Review Article
Vitamin D deficiency in Children: An update on its Prevalence, Therapeutics and Knowledge gaps
Suchitra Surve1, Sanjay Chauhan1, Yeshwant Amdekar2 and Beena Joshi1*
1ICMR-National Institute of Research in reproductive Health, Mumbai, India
2B.J Wadia Hospital for Children, Mumbai, India
Corresponding author: Beena Joshi, Scientist ‘E’, Department of Operational Research, National Institute for Research in Reproductive Health (ICMR), Mumbai, Maharashtra, India; Tel: 91-22-24192043; Fax: 91-22-24139412; E-mail: bjoshithane@gmail.com
Citation: Surve S, Chauhan S, Amdekar Y, Joshi B. Vitamin D deficiency in Children: An update on its Prevalence, Therapeutics and Knowledge gaps. Indian J Nutri. 2017;4(3): 167.
Copyright © 2017 Surve S, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Indian Journal of Nutrition | ISSN: 2395-2326 | Volume: 4, Issue: 3
Submission:19/07/2017; Accepted: 14/08/2017; Published: 23/08/2017
Abstract
Background: Vitamin D Deficiency (VDD) is known to have adverse implications from conception throughout lifespan. Vitamin D is important in children mainly because of its profound effect on growth and development and more importantly bone health. Recent evidence suggests maintaining 25(OH)D levels above 20 ng/ml for maximizing health benefits. Despite ample sunshine, VDD is prevalent among 50-90% of Indian children. For understanding functional relevance of this widely prevalent VDD, a comprehensive overview of evidence in children is required. The aim of this article is to review studies related to VDD among children. This paper collates information from various epidemiological studies among children pertaining to prevalence, morbidities and therapeutics thus highlighting the current research gaps and challenges in Indian context.
Methods: Literature search with keywords focusing on Vitamin D deficiency, Vitamin D, child health, Pediatric, Hypovitaminosis D and Bone health was carried out through computerized databases- Medline, Embase, PubMed, Cochrane and Google.
Results: Data in pediatric age group is heterogeneous with limited evidence in under five age group. The VDD among children may be defined either on basis of a “population based reference value” or a “functional health based reference value”. Functional cut offs refer to the concentration of 25(OH)D at which PTH begins to increase and needs more validation in view of varied response among children. Determinants of circulatory levels of Vitamin D metabolites and evaluation of complete 25(OH)D statuses including Vitamin D Binding Protein (DBP) in children also needs further evidence.
Importantly, the use of vitamin D supplements among children should be done cautiously. The Consensus by Indian Academy of Pediatrics may be followed in this context until Indian specific evidence based guidelines are formulated.
Conclusion: There is a need to explore Vitamin D paradox with regards to cut offs and risk factors such as genetic, environmental and behavioral factors in Indian children.
Keywords:
Vitamin D deficiency; Vitamin D; Child health; Pediatric; Hypovitaminosis D; Bone health
Background
Vitamin D Deficiency (VDD) is known to have adverse implications right from conception throughout lifespan. It is important in children mainly because of its profound effect on growth and development. Since approximately 40%-60% of total skeletal mass at maturity is accumulated during childhood and adolescence, it has major implications on adult bone health. It regulates calcium and phosphorus balance for bone mineralization and remodeling [1]. Rickets is a severe form of VDD and manifests as bone deformities, bone pain and weakness. It however represents only tip of iceberg as apart from its conventionally understood actions on bone health and calcium homeostasis, Vitamin D is believed to have effect on body’s endocrine system, immune system, cardiovascular system, neuropsychological functioning and neuromuscular performance [2]. The active form of Vitamin D ie. 1,25(OH)2D is capable of regulating a wide variety of genes which regulates cell growth and differentiation. Vitamin D is therefore essential not only for skeletal growth, but also for improving immune status in children. Hence elucidating relevance and applicability of insufficient Vitamin D levels among children is of paramount importance..
Vitamin D metabolism and diagnostics
Nearly 90% of Vitamin D requirement (Vitamin D3) is met by adequate exposure of the skin to sunlight through the action of Ultraviolet B Radiations (UVR) and rest 10% is said to meet through diet [3]. The commonest dietary sources of Vitamin D3 are animal food sources e.g., fatty fish (e.g., salmon, mackerel and tuna) cod liver oil, milk, etc. whereas Vitamin D2 is found in plant sources like sunexposed yeast and mushrooms. The FAO/WHO expert Consultation states that humans can get Vitamin D from abundant sunshine, by exposing 18% of body surface area (without sunscreen) to mid-day sun for 30-45 min [4].
Both the forms of Vitamin D are metabolized in liver to produce first 25(OH)D (Calcidiol) and then 1,25,(OH)2 Vitamin D (Calcitriol) in kidney which is an active but unstable metabolic form. The majority of circulating 25(OH)D and 1,25(OH)2D is tightly bound to Vitamin D Binding Protein (DBP) which is an albumin-like protein produced in the liver. Internalization of DBP-bound 25OHD by the megalin/cubulin endocytic pathway in the proximal tubule epithelium allows for hydroxylation of 25OHD by 1-É‘ hydroxylase to produce 1,25(OH)2D, the active metabolite of Vitamin D for systemic/endocrine functions .
Assessment of Vitamin D status of an individual is best reflected by measurement of 25-hydroxyVitamin D [25(OH) D] as it is the major circulating form of Vitamin D with a half-life of 2-3 weeks. 1, 25(OH)2D (Calcidiol) is the active form with a half-life of only 4 hours and it is not a good indicator of Vitamin D stores as VDD causes PTH elevation that induces increased 1- alpha hydroxylase activity, thereby resulting in normal or increased levels of 1,25(OH)2D and it circulates at the concentration that is 100-1000 fold less than 25(OH) D [5].
Immunoassays such as Radioimmunoassay (RIA), Enzyme Linked Immunosorbant Assay (ELISA), Chemiluminescence immunoassay and protein binding assays are used in routine testing of 25(OH)D in clinical laboratories. High Performance Liquid Chromatography (HPLC) or tandem mass spectrometry though considered as the gold standard for Vitamin D metabolites are expensive, cumbersome and time consuming and therefore hardly used commercially [1].
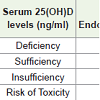
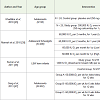
Different Classifications that have been used so far to classify Vitamin D deficiency on basis of 25(OH)D status are as shown in [Table 1] [1,6-8].
Recent evidence suggests maintaining levels above 20 ng/mL for maximizing health benefits like good immune function and overall growth [9].
Currently, there is a high prevalence of VDD worldwide, Despite ample sunshine, it prevails to 50-90% among children in India [10]. Various studies conducted in different parts of the country have reported a widespread prevalence of VDD in all age groups. For understanding functional relevance of this widely prevalent VDD, a comprehensive picture of pediatric studies is required. VDD needs more contemplation among children as similar cut offs may not be directly applied in view of varied metabolism in children hence remains imperceptibly controversial. There is a need to unravel various aspects of VDD in children to guide in the diagnosis and treatment of VDD in children. Some reviews have highlighted the need for large randomized clinical trials to investigate the non-skeletal benefits of Vitamin D [11]. The need of research to provide a meticulous picture of the ongoing VDD is highly endorsed as it will help policymakers to plan supplementation and fortification strategies for preventing VDD [11-15].
The aim of this article is to review studies related to Vitamin D deficiency in Indian children and to throw light on the debate of Vitamin D paradox whether virtual or real. This paper collates information from various epidemiological studies conducted among healthy children, studies that analyze the association of VDD with other morbidities and studies that assess the outcome of Vitamin D supplementation among children thus highlighting the current research gaps and management challenges in Indian context.
Methods
Literature search with keywords focusing on relevant topics such as Vitamin D deficiency, Vitamin D, child health, Pediatric, Hypovitaminosis D, Bone health was carried out through computerized databases-Medline, Embase, PubMed, Cochrane and Google. The personal knowledge of authors and experience in the field has helped in identifying the articles and databases not revealed by computer searches. References of various manuscripts were searched to identify the missing information. The literature search was limited to articles in Indian context over last 20 years. About eighty references were reviewed and studies pertaining to Vitamin D deficiency in pediatric age group were included. Few studies conducted among adult population pertaining to genetic factors and Vitamin D- PTH axis were also included. The studies and their findings are described in brief data is compiled and interpreted to give a comprehensive picture of Vitamin D deficiency among Indian children.
Various studies discussed here have used different cut-off levels to define Vitamin D deficiency, insufficiency and sufficiency levels on basis of 25(OH)D levels either as nM (nanomoles per liter) or ng/mL. For uniformity and ease of comparison, in this review all the data on 25(OH)D levels are presented in a single concentration unit for serum 25(OH)D levels-ng/mL.
Prevalence of VDD in Indian children
This section has been classified into three different age groups ofchildren ie (i) 0-6 months (ii) 6 months to 60 months and (iii) Beyond60 months up to adolescent period.
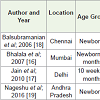
Children aged 0-6 months (Newborns and Breast feeding group): Studies as summarized in the Table 2 were conducted in the age group of newborns up to 6 months and showed prevalence from 62-95.7%. Subnormal maternal Vitamin D status was shown to have a significant association with Vitamin D deficiency in newborns and breastfeeding babies and subclinical Vitamin D deficiency is reported among exclusively breast fed infants with radiological rickets at 25OHD < 10 ng/ml [16,17]. These studies have revealed predisposition of lower 25(OH)D concentrations to neonatal hypocalcaemia and infantile rickets with biochemical evidence of hyperparathyroidism among infants manifesting with hypocalcemic seizures with Vitamin D deficiency [18]. Significant association between a deficient Vitamin D status (45.5%) and low birth weight has also been observed [19].
However, though similar cut offs were not used in the newborn studies done so far, the cutoff of 25(OH)D for diagnosing hypovitaminosis D in the newborn was best considered as 30 nmol/L (12 ng/mL) on basis of raised PTH in a few studies [20].
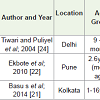
6 months-60 months: Studies summarized in the Table 3 were mainly conducted in the age group of 9 months -5 years with the prevalence of 46-80%. Seasonal variation was observed among 41.6% pre-school children [21]. The study results from slum area in Pune showed that underprivileged toddlers who were deprived of sunlight had a much greater incidence of hypovitaminosis D compared to toddlers in slums exposed to sunlight [22].
Effect of atmospheric pollution on Vitamin D status among toddlers was assessed in a study from Delhi which reported low 25(OH)D values among children residing in a high polluted area [23]. It was hypothesized that the haze caused by aero pollutants reduced UVB rays resulting in reduced cutaneous vitamin D synthesis. However, some studies have reported 80-84% prevalence irrespective of sun exposure and pollution status [24]. However the sample size was inadequate to create conclusive evidence in both the studies.
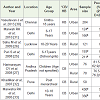
Beyond 60 months up to adolescent period: Prevalence of VDD among 5-20 years children varied between 37.8%-97.5%. Significant correlation between serum 25-hydroxy Vitamin D and estimated sun exposure (r 0•185, P=0•001) and percentage body surface area exposed (r 0•146, P=0•004) was observed among healthy schoolgirls [25]. The seasonal variation has also been reported among rural adolescent girls with manifestation of low serum 25 OHD levels at latitude 26 degree North [26]. The clinical evidence of Vitamin D deficiency was noted only among 10.8%-11.5% of the children exhibiting Vitamin D deficiency.
No significant difference was reported between 25(OH)D levels of urban and rural children, however, low dietary calcium and higher phytate to calcium ratio were significantly associated with low 25(OH)D among rural children [27]. Phytate is the principal storage form of phosphorus in the bran portion of grains and other seeds and is largely consumed by the rural population. It is indigestible to humans and chelates micronutrients such as calcium and iron. High serum phosphate levels further increase production of FGF23 in bone osteocytes via the action of 1,25(OH)2D. This further results in suppressed PTH and 1,25(OH)2D synthesis. These speculated mechanisms were possible explanations for low 25(OH)D levels in rural subjects on a high phytate and/or low calcium diet, despite plentiful sun exposure.
Apart from this, environmental pollutants like fluoride have also been reported to affect bone metabolism severely in the conjunction with inadequate calcium intake, especially in children resulting in low 25(OH)D levels [Table 4] [28,29].
Vitamin D association with other comorbidities in children
Vitamin D is believed to act as a potent antioxidant protecting against free radical damage and an inducer of cellular differentiation, protecting against carcinogenesis [34,35]. It also has a role to fight against serious infections, by controlling T Cell Antigen Receptor (TCR) signaling and activation of human T cells [36]. VDD can therefore be associated with an increase in risk for certain diseases. However, since these diseases are multifactorial, optimum Vitamin D levels may not prevent these diseases, but their risk is lowered.
In pediatric age group, subclinical Vitamin D deficiency and nonexclusive breastfeeding in the first 4 months of life were found to be significant risk factors for acute respiratory tract infections and tuberculosis [37-39]. Significant inverse relationship between Vitamin D levels and severity of Asthma was observed in some studies [40]. Topical Vitamin D (Calcipotrol) has been proven to be effective in management of Psoriasis [41]. Studies have also depicted association of Vitamin D intake and reduced risk of T1DM and 30% reduction in risk [42-44].
Therapeutics of Vitamin D deficiency among children
Vitamin D supplementation studies in healthy Indian children: Supplementation studies, among healthy adolescents have shown a significant improvement in Vitamin D status and bone health. However, large proportion of the population could not attain sufficient levels of Vitamin D [Table 5] [12,30,45-47].
Though studies have shown Vitamin D supplementation decreases and protects against respiratory tract infections, recently published Cochrane Review revealed that under five Children may not demonstrate benefit of Vitamin D supplementation on the incidence of pneumonia [48-50]. This observation was based on findings from one large trial in Afghanistan.
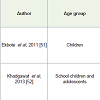
Vitamin D fortification studies in children: Two Vitamin Dfortification studies in healthy children were reported. Significantincrease in serum calcium and Vitamin D levels and also in BoneMineral Content (BMC) was observed in underprivileged toddlers,fed with fortified laddoos. In another study, 776 school children weregiven fortified milk and significant improvement in their Vitamin Dstatus was observed [Table 6] [12,51,52].
Current guidelines for management of Vitamin D deficiency: Several therapeutic regimens have been pursued to rectify the deficiency of Vitamin D. Short term administration of Vitamin D2or D3 2000 units daily or Vitamin D2 50,000 units weekly has yielded equivalent outcomes in treating hypovitaminosis D in young children. The most common recommended regimens are either Vitamin D 1000-5000 units/day for several weeks or single IM injection of 6 lakh units (Stoss therapy) or 50,000 U of Vitamin D2 weekly for 8 weeks. The total therapeutic dose of Vitamin D has been reported to be more predictive of Vitamin D sufficiency rather than the frequency of dosing (daily, weekly or monthly). Therefore, treatment regimensfor a given patient can be individualized to ensure compliance, sinceno difference in the efficacy or safety was reported in these commontreatment regimens [53].
Considering the increased prevalence of VDD and the confusion about supplementation and treatment of vitamin D deficiency for various age groups, the Indian Academy of Pediatrics has recently published a set of recommendations for prevention and treatment of vitamin D and calcium deficiency in July 2017. According to these guidelines, for the prevention of rickets in premature infants, 400 IU of vitamin D and 150-220 mg/kg of calcium, and in neonates, 400 IU of vitamin D and 200 mg of calcium are recommended daily. For prevention of rickets and hypocalcaemia in infants (after neonatal period) up to 1 year of age, and from 1-18 years, 400 IU and 600 IU vitamin D/day and 250-500 mg/day and 600-800 mg/day of calcium, respectively are recommended. For treatment of rickets in premature neonates, infants up to 1 year and from 1-18 years, 1000 IU, 2000 IU and 3000-6000 IU of vitamin D daily, respectively and elemental calcium of 70-80 mg/kg/day in premature neonates and 500-800 mg daily for all children over that age are recommended. It is recommended that larger doses of vitamin D up to 60,000 IU/week for 6 weeks may be given from 3 months to 18 years of age [54].
Research gaps in Indian context
Genetic factors affecting Vitamin D status: Genetic factors such as polymorphisms in 7 dehydroxylase reductase, DBP, 1 alpha hydroxylase, VDR, 25 hydroxylase, 24 hydroxylase affect expression of genes which modulates Vitamin D metabolism resulting in Vitamin D deficiency. DBP is encoded by Gc located on chromosome 4q11–q13. While DBP is relatively stable in most healthy populations, a recent study showed that there are gene polymorphisms associated with race and ethnicity that could alter DBP levels and binding affinity. In cases of low DBP or gene polymorphisms, 25(OH)D - DBP complex will not form which will have implications in Vitamin D status of an Individual. The genetic variance of the common GC T436K SNP affecting circulating levels of the DBP protein and resulting in low circulating 25-OHD has been reported in young children [55]. The DBP and Vitamin D status has also been studied in a very few Indian studies in context to adult population [56]. However, a clear association between these polymorphisms and Vitamin D status is yet to be established pertaining to Indian children [57,58].
Epigenetic factors cause heritable changes in the gene expression without changing the DNA sequence. Post-translational modifications of histones-methylation, acetylation and phosphorylation, and aberrant expression of microRNAs could be some likely mechanisms. Interaction between genetic and environmental factors, modulated by epigenetic factors has also been reported in few studies [59].
Vitamin D and PTH axis: The cutoff values for deciding Vitamin D deficiency are mainly based on adult studies in relation to fracture risk, intestinal calcium absorption, or bone mineral density [60]. The metabolic cutoff values are defined at the level at which serum levels of Parathyroid Hormone (PTH) increase and serum 25OHD level decreases below a variably defined range of 37.5-75 nmol/l (15-30 ng/ ml) [8,61,62].
In children, a few studies have attempted to investigate the Vitamin D and PTH axis and also demonstrated inverse relationships between 25OHD and PTH [33,63-65]. However the deflection points in pediatric population may vary. The vast majority of children with abnormal bone metabolites had 25(OH)D levels less than 13.6 ng/ ml and PTH levels > 50 ng/l. Vitamin D deficiency, based on PTH elevation, was best defined by a 25OHD level of 13.6 ng/ml (34 nmol/l) children with VDD, without raised PTH or signs of rickets are not at an increased risk of fractures [66,67].
Lack of Indian cut off values for diagnosis of Vitamin D deficiency: Indian RDA guidelines 2010 mentions increasing prevalence of Vitamin D deficiency in India, however adequate sun exposure and physical activity is recommended for Vitamin D and no specific suggestions are made on intakes for different age groups [68]. This is because very few Indian foods are fortified with Vitamin D, and that too, with small amounts [69]. However, recently published nutrition Guidelines by NIN ,ICMR have calculated Vitamin D2 content of plant foods and D3content of animal foods using LCMS. This new analytical method Vitamin D data in food will help in dietary intake assessment of Vitamin D and may be taken up in future studies.
No Indian guidelines for identifying cut offs for Indian children have yet been established. Similarly though IAP has come up with the guidelines for Vitamin D supplementation among children, no supplementation strategy of Vitamin D with special regard to under five children is yet defined by the Government.
Discussion
Prevalence and attributing factors
Various studies have discussed different aspects of Vitamin D deficiency among children. It is observed that inadequate sun exposure contributes significantly to Vitamin D deficiency in children. Pollution results in high haze score which hinders solar UVB rays reaching the ground because of high haze score. This hampers Vitamin D synthesis. The lack of space, overcrowded tenements prevent the direct sunlight reaching inside most parts in urban regions and gives limited scope for outdoor activities among children. The extreme discomfort of the scorching heat also keeps children away from the midday sun exposure. This specially holds true among under-five children who are either in preschools indoors during 10 am to 3 pm making them more susceptible for Vitamin D deficiency owing to inadequate sun exposure. High phytate and/ or low calcium diet are likely mechanisms for Vitamin D deficiency among children despite adequate sun exposure. The cooking practices also alter Vitamin D content of foods as Vitamin D is stable up to 200 °C. Since most cooking fats and oils have smoke points above 180 °C, shallow and deep-frying of food degrades Vitamin D once it comes out into the cooking medium. All these factors may contribute to Vitamin D deficiency among children.
Defining Vitamin D cutoffs in Indian Children
Substantial amount of data has been generated on VDD among children is demonstrating heterogeneity. Majority of the studies have focused on school going children and adolescents. The data in under-five children is very limited and does not give a comprehensive picture. Most of the studies have used 20 ng/ml as cut off for defining VDD in children. However whether to apply same cut off in children s a matter of concern. As suggested in few reviews published before, VDD among children may be defined either on basis of a locally developed “population based reference value” which may show region specific trends or a “functional health based reference value” hich physiologically defines hypovitaminosis D as the concentration of 25(OH)D at which PTH begins to increase. Even though, optimal level of 20 ng/ml is essential for maximizing the health benefits, the biochemical definition of VDD does carry importance in children as deflection points vary in children. The “optimal” 25OHD threshold which separates blood markers of abnormal bone metabolism from normal bone metabolism may differ in children as it is observed that PTH elevation levels in response to low 25(OH)D levels in pediatric age group is not as same as that that of adults. Therefore simply estimating deficiency on the basis of population based reference values may not be appropriate. The studies conducted on basis of Vitamin D-PTH axis among children have identified lower cut off values among children on basis of PTH response which is analogous with the Pediatric Endocrinology Society classification. However there is no clear consensus for defining VDD among Indian children based on these studies on Vitamin D-PTH axis. Therefore further research is needed focusing especially on under-five children to create evidence on VDD for planning any supplementation or fortification strategies for children.
Newer perspectives
The Vitamin D among children needs further exploration. Factors like D binding protein to which majority of 25(OH)D is bound may have some role in Vitamin D deficiency in children. Determinants of circulatory levels of Vitamin D metabolites in children have not yet been evaluated among Indian children. Alterations in DBP levels may be potential confounders on the interpretation of total 25(OH) D concentrations among children. The biological relevance of the DBP-bound Vitamin D metabolites versus the DBP-unbound or “free fraction” of Vitamin D has not yet been established. Further research is required to estimate complete 25(OH)D status including free 25(OH) index, Vitamin D Binding Protein (DBP) to understand whether the free 25(OH)D index as compared to total 25(OH)D levels is a better marker of 25(OH)D tissue availability.
The information regarding association between genetic and/ or epigenetic factors and Vitamin D status is also inconclusive and warrants further study. Future studies may consider all these aspects. Recently published nutrition Guidelines by NIN, ICMR may be referred in dietary intake assessment of Vitamin D and can be considered in future studies.
Therapeutics
Vitamin D sufficiency status may not be treated as a “feel good status” for the children and irrational use of massive doses to be avoided. Advocating Educational Programs in physical activity especially among under-five children should be encouraged in preschools. Supplementation strategies advised by Indian Academy of Pediatrics (IAP) such as supplementation in newborn period and Infancy may be followed till any strategic plan is made at national level.
Conclusion
There is abundant room for further progress in determining Vitamin D status in children by understanding overall Vitamin D status of children including genetic, environmental and behavioral factors and needs further research.
Acknowledgement
The authors acknowledge the encouragement received fromICMR (NIRRH/REV/497/07-2017)
References
- Misra M, Pacaud D, Petryk A, Collett-Solberg PF, Kappy M (2008) Vitamin D deficiency in children and its management: Review of current knowledge and recommendations. Pediatrics 122: 398-417.
- Ford JA, McIntosh WB, Butterfield R, Preece MA, Pietrek J, et al. (1976) Clinical and subclinical Vitamin D deficiency in Bradford children. Arch Dis Child 51: 939-943.
- Specker BL, Valanis B, Hertzberg V, Edwards N, Tsang RC (1985) Sunshine exposure and serum 25-hydroxy Vitamin D concentrations in exclusively breast-fed infants. J Pediatr 107: 372-376.
- FAO (1998) Preliminary report on recommended nutrient intakes: Joint FAO/WHO Expert Consultation on Human Vitamin and mineral requirement. Bangkok.
- Holick MF (2009) Vitamin D status: Measurement, interpretation and clinical application. Ann Epidemiol 19: 73-78.
- Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, et al. (2011) Evaluation, treatment and prevention of Vitamin D deficiency: An endocrine society. J Clin Endocrinol Metab 96: 1911-1930.
- Ross AC, Manson JE, Abrams SA, Aloia JF, Brannon PM, et al. (2011) The 2011 report on dietary reference intakes for calcium and Vitamin D from the institute of medicine: What clinicians need to know? J Clin Endocrinol Metab 96: 53-58.
- Lips P (2001) Vitamin D deficiency and secondary hyperparathyroidism in the elderly: Consequences for bone loss and fractures and therapeutic implications. Endocr Rev 22: 477-501.
- Harinarayan CV, Joshi SR (2009) Vitamin D status in India-Its implications and remedial measures. J Assoc Physicians 57: 40-48.
- Rathi N, Rathi A (2011) Vitamin D and child health in the 21st Century. Indian pediatr 48: 619-625.
- Ritu G, Gupta A (2014) Vitamin D deficiency in India: Prevalence, causalities and interventions. Nutrients 6: 729-775.
- Balasubramanian S, Dhanalakshmi K, Amperayani S (2013) Vitamin D deficiency in childhood -A review of current guidelines on diagnosis and management. Indian Pediatr 50: 669-675.
- Londhey V (2011) Vitamin D deficiency: Indian Scenario. J Assoc Physicians 59: 695-696.
- Urvashi Mehlawat, Priyanka Singh, Shubhra Pande (2014) Current status of Vitamin-D deficiency in India. IPP 2: 328-335.
- Bhalala U, Desai M, Parekh P, Mokal R, Chheda B (2007) Subclinical hypovitaminosis D among exclusively breastfed young infants. Indian Pediatr 44: 897-901.
- Jain V, Gupta N, Kalaivani M, Jain A, Sinha A, et al. (2011) Vitamin D deficiency in healthy breastfed term infants at 3 months & their mothers in India: Seasonal variation & determinants. Indian J Med Res 133: 267-273.
- Balasubramanian S, Shivbalan S, Kumar PS (2006) Hypocalcemia due to Vitamin D deficiency in exclusively breastfed infants. Indian Pediatr 43: 247-251.
- Nageshu S, Krishna K, Krishna L, Shyamasundara Bhat B, Suma HR, et al. (2016) A study of prevalence of Vitamin D deficiency among pregnant women and its impact on feto maternal outcome. Int J Reprod Contracept Obstet Gynecol 4: 1174-1180.
- Zeghoud F, Vervel C, Guillozo H, Walrant-Debray O, Boutignon H, et al. (1997) Subclinical vitamin D deficiency in neonates: Definition and response to vitamin D supplements. Am J Clin Nutr 65: 771-778.
- Basu S, Gupta R, Mitra M, Ghosh A (2015) Prevalence of vitamin D deï¬ciency in a pediatric hospital of eastern India. Indian J Clin Biochem 30: 167-173.
- Ekbote VH, Khadilkar AV, Mughal MZ, Hanumante N, Sanwalka N, et al. (2010) Sunlight exposure and development of rickets in Indian toddlers. Indian J Pediatr 77: 61-65.
- Agarwal KS, Mughal MZ, Upadhyay P, Berry JL, Mawer EB, et al. (2002) The impact of atmospheric pollution on vitamin D status of infants and toddlers in Delhi, India. Arch Dis Child 87: 111-113.
- Tiwari L, Puliyel JM (2004) Vitamin D level in slum children of Delhi. Indian Pediatr 41: 1076-1077.
- Puri S, Marwaha RK, Agarwal N, Tandon N, Agarwal R, et al. (2008) Vitamin D status of apparently healthy school girls from two different socioeconomic strata in Delhi: Relation to nutrition and lifestyle. Br J Nutr 99: 876-882.
- Sahu M, Bhatia V, Aggarwal A, Rawat V, Saxena P, et al. (2009) Vitamin D deficiency in rural girls and pregnant women despite abundant sunshine in northern India. Clin Endocrinol (Oxf) 70: 680-684.
- Harinarayan CV, Ramalakshmi T, Prasad UV, Sudhakar D (2008) Vitamin D status in Andhra Pradesh: A population based study. Indian J Med Res 127: 211-218.
- Khandare AL, Harikumar R, Sivakumar B (2005) Severe bone deformities in young children from vitamin D deficiency and fluorosis in Bihar-India. Calcif Tissue Int 76: 412-418.
- Harinarayan CV, Kochupillai N, Madhu SV, Gupta N, Meunier PJ (2006) Fluorotoxic metabolic bone disease: An osteo-renal syndrome caused by excess fluoride ingestion in the tropics. Bone 39: 907-914.
- Vasudevan J, Reddy MG, Jenifer A, Thayumanavan S, Devi U, et al. (2014) Prevalence and factors associated with vitamin D deficiency in Indian children: A hospital based cross sectional study. Pediatr Oncall J 2: pp. 71.
- Marwaha RK, Tandon N, Agarwal N, Puri S, Agarwal R, et al. (2010) Impact of two regimens of vitamin D supplementation on calcium - vitamin D - PTH axis of schoolgirls of Delhi. Indian Pediatr 47: 761-769.
- Khadilkar AV (2010) Vitamin D deficiency in Indian adolescents. Indian Pediatr 47: 755-756.
- Marwaha RK, Sripathy G (2008) Vitamin D & bone mineral density of healthy school children in northern India. Indian J Med Res 127: pp. 239-244.
- Holick MF (2004) Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers and cardiovascular disease. Am J Clin Nutr 80: 1678S-1688S.
- Bartoszewska M, Kamboj M, Patel DR (2010) Vitamin D, muscle function and exercise performance. Pediatr Clin North Am 57: 849-861.
- Walker VP, Modlin RL (2009) The vitamin D connection to pediatric infections and immune function. Pediatr Res 65: 106R-113R.
- Wayse V, Yousafzai A, Mogale K, Filteau S (2004) Association of subclinical Vitamin D deficiency with severe acute lower respiratory infection in Indian children under 5 y. Eur J Clin Nutr 58: 563-567.
- Ramaiah JD, Reddy PS, Sheshacharyulu M, Radhakrishna KV, Subrahmanyam GV (2014) Association of vitamin D deficiency with sever acute lower respiratory tract infection in children of less than 2 year age. Asian J Biochem Pharm Res 4: 11-19.
- Dhingra A, Dhanda MS, Goel AK, Kumar N, Madan HS, et al. (2015) Hypovitaminosis and tuberculosis: A comparative study in children of mewat. Indian J Sci Res 6: 81-84.
- Eleazar Suchiang, Jagdish Singh, Anil Kumar Saroj, Mordhwaj Singh (2016) Assessment of 25 (oh) vitamin D3 in asthmatic children in S.P.M.C.H.I, Jaipur. J Dent Med Sci 15: pp. 16-22.
- Oranje AP, Marcoux D, Svensson A, Prendiville J, Krafchik B, et al. (1997) Topical calcipotriol in childhood psoriasis. J Am Acad Dermatol 36: 203-208.
- Mutlu A, Mutlu GY, Özsu E, Çizmecioğlu FM, Hatun Ş (2011) Vitamin D deficiency in children and adolescents with type 1 diabetes. J Clin Res Pediatr Endocrinol 3: 179-183.
- VV Borkar, Devidayal, Verma S, Bhalla AK (2010) Low levels of vitamin D in north Indian children with newly diagnosed type 1 diabetes. Pediatr Diabetes 11: 345-350.
- Zipitis CS, Akobeng AK (2008) Vitamin D supplementation in early childhood and risk of type 1diabetes: A systematic review and meta-analysis. Arch Dis Child 93: 512-517.
- Khadilkar AV, Sayyad MG, Sanwalka NJ, Bhandari DR, Naik S, et al. (2010) Vitamin D supplementation and bone mass accrual in underprivileged adolescent Indian girls. Asia Pac J Clin Nutr 19: 465-472.
- Kumar GT, Sachdev HS, Chellani H, Rehman AM, Singh V, et al. (2011) Effect of weekly vitamin D supplements on mortality, morbidity, and growth of low birth weight term infants in India up to age 6 months: Randomized controlled trial. BMJ 342: pp. d2975.
- Garg MK, Marwaha RK, Khadgawat R, Ramot R, Obroi AK, et al. (2013) Efficacy of vitamin D loading doses on serum 25-hydroxy vitamin D levels in school going adolescents: An open label non-randomized prospective trial. J Pediatr Endocrinol Metab 26: 515-523.
- Charan J, Goyal JP, Saxena D, Yadav P (2012) Vitamin D for prevention of respiratory tract infections: A systematic review and meta-analysis. J Pharmacol Pharmacother 3: 300-303.
- Martineau Adrian R, Jolliffe David A, Hooper Richard L, Greenberg Lauren, Aloia John F, et al. (2017) Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 356: pp. i6583.
- Yakoob MY, Salam RA, Khan FR, Bhutta ZA (2016) Vitamin D supplementation for preventing infections in children under five years of age. Cochrane Database of Systematic Reviews.
- Ekbote VH, Khadilkar AV, Chiplonkar SA, Hanumante NM, Khadilkar VV, et al. (2011) A pilot randomized controlled trial of oral calcium and vitamin D supplementation using fortified laddoos in underprivileged Indian toddlers. Eur J Clin Nutr 65: 440-446.
- Khadgawat R, Marwaha RK, Garg MK, Ramot R, Oberoi AK, et al. Impact of vitamin D fortified milk supplementation on vitamin D status of healthy school children aged 10-14 years. Osteoporos Int 24: 2335-2343.
- Gordon CM, Williams AL, Feldman HA, May J, Sinclair L, et al. (2008) Treatment of hypovitaminosis D in infants and toddlers. J Clin Endocrinol Metab 93: 2716-2721.
- Khadilkar A, Khadilkar V, Chinnappa J, Rathi N, Khadgawat R, et al. (2017) Prevention and treatment of vitamin D and calcium deficiency in children and adolescents: Indian academy of pediatrics (IAP) guidelines. Indian Pediatr 54: 567-573.
- Thomas O. Carpenter, Jane H. Zhang, Esteban Parra, Bruce K. Ellis, Christine Simpson, et al. (2013) Vitamin D binding protein is a key determinant of 25-hydroxyvitamin D levels in infants and toddlers. J Bone Miner Res 28: 213-221.
- Goswami R, Saha S, Sreenivas V, Singh N, Lakshmy R (2017) Vitamin D-binding protein, vitamin D status and serum bioavailable 25(OH)D of young Asian Indian males working in outdoor and indoor environments. J Bone Miner Metab 35: 177.
- Wang TJ, Zhang F, Richards JB, Kestenbaum B, van Meurs JB, et al. (2010) Common genetic determinants of Vitamin D insufficiency: A genome-wide association study. Lancet 376: 180-188.
- McGrath JJ, Saha S, Burne TH, Eyles DW (2010) A systematic review of the association between common single nucleotide polymorphisms and 25-hydroxyVitamin D concentrations. J Steroid Biochem Mol Biol 121: 471-477.
- Hossein-nezhad A, Holick MF (2012) Optimize dietary intake of Vitamin D: An epigenetic perspective. Curr Opin Clin Nutr Metab Care 15: 567-579.
- Vieth R, Ladak Y, Walfish PG (2003) Age-related changes in the 25-hydroxyvitamin D versus parathyroid hormone relationship suggest a different reason why older adults require more vitamin D. J Clin Endocrinol Metab 88: 185-191.
- Malabanan A, Veronikis IE, Holick MF (1998) Redefining vitamin D insufficiency. Lancet 351: 805-806.
- Steingrimsdottir L, Gunnarsson O, Indridason OS, Franzson L, Sigurdsson G (2005) Relationship between serum parathyroid hormone levels, vitamin D sufficiency and calcium intake. JAMA 294: 2336-2341.
- Hill TR, Cotter AA, Mitchell S, Boreham CA, Dubitzky W, et al. (2010) Vitamin D status and parathyroid hormone relationship in adolescents and its association with bone health parameters: Analysis of the Northern Ireland young heart’s project. Osteoporos Int 21: 695-700.
- El-Hajj Fuleihan G, Nabulsi M, Choucair M, Salamoun M, Hajj Shahine C, et al. (2001) Hypovitaminosis D in healthy school children. Pediatrics 107: E53.
- Outila TA, Kärkkäinen MU, Lamberg-Allardt CJ (2001) Vitamin D status affects serum parathyroid hormone concentrations during winter in female adolescents: Associations with forearm bone mineral density. Am J Clin Nutr 74: 206-210.
- Navoda Atapattu, Nick Shaw Wolfgang Högler (2013) Relationship between serum 25-hydroxyvitamin D and parathyroid hormone in the search for a biochemical definition of vitamin D deficiency in children. Pediatr Res 74: 552-556.
- Munns CF, Shaw N, Kiely M, Specker BL, Thacher TD, et al. (2016) Global consensus recommendations on prevention and management of nutritional rickets. J Clin Endocrinol Metab 101: 394-415.
- National institute of nutrition (2009) A report of the expert group of the indian council of medical research: Nutrient requirements and recommended dietary allowances for Indians. Indian council of medical research, Hyderabad. pp. 1-334.
- Ritu G, Gupta A (2014) Fortification of foods with vitamin D in India. Nutrients 6: 3601-3623.
- Longavah T, Ananthan R, Bhaskarachary K, Venkaih K (2017) Indian food composition tables: National institute of nutrition. Hyderabad, Telangana, India. pp. 1-522.