifidobacteria recognized so far [190], the most represented in the
gastrointestinal tract of human adults or infants, are Bifidobacterium
pseudocatenulatum, B. catenulatum, B. adolescentis, B. longum, B.
infantis, B. breve, B. angulatum and B. dentium [191]. In a human
trial, the administration of the few strains of bifidobacteria resulted
in a significant increase of folate concentration in feces. Even though
the effect on plasmatic levels needs further investigation, folateproducing
bifidobacteria provide a complementary endogenous
source of the vitamin and may contribute to prevent folate deficiency,
which is often associated with premalignant changes in the colonic
epithelia.Prevalence of folate deficiency - especially among women of
childbearing age- is a growing concern and thereby folate fortification
programs should be implemented [192]. Foods can be naturally
fortified with folate synthesized by LAB and bifidobacteria during
manufacture of fermented foods [193,194].
B. adolescentis and B. dentium are capable of de novo folate
production, while B. longum needs to be provided with pABA (paraaminobenzoic
acid), and B. animalis requires folate. Several strains of
bifidobacteria have been screened for their ability to produce folate
in low-folate or folate-free media. Twenty-four strains of B. bifidum,
B. infantis, B. breve, B. longum, and B. adolescentis were cultured
in a low - folate semi synthetic medium and significant differences
in vitamin accumulation were found among the species tested [195].
All B. bifidum and B. infantis strains were classified as high folate
accumulators, while B. breve, B. longum, and B. adolescentis produced
lower amounts of the vitamin. For all the strains, extracellular folate
accounted for most of the accumulated vitamin [196]. In other
studies, the highest folate accumulation in reconstituted skim-milk
was obtained after incubation with B. breve and B. infantis or B.
longum strains [193].
Ability to produce the vitamin in the folate-free medium
was found only in 17 strains belonging to nine different species
(B. adolescentis, B. breve, B. pseudocatenulatum, B. animalis, B. bifidum, B. catenulatum, B. dentium, B. infantis, and B. longum)
[195]. The highest extracellular folate levels (between 41 and 82 ng
mL−1) were produced by four strains of B. adolescentis and two of B.
pseudocatenulatum. Only one out of 15 B. longum strains grew in
folate free-medium. The highest extracellular folate levels (between 41
and 82 ng mL−1) were produced by four strains of B. adolescentis and
two of B. pseudocatenulatum. Only one out of 15 B. longum strains
grew in folate free-medium. These same strains of B. adolescentis and
B. pseudocatenulatum, when given to 23 healthy volunteers in a pilot
human study, significantly increased folate concentration in the feces
of the subjects [197]. These results corroborate the assumption that
the increase of folate levels was markedly due to the effective growth of
the folate-producing bifidobacteria. The results from various studies
support the evidence that folate-producing probiotic strains may
represent an endogenous source of vitamin, preventing its’ deficiency
in the colon. Localized folate production in the large intestine may
provide the proliferating enterocytes with this essential vitamin with
potential effects in reducing colonic carcinogenesis [198].
The strains Streptococcus thermophilus CRL803/CRL415 and L.
bulgaricus CRL871 were reported to be suitable for the elaboration
of yogurt naturally bio-enriched in this vitamin [199]. High folate
concentration (up to 150 μg/l) can be reached in yogurt as a result
of the ability of S. thermophilus to produce this vitamin [200].
Among bifidobacteria, B. catenulatum ATCC 27539 was shown to
produce high levels of folate in vitro [201], and B. lactis CSCC5127, B.
infantis CSCC5187, and B. breve CSCC5181 strains increased folate
concentration during fermentation of reconstituted skim milk [202].
Similarly, L. amylovorus CRL887 can be used for natural folate bioenrichment
of fermented milk [203].
Prevention of depression and anxiety postpartum:
Depression and anxiety in pregnancy and after birth affects 10-15
per cent of women, although many are not recognised or treated. There
is mounting evidence from animal studies that, the ‘microbiomegut-
brain axis’ - the biochemical signalling that takes place between
the gastrointestinal tract and the central nervous system - may be
important for mental health. Maternal depression can produce longlasting
effects on children’s cognitive, social-emotional and health
outcomes [204,205]. Anxiety often coexists with depression. Despite
this, most women with post natal depression are either not recognised
as being depressed, are unable to access psychological therapy or
are reluctant to take antidepressant medication in pregnancy or
while breastfeeding [206]. Furthermore it takes several weeks for
the therapeutic effect of antidepressants to appear and there is a 15–
30% discontinuation rate [207]. Furthermore, it has been suggested that fermented foods (prebiotics) alter dietary items before they are
ingested, resulting in phytochemical transformation into bioactive
chemicals which reduce oxidative stress and inflammation [208].In 2005 it was first suggested that probiotics might be an adjuvant
therapy for major depression [209]. The study suggests that in
depression and similar conditions where depression is a common
symptom, lactobacilli levels may be low owing to migration of
bacteria from the colon into the small intestine resulting in small
intestinal bacterial overgrowth (SIBO). In patients with MDD (major
depressive disorder), SIBO is likely to occur because it is often the
result of intestinal stasis or low stomach acid secretion. Patients with
depression are known to have low levels of stomach acid production
and intestinal stasis. Cytokines linked to depressive symptoms,
particularly interleukin1-beta (Il-1b) and tumor necrosis factor
alpha(TNFa), are capable of inhibiting gastric acid secretion. In
addition, physical inactivity, common to depression, is associated with
SIBO [209]. Others have also suggested that probiotic enhancement
of gut microbiota may improve mood outcomes [210].
The significance of small intestine bacterial overgrowth in cases
of depression can lead to functional abdominal complaints, it can
cause malabsorption of fat, carbohydrate, protein, B vitamins and
other micronutrients, in turn leading to weakening of host defence
against SIBO. Patients with depression are known to have low levels
of folic acid, vitamins B12, B6 and zinc [211-214]. Low levels of vitamin
B6 is associated with diminished conversion of alpha linolenic acid
into mood regulating eicosapentaenoic acid (EPA). Non-digestible
oligosaccharides can increase the availability of nutrients including
zinc, effects that are attributed to increased bifidobacterium. It is
interesting to note that treatment of SIBO has led to improvements
in depression, memory and concentration among CFS patients [215].
A recent systematic review identified 10 clinical trials of the effect
of probiotics on symptoms of depression [216]. Seven studies were in
healthy subjects, 2 in chronic fatigue syndrome and one in depression.
Three of 5 studies reported improved mood with probiotics, and 5
of 7 studies reported improvements in stress and anxiety. A recent
study that was published after these reviews reported that obese
women treated with a weight-reduction programme and probiotic
had reduced symptoms of depression compared with the comparison
group, but this effect was not seen in men [217]. There was no effect
on anxiety.
In a very recent, first double-blind RCT of probiotics that has
evaluated symptoms of depression and anxiety in the postpartum
period [206], a significantly lower prevalence of symptoms of
depression and anxiety was seen postpartum in women supplemented
with the probiotic HN001 during and after pregnancy than in those
given a placebo. Furthermore, the number of women reporting
clinically significant levels of anxiety on screening was significantly
lower in the probiotic group. In this study infant colic was associated
with higher depression and anxiety scores. There has been a suggestion
in the literature that probiotic supplementation may benefit maternal
mood by reducing infant colic. One study reported that direct
probiotic supplementation of infants reduced infant colic and this in
turn was associated with lower rates of maternal depression [218].
RCT of 40 people with major depressive disorder treated with a combination of three probiotics (Lactobacillus acidophilus,
Lactobacillus casei, and Bifidobacterium bifidum) or placebo, also
found a significant reduction in symptoms of depression on the Beck
Depression Inventory (BDI) in the treatment group [219].
Many Lactobacillus and Bifidobacterium strains have been
studied with respect to mental health and these genera seem to show
the most beneficial effects [220]. Maternal stress during pregnancy
can result in a reduction of both lactobacilli and bifidobacterium in
offspring, relative to controls [221].
There is also evidence from human studies indicating that stress
can negatively affect microflora [222,223]. Emotional stress can lead
to acute and long term reductions in lactobacilli and Bifidobacterium
[224]. Bifidobacterium appear to be extremely sensitive to emotional
stress. Restraint stress and excess physical demands can also lead to
decreases in lactobacilli and bifidobacterium in humans [221].
The gut contains over 100 million neurons; the GI tract is
ultimately a meeting place of nerves, microorganisms and immune
cells. Microorganisms are responsive to the host’s neuroendocrine
environment and, conversely, bacteria can influence the
neuroendocrine environment by the production of neurochemicals
such as gamma amino butyric acid (GABA), serotonin, and
various biologically active peptides. Animal studies indicate that GI
microorganisms can directly activate neural pathways, even in the
absence of an immune response [226,227]. Probiotic bacteria may
influence mood by their effect on cytokine production.
Preventing skin disease in newborn:
The intestinal microflora in atopic dermatitis has been shown to
contain significantly lower levels of bifidobacterium and higher levels
of staphylococcus. Percentages of bifidobacteria are significantly
lower in patients with severe atopy versus those with mild symptoms
[209]. Probiotics should be consumed by pregnant and lactating
women and their breastfed infants to prevent the development of
atopic dermatitis [228].A probiotic extract being developed from the human microbiome
could offer drug-free topical therapy for patients with atopic
dermatitis, and could protect against pathogenic biofilms [229]. The
extract has anti-inflammatory effects as well. Staphylococcus aureus
biofilms — both methicillin-resistant (MRSA) and methicillinsusceptible
(MSSA) — play an important role in patients with
moderate and severe atopic dermatitis. The topical extract can disrupt
these biofilms and has tremendous potential to not only decrease
infection risk, but to improve other aspects of the disease as well,
since the bacterial colonization likely contributes to inflammation
and skin barrier disruption. The effects are similar to dexamethasone.
In other clinical studies with infants allergic to cow’s milk,
atopic dermatitis was alleviated by ingestion of probiotic strains L.
rhamnosus GG and B. lactis BB-12 [230-232].
The development of the infant microbiome is a key area of study,
and its known that there are a variety of contributing factors and
situations that impact differences in microbial colonization among
infants. Differences in the microbiome have been linked to increased
allergy risk. Presently, there is support for the use of probiotics to prevent eczema. Since eczema is a precursor to a variety of atopic
conditions (eg, food allergies and asthma), and because it causes
a great deal of suffering, reducing or preventing it, is important.
About 10.7% of children younger than 18 have eczema, and about
37% of those with moderate to severe eczema go on to develop food
allergies, according to the American College of Allergy, Asthma and
Immunology [233,234].While researchers continue to learn about
the connection, controlling eczema may be one way to reduce the
development of food allergies.
Two separate systematic meta-analyses found a reduction in
eczema risk among the offspring of mothers who took probiotic
supplements during pregnancy [235,236]. However, these two
reviews had some limitations, so they were considered low quality
and were not according to the current research [237]. According to
the World Allergy Organization-McMaster University Guidelines
for Allergic Disease Prevention (GLAD-P): Probiotics, clinicians
should recommend probiotics to women at high risk of having an
allergic child, those who breast-feed infants who are at high risk, and
to infants who are predisposed to the development of allergy [238].
According to the guidelines, “High risk for allergy in a child is defined
as biological parent or sibling with existing or history of allergic
rhinitis, asthma, eczema, or food allergy.” In addition, the guidelines
say that although the quality of the evidence may be low, the possible
net effect is worth supplementation, since the risk of negative impact
is low.
Probiotics and Infant colic:
Babies who cry and fuss for more than 3 h daily have colic.
The condition generally starts at 3 weeks of age, occurs on more
than 3 days/week, and resolves after 3 months of age (hence the
“rule of threes” . The most common description of colic is intense,
“paroxysmal” crying that is markedly different from normal fussing
and crying. It can also occur as prolonged, unpredictable crying, and
the infant is restless and inconsolable. Crying may occur any time of
the day without obvious cause but is most common after the evening
feeding. The colicky episode is often accompanied by distention of
the abdomen and cold feet. Often the baby seems to feel better after
passing gas or a stool.The neonatal microbiota is highly different compared to the adult
one, since the first is characterized by rapid changes as mentioned
earlier [239]. At birth, the newborn is exposed to a set of bacteria
including staphylococci, enterobacteria, and enterococci that
immediately colonize the gastrointestinal tract. In the first days of
life, the gut is inhabited mainly by Bifidobacterium, Lactobacillus,
Clostridium, and Bacteroides. From one to five months of life, the
population of the gastrointestinal tract consists of Bifidobacteriales,
Lactobacillales, and Clostridiales. At one year of age, the microbiota
is similar to the adult one [240,241].
Infant colic previously was felt to be unresponsive to any
treatment. Microbial dysbiosis began to be linked to this condition
and was confirmed by several groups [242-244], and it was linked to
gut inflammation [245]. Therefore, colic might represent a condition
for which probiotic treatment would be useful. Several meta-analyses
have shown that the probiotic L. reuteri, isolated from a Peruvian mother’s breast milk, reduces crying time and irritability in this
condition [246-248].
Probiotics and Arthritis:
Very recently, dysbiosis have been included in the list of
triggers leading to rheumatoid arthritis (RA). Rheumatoid arthritis
is a systemic autoimmune disease characterized by autoantibody
formation leading to the chronic inflammation of multiple joints.
RA is also known to affect other internal organs, including the lungs,
heart, and kidneys [249].People with inflammatory arthritis have been shown to have
inflammation of the intestinal tract, which results in increased
intestinal permeability. This enables certain bacteria to cross
the intestinal barrier, get into the bloodstream and trigger an
inflammatory response. Probiotics may be able to help decrease
the inflammation associated with increased intestinal permeability.
Probiotics appear to have an impact on inflammation, reducing
common biomarkers of inflammation, including C-reactive protein.
They help to decrease the inflammation associated with increased
intestinal permeability.
In a recent study by Zamini et al [250], daily probiotic
capsules containing Lactobacillus acidophilus, Lactobacillus casei
and Bifidobacterium bifidum were given for 8 weeks. Probiotic
supplementation group resulted in improved Disease Activity Score of
28 joints (DAS-28). There was a significant decrease in serum insulin
levels, homeostatic model assessment-B cell function (HOMA-B) and
serum high-sensitivity C-reactive protein (hs-CRP) concentrations.
In a study by Mohammed et al [251], the efficacy of probiotics
as an adjuvant therapy for rheumatoid arthritis was studied. The
meta-analyses indicated that pro-inflammatory cytokine IL-6 was
significantly lower in the probiotics compared with the placebo group
but disease activity score could not find any difference.
A study by Chen et al. evaluated the gut microbiota profile in 40
patients with RA and 32 healthy controls. They found decreased gut
microbial diversity in RA compared to controls, which additionally
correlated with disease duration and with levels of serum rheumatoid
factor [252].
Alipour et al. showed that L. casei 01 supplementation decreased
serum high-sensitivity C-reactive protein (hs-CRP) levels, reduced
tender and swollen joint counts, and improved global health (GH)
score. A significant difference was also observed between the two
groups with respect to circulating levels of interleukin (IL)-10, IL-12,
and tumour necrosis factor (TNF)-α, in favour of the probiotic group
[253].
Possible negative effects of probiotics use:
Many probiotic products are used on the simple assumption that
probiotics can retain health and well being, and potentially reduce
long-term risk of diseases of the bowel, kidney, respiratory tract and
heart. Multicentre large randomised controlled trials are needed to
authenticate such an assumption before making it a regular practice.
Study by Tannock et al. [253] mentions an important observation that
the ingestion of probiotic strains has not led to measurable long-term
colonization and survival in the host. Invariably, the microorganisms are retained for days or weeks, but no longer. Thus, use of probiotics
likely confers more transient than long-term effects, and so continued
intake appears to be required. However, in newborn children where
a commensally flora has not yet been established, it is assumed that
probiotic microorganisms could become primary colonizers that
remain long-term, perhaps even for life.When ingested orally or used vaginally, probiotics are generally
considered safe and are well tolerated. One theoretical concern
associated with probiotics is the potential for these organisms to cause
systemic infections. Although rare, probiotic-related bacteremia and
fungemia have been reported [255]. It is estimated that the risk of
developing bacteremia from ingested Lactobacillus probiotics is less
than 1 per 1 million users [256], and the risk of developing fungemia
from Saccharomyces boulardii estimated at 1 per 5.6 million users,
and is estimated to be lower in healthy individuals [257]. There have
been no reports of bifidobacterium sepsis associated with the use
of probiotics in healthy individuals [258]. Risk factors for systemic
infections include immune suppression, critical illness, central
venous catheters, and impairment of the intestinal epithelial barrier.
Probiotics administered orally to combat urogenital infections
are not systemically absorbed but rather get to the site of action by
passage through the gastrointestinal system and ascending into the
vagina [142].
Annually, over one billion doses of probiotics are administered
worldwide, and those administered for urogenital health have
been well tolerated [115,152,256,259-261]. In addition, the mouth,
gastrointestinal tract, and female genitourinary tract are inhabited
by Lactobacillus [256]. Yet, endocarditis and bacteremia caused
by lactobacilli are extremely rare. Most cases occur in patients
with chronic diseases or debilitating conditions that provide direct
access to the bloodstream from a leaky gut. Only 1.7% of 241 cases
of bacteremia, endocarditis, and localized infections associated with
Lactobacillus that were investigated by Cannon et al. were considered
to have a possible link with heavy consumption of dairy products [262].
Only one case had a Lactobacillus isolate that was indistinguishable
from a probiotic strain. There was no connection between the species
of Lactobacillus isolated and the type of infection or mortality. A
recent study that directly instilled a six-strain bacterial product into
the intestine of patients with severe, potentially fatal pancreatitis
portrayed probiotics as being dangerous [262]. However, the product
had never been proven to be probiotic, it was administered as a drug
unlike 99.9% of probiotics, the randomization process led to patients
with multiorgan failure being given large doses of live bacteria, and
the authors failed to provide a rationale for the study in an appropriate
animal model. All this led to warranted adverse publicity for the field
of probiotics [263].
Probiotics do not appear to pose any safety concerns for pregnant
and lactating women. Systemic absorption is rare when probiotics are
used by healthy individuals [264].
Future Researches:
One of the studies to watch for is the Probiotics in Pregnancy
(PiP) Study, a multicentre, multinational study that has recruited
pregnant women to research the effect of L rhamnosus HN001 in early pregnancy through breast-feeding. Researchers expect administration
to reduce the rate of infant eczema and atopic sensitization at 12
months. Researchers also are studying the impact of supplementation
on GDM, bacterial vaginosis, and group B streptococcal vaginal
colonization before birth, and depression and anxiety postpartum
[265].Another study to keep an eye on is The Environmental
Determinants of Diabetes in the Young (TEDDY), also a multicentre,
multinational study. The TEDDY study is exploring the causes of
type 1 diabetes mellitus to understand what interventions could help
reduce the risk of it’s development. These researchers are exploring
the interaction between genes and environmental contributors.
The study is ongoing, but in 2015, researchers presented early data
that showed a reduction of 33% in autoimmunity, based on the
development of auto antibodies after infant supplementation with
probiotics starting in the first month of life. Although promising,
more research is needed to confirm these findings [266].
Probiotic foods are a safe way for pregnant women to introduce
and consume healthful microbes during pregnancy and may provide
other positive nutritional benefits. These may include foods such
as fermented sauerkraut (a source of fibre), and yogurt and kefir
(providing calcium and vitamin D). Pregnant patients should avoid
unpasteurized milk and juice products because of the risk of foodborne
illness. Pregnant women who fall into the high-risk category are good
candidates for probiotic supplements. In fact, regular consumption of
safe, whole, fermented, and probiotic foods may benefit all patients.
Finally, research on the benefits of probiotics is growing and dietitians
should continue to follow the research in this area to provide the best
evidence-based guidelines to use in practice.
References
10. Monash University. Dietary Fibre and natural prebiotics for gut health: FAQs. Monash University.
14. Vandenplas Y, De Greef E, Veereman G (2004) Prebiotics in infant formula. Gut Microbes 5: 681–687.
37. Cullen G, O'Donoghue D (2007) Constipation and pregnancy. Best Pract Res Clin Gastroenterol 21: 807.
93. Gregor MF, Hotamisligil GS (2011) Inflammatory mechanisms in obesity. Annu Rev Immunol 29: 415-445.
104. Shore SA (2008) Obesity and asthma: possible mechanisms. J Allergy Clin Immunol 121: 1087-1093.
184. Hill MJ (1997) Intestinal flora and endogenous vitamin synthesis. Eur J Cancer Prev 6: S43-S45.
187. Rossi M, Amaretti A, Raimondi S (2011) Folate Production by Probiotic Bacteria. Nutrients 3:118-134.
193. Lin MY, Young CM (2000) Folate levels in cultures of lactic acid bacteria. Int Dairy J 10: 409-413.
Citation
Tania SG. Probiotics in Pregnancy. Indian J Nutri. 2021;8(2): 231.