Review Article
Microalgae (Spirulina Platensis) and Its Bioactive Molecules: Review
Smriti Kana Pyne1*, Paramita Bhattacharjee1 and Prem Prakash Srivastav2
1Department of food Technology and Biochemical Engineering, Jadavpur University, Kolkata, India
2Department of Agricultural and Food Engineering, IIT Kharagpur, India
Corresponding author: Smriti kana Pyne, Department of food Technology and Biochemical Engineering, JadavpurUniversity, Kolkata, India; Tel: 9476249230; E-mail:smritipyne10@gmail.com
Citation: Pyne PK, Bhattacharjee P, Srivastav PP. Microalgae (Spirulina Platensis) and Its Bioactive Molecules: Review. Indian J Nutri. 2017;4(2): 160.
Copyright © 2017 Pyne SK, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Indian Journal of Nutrition | ISSN: 2395-2326 | Volume: 4, Issue: 2
Submission: 13/04/2017; Accepted: 10/05/2017; Published: 22/05/2017
Abstract
Spirulina platensis, a species of blue green algae of the cyanobacteria family is currently of global nutritional significance. It’s high content of proteins, vitamins, antioxidants, antimicrobials and anti-carcinogenic biomolecules is being researched upon. Various nutritional potencies have been attributed to metabolites from the cyanobacteria, Spirulina platensis. Spirulina serve as very viable potential sources of bioactive products with commercial importance. Therefore, more should be done in the culture, isolation, and purification of microalgae to enable beneficial harvest of their important inclusions.
Keywords:
Spirulina platensis; Nutritional composion; Antioxidant
Introduction
Spirulina platensis is currently of global nutritional significance to redress malnutrition especially in children. This blue green algae of the cyanobacteria family grown in temperate waters around the world that is considered as a functional food due to its rich source of proteins, vitamins, minerals, healthy fatty acids and other healing phytonutrients, such as numerous active plant pigments. From ancient times a blue green microalgae has been used as a source of food supplement for its high protein content and nutritional value [1]. Due to its high content of valuable substances spirulina has benefit for use as bioactive substance. It has been also demonstrated several beneficial health effects due to their ability to act as scavengers for reactive oxygen species.
Spirulina platensis is a potential source of high value compoundswith functional properties e.g., phycocyanins, carotenoids, phenolicacids and omega-3 and omega-6 polyunsaturated fatty acids [2].
Recently algae are widely being studied for its nutritional reasons and also for its therapeutic properties. Several studies have shown that Spirulina or its extracts could prevent or inhibit cancer in humans and animals for its immuno-promoting effects [3,4]. S. platensis was also has antimicrobial activity to inhibit replication of several viruses such as Herpes simplex and HIV-1 [5-7]. Moreover Spirulina contains a whole spectrum of natural mixed carotene and xanthophylls phytopigments which together seem to be relatedto its antioxidant activity [8-10]. Now the new food ingredients obtained by extraction of natural products is growing the demand of interest in functional foods. In this way, several studies showed that Spirulina platensis or its extracts could possess physiological profits as ntioxidant, antimicrobial, anti-inflammatory, antiviral or antitumorproperties.
Nutritional Composition of Algal Biomass
Spirulina is an incredible natural source of nutrition which has been used since ancient times. Spirulina is a major alga which is used as food supplement. The different nutritional compositions of microalgae have been analyzed since 1970. They were shown to be excellent sources of protein, amino acid, minerals etc. [11]. Itscomposition is discussed below:
Protein
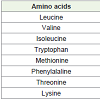
Spirulina consists of essential amino acids mainly leucine, valine and isoleucine along with tryptophan, methionine, phenylalanine, threonine and lysine [12]. Among the non-essential amino acids the algae is rich source of aspartate and glutamate. The protein denatures at and above 67°C, in neutral aqueous solution (Table 1).
Table 1: Protein composition of Spirulina microalgae [12].
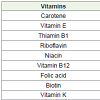
Vitamins
Spirulina is a great source of beta-carotene (provitamin A) and vitamin B-12. Vit B-12 is very useful in treatment of pernicious anemia [13-16]. The vitamins content of Spirulina platensis are described in Table 2.
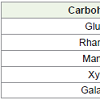
Carbohydrates - Glucose, rhamnose, mannose, xylose andgalactose etc are found in microalgae biomass (Table 3) [17].
Lipids
Spirulina contains 4-7% lipids. Spirulina mainly has essential fatty acids like GLA which is required for arachidonic acid and prostaglandin synthesis. Spirulina is one of the few natural dietary sources of Gamma Linolenic Acid (GLA), an omega 6 fatty acid (Table 4) [18,19].
Table 4: Lipid composition of Spirulina microalgae [18].
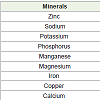
Minerals
Spirulina is an adequate source of iron. The mineral compositionof Spirulina powder is described in Table 5.
Table 5: Minerals composition of Spirulina microalgae [18].
Pigments
Spirulina has a high ratio of plants pigments and phytonutrients, including phycocyanin, carotenoids, xanthophylls and chlorophyll. Phycocyanin is a blue pigment (bilirubin) found only in blue-green algae [20,21]. Pigments are usually associated with thylakoid lamella of the algae (Table 6).
Table 6: Phytonutrients of spirulina algae [22].
Harvesting and Post Harvesting Processing of AlgalBiomass
Biomass harvesting requires one or more solid liquid separation methods. Biomass can be harvested by any of the three methods viz, 1. Flocculation-The commonly used salts include ferric chloride, aluminum sulphate and ferric sulphate. 2. Centrifugation-Centrifugal recovery can be rapid but it is energy intensive [23]. So the application of centrifugation for recovery of biomass for commercial purpose is minimal and 3. Filtration-Filter presses operating under pressure or vacuum are reasonable for bulk recovery. Rotary drum vacuum or pressure filters are also preferred for harvesting algal biomass.
The harvested biomass slurry must be processed rapidly or it can be spoiled within a few hours in a hot climate. Dehydration or drying method is mostly used. Absence of water in the biomass enhances the recovery of lipid soluble components such as astaxanthin and beta carotene. Different drying methods are employed for the drying of biomass. It includes spray drying, cross flow drying, oven drying, sun drying, recirculatory hot air drying, freeze drying etc. [24]. Spray drying is already a commercial method widely used and product quality obtained is also high having additional advantages as uniform appearance and relatively easy packaging. The freeze drying method is also recommended because of no or minimal product degradation and higher biomass quality [25]. However freeze drying is too expensive for use in large scale.
Nutritional Value of Spirulina Platensis
Arthrospira (Spirulina), the richest sources of proteins is about 60-70%. Attempting a study on using Spirulina as a protein supplement, it was observed that it can be substituted up to 40% of protein content in diets. Rabelo have explained on the development of cassava doughnuts enriched with S. Platensis biomass [26]. Unlike many other cyanobacteria that have proven toxicity, no such property has been attributed to Spirulina. Becker et al. reported that organic nitrogenous rich Spirulina constituents were used as food supplement in Chad Republic (Africa). The alga strain growths in an open air pilot production unit were determined with respect to its amino acids, vitamins and nutritive value. It contained 62% protein, high digestibility and vitamins like β-carotene. Lyophilized Spirulina platensis had a nutritional quality between that of wheat gluten and casein supplemented with methionine. Usharani showed that the drum dried preparations of Spirulina platensis were fairly similar in quality to that of casein sample with methionine [27,28]. It also reported on the water-soluble sugars constituting the major carbohydrates of Spirulina. The low amount of starch was due to the high α and β amylase activity of in Spirulina. The in vitro digestibility of Spirulina had been reported using an amylase enzyme. A sufficient amount of protease activity was mainly involved in protein turn over rather than in storage hydrolysis. Markou studied on the production and nutritive value of Spirulina platensis on swine wastes [29]. The alga contained 55 to 61% crude protein. Natives of the chad region still supplements their diet with Spirulina harvested from lakes. They fed Spirulina to nourish adults. It showed positive nitrogen balance without its harmful effects on human body. Gerde reported on the characteristics of the protein of fresh water mass cultures of Spirulina platensis [30]. The total protein was 50-65% of which nearly 9.9% was non-protein nitrogen. Total protein was extracted by three successive extractions methods using water as a solvent. The isoelectric point was found to be 3.0. The polyacrylamide gel electrophoretic pattern showed seven bands. The in vitro digestibility was found to be 85% when assayed with a pepsin pancreatic system. Khan also reported the biochemistry and industrial potential of Spirulina [31]. It’s consumption by human populations indicated a lack of toxic effects on human bodies. It could be used as a source of chemical and other basic commodities like enzymes, vitamins, lubricants, pigments, etc. Bonotto reported that many species of microalgae and blue green algae under suitable conditions grew fastly and produced large biomass with high protein content [32]. They constituted a source of single cell protein supplemented with the algal biomass. The carotenoids composition of the blue green alga, Spirulina was estimated using High Performance Liquid Chromatography. Freezedried Spirulina had a total xanthophylls concentration of 5787 mg/kg. Manjit Kaur carried out the biochemical studies of Spirulina sugars and enzymes [33]. The content of total water soluble sugars were more (7.31%) than acid soluble sugars (6.19%) and the activities of α amylase (8.5%), β amylase (5.33%) and protease (1.07%) were also recorded simultaneously. The activity of acid phosphatase showed insignificant statistically. Powdered Spirulina contained 480 mg of iron per kg. The sample grown in chelated iron showed a higher percentage of available iron in the wet algae (15-20%) than the dried algae (8-10%) as compared to the control which had traces of iron. Water extract of Spirulina was found to inhibit the lipid peroxidation to 76% and the alcohol extract to 65%. The chemical antioxidants like α tocopherol, Butylated hydroxyanisole and β carotene gave 35, 45 and 48% respectively. Mendiola screened on functional compounds of supercritical fluid extracts from Spirulina platensis [34]. It can also be inferred that extraction at 220 bars and 55°C yields the best extracts in terms of antioxidant and antimicrobial activities. Under these conditions, a good fractionation was also achieved. Aqueous extract of Spirulina has a protective effect against apoptotic cell death due to free radicals. This potential application of incorporating Spirulina into food products and beverages enhance their antioxidant capacity [35]. Microalgae are commercially important due to its mass production ability, high quality protein, lower production cost, no toxicity, high nutritional value easily digestible, can add natural color in food, used in cosmetics and pharmaceuticals products [36]. Nutraceutical properties due to presence of amino acids, pigments and essential fatty acid which acts as an anti-inflammatory that sometimes alleviates symptoms of arthritic conditions.
Sundararaman studied the bioactive potential of marine Cyanobacteria in the animal based systems where twelve different strains were administered to male albino wistar rats [37]. Spirulina platensis, a cyanobacterium was evaluated as an ameliorant of Hexachloro Cyclohexane (HCH), an organochlorine insecticide, idely used in India. HCH induced dietary toxicity in retinal deficient male albino rats by feeding HCH mixed with diets free of vitamin. Growth rate was considerably reduced in rats fed with vitamin A free diet with and without HCH. The body weight gain increased in rats fed with alga supplemented diets with or without HCH.
Lactococcus lactis, Streptococcus thermophilus, Lactobacillus casei, Lactobacillus acidophilus, Lactobacillus delbrueckii and Lactobacillus bulgaricus were grown in rich media as well as in minimal saline medium with and without addition of extracellular products from cultures of Spirulina platensis. The stimulatory effect was observed in media with pH adjusted to 5.3, 6.3 and 7.0. The results showed significantly increments of bacterial growth in all media enriched with extracellular algal filtrates. Phycocyanin, a blue colourant from the cyanobacterium Spirulina platensis was evaluated for its natural food colour by toxicity evaluation studies in albinorats. While testing for mutagenicity, acute, subchronic and chronic toxicities and teratogenicity in animal experimentations, Chamorro have shown that Spirulina did not exhibit any potential for organ or system toxicity even though the doses given were elevated above those for expected human consumption [38]. Likewise, dietary supplementation of Spirulina has helped in alleviating the incidence of anemia experienced during pregnancy and lactation. L. Anitha studied the effect of supplementation of Spirulina on Blood Glucose, Glycosylated Hemoglobin and Lipid Profile of Male Noninsulin Dependent Diabetics [39]. It can be also concluded that Spirulina is potent neutraceutical both as a hypoglycemic and hypolipidemic agent.
Production and Extraction of Bioactive Molecules from S.Platensis
Extraction techniques are widely employed for the isolation of bioactive compounds from natural sources. Current techniques of solid-liquid extraction are essentially based on diffusion and osmosis. However they are usually time consuming and some cases are liable to cause degradation or unwanted chemical changes in the products. Different methods are employed for the extraction of intracellular components from biomass. Freezing and thawing of cells, homogenization using a mortar and pestle, using a blender at high rpm, ultra sound-assisted extraction. Supercritical fluid extraction and accelerated solvent extraction were commonly employed for the extraction of bioactive components from Spirulina biomass [40]. Of all methods ultra sound assisted and super critical fluid extraction have recently got more attention among researchers because of its first and maximum response. Supercritical fluid extraction has certain disadvantages like the extraction temperature may cause degradation of heat sensitive bioactive, expensive process and also the extraction yield is equal or in some cases lower than the normal acid extraction yield [41]. Solvent extraction procedures are widely employed till date for extraction of bio molecules from biomass. The solvent used for the extraction of bioactive from microalgae varies and includes hexane, methanol, acetone, petroleum ether, ethanol and water. Of all solvents ethanol was found to be the best extraction method solvent for anti-oxidant because unlike hexane or petroleum ether, is generally considered GRAS (Generally Recognized as safe) and therefore can be used as a safe solvent for the food industry. Moreover, the antioxidants yield obtained with ethanol are the highest (10.9%) than other solvents (1.4-7.1), providing a good efficiency of the extraction process [42]. The blue green algae S. platensis is a potential source of GLA, an essential polyunsaturated fatty acid of excellent economic interest [43]. Natural sources of GLA contain variable amounts of this fatty acid which rarely exceed 25%. Hence there has always been a keen interest in producing higher concentration of GLA. Lipid fractionation of Spirulina sp can be achieved on silica gel column chromatography and preparative TLC. The fatty acid composition of microalgae can be determined after trans-methylation by gas chromatography using highly polar EGSS-X column [44]. At the molecular and cellular levels antioxidants serve to deactivate certain particles called free radicals. Free radicals are the natural by-products of many oxidative metabolic processes within cells. These free radicals can cause damage to cell walls, certain cell structures and genetic material within the cells. It has been also demonstrated that the use of the combined protocol PLE-TLCHPLCDAD allows in a fast and simple way the extraction, purification and preliminary characterization of several antioxidant compounds from Spirulina platensis microalgae. According to Miranda the main phenolic compounds of Spirulina were salicylic, trans-cinnamic, synaptic, chlorogenic, quimic and caffeic acids [45]. But the formation of these compounds in cyanobacteria and their importance were still unknown. For green plants, Rechner reported that phenylalanine could be converted by an ammonia lyase to trans-cinnamic acid that was transformed to cumaric acid which was in turn converted to caffeic acid [46]. These compounds are used to produce flavonoids, with antioxidant activity. Luciane reported that the antioxidant potential of the phenolics present in the cyanobacterium Arthospira platensis and also indicated that the quantities of phenolics could be increased by altering the culture conditions, suggesting that it was possible to increase the antioxidant potential of S. platensis biomass for use as a nutritional supplement. The highest quantities of phenolics were obtained at 35°C in medium supplemented with 1.875 g.L-1 or 2.5 g.L-1 of sodium nitrate. The phenol-containing methanol extracts obtained from S. Spirulina was grown in medium containing 1.875 g.L-1 of sodium nitrate and 35% in medium containing 2.5 g.L-1 of sodium nitrate.
Antioxidant Potential and Therapeutic Benefits
Apart from its importance as a food additive for supplementary dietary proteins, there are also a lot of potentials for medical and therapeutic applications. For example, A. platensis plays a hepatoprotective role [15]. This role which has to do with the antioxidant activity of Spirulina has been previously asserted by various researchers. The antioxidant activity of Spirulina is described to the presence of two phycobiliproteins: phycocyanin and allophycocyanin, as determined by its action against OH radical generated from ascorbate/iron/H2O2 system. The activity was found to be proportional to the concentration of the phycobiliproteins and was mainly due to the phycocyanin content. As an antioxidant effect, oxygen stress was inhibited by phycocyanin and phycocyanobilin from Spirulina leading to protection against diabetic nephropathy. In an earlier experiment to determine the radical scavenging activity of C-phycocyanin, an intraperitoneally administered C-phycocyanin was found to reduce the peroxide values of CCl4-induced lipid peroxidation in rat liver microsomes Spirulina fusiformis also has shown some free radical scavenging activities. In a more recent finding, aqueous extract of S. platensis showed suppressive potency, through free radical scavenging activity, against cyclophosphamideinduced lipid peroxidation in goat liver homogenates.
Conclusion
An important issue surrounding production of blue green algaeis the possibility of harvesting other blue green algae containing cytotoxin. There is a risk when harvesting algae from natural bodies of water with mixed cultures of microscopic algae. For better commercialization of the Spirulina it is important to focus on its quality aspects. Algal toxin is capable of causing widespread poisoning to animals and humans. Various research activities are going-on in different parts of the world for extracting bioactive of therapeutic potentials from this microalgae, so that it can be further exploited for health claims. The research should focus on developing safe extraction strategies procedures for bioactive compounds, so that it is a non-toxic and fit for human consumption. There is considerable scope for research in Spirulina processing for developing various types of food products with extended shelf life.
Acknowledgement
Authors are acknowledged to the University Grants Commission,India for proving financial help.
References
- Burtin P (2003) Nutritional value of seaweeds. Electron J Environ Agric Food Chem 2: 448.
- Lordan S, Ross RP, Stanton C (2011) Marine bioactives as functional food ingredients: Potential to reduce the incidence of chronic diseases. Mar Drugs 9: 1056-1100.
- Hirahashi T, Matsumoto M, Hazeki K, Saeki Y, Ui M, et al. (2002) Activation of the human innate immune system by Spirulina: Augmentation of interferon production and NK cytotoxicity by oral administration of hot water extract of Spirulina platensis. Int Immunopharmacol 2: 423-434.
- Subhashini J, Mahipal SV, Reddy MC, Reddy MC, Rachamallu A, et al. (2004) Molecular mechanisms in C-Phycocyanin induced apoptosis in human chronic myeloid leukemia cell line-K562. Biochem Pharmacol 68: 453-462.
- El-Sheekh MM, Daboor SM, Swelim MA, Mohamed S (2014) Production and characterization of antimicrobial active substance from Spirulina platensis. Iran J microbiol 6: 112-119.
- Ozdemir G, Karabay NU, Dalay MC, Pazarbasi B (2004) Antibacterial activity of volatile components and various extracts of Spirulina platensis. Phytother Res 18: 754-757.
- Ayehunie S, Belay A, Baba TW, Ruprecht RM (1998) Inhibition of HIV-1 replication by an aqueous extract of Spirulina platensis (Arthrospira platensis). J Acquir Immune Defic Syndr Hum Retrovirol 18: 7-12.
- Bhat VB, Madyastha KM (2000) C-Phycocyanin: A potent peroxyl radical scavenger in vivo and in vitro. Biochem Biophys Res Commun 275: 20-25.
- Miranda MS, Cintra RG, Barros SB, Filho MJ (1998) Antioxidant activity of the microalgae Spirulina maxima. Braz J Med Biol Res 31: 1075-1079.
- Pineiro Estrada JE, Bermejo Bescos P, Villar del Fresno AM (2001) Antioxidant activity of different fractions of Spirulina platensis protean extract. Farmaco 56: 497-500.
- Sánchez M, Bernal-Castillo J, Rozo C, RodrÃguez I (2003) Spirulina (Arthrospira): An edible microorganism: A review. Universitas Scientiarum 8: 7-24.
- Colla LM, Reinehr OC, Reichert C, Costa JA (2007) Production of biomass and neutraceutical compounds by Spirulina platensis under different temperature and nitrogen regimes. Bioresour technol 98: 1489-1493.
- Vijayarani D, Ponnalaghu S, Rajathivya J (2012) Development of value added extruded productusing Spirulina. Int J Health Sci Res 2: 42-47.
- Saranraj P, Sivasakthi S (2014) Spirulina platensis-food for future: A review. Asian J Pharm Sci Technol 4: 26-33.
- Ibanez E, Herrero M, Mendiola JA, Castro-Puyana M (2012) Extraction and characterization of bioactive compounds with health benefits from marine resources: Macro and micro algae, cyanobacteria, and invertebrates. In: Hayes M (ed), Marine bioactive compounds: Source, Characterization and Applications Springer, New York, USA pp. 55-59.
- Belay A (2008) Spirulina (Arthrospira): Productionand quality assurance. In: Gershwin ME, Belay A (eds) Spirulina in human nutrition and health. CRS press, Taylor and Francis group, New York, pp. 1-25.
- Yang L, Zhang LM (2009) Chemical structural and chain conformational characterization of some bioactive polysaccharides isolated from natural sources. Carbohydr polym 76: 349-361.
- Otles S, Pire R (2001) Fatty acid composition of chlorella and Spirulina microalgae species. J AOAC Int 84: 1708-1714.
- Pulz O, Gross W (2004) Valuable products from biotechnology of microalgae. Appl microbiol biotechnol 65: 635-648.
- McCarty MF (2007) Clinical potential of Spirulina as a source of phycocyanobilin. J Med Food 10: 566-570.
- Balch PA, Balch JF (2000) Prescription for nutritional healing. Avery, Health and Fitness pp. 776.
- Koru E (2012) Earth food Spirulina (Arthrospira): Production and quality standarts. INTECH Open Access Publisher.
- Grima ME, Belarbi EH, Acién Fernández FG, Medina RA, Chisti Y (2003) Recovery of microalgal biomass and metabolites: Process options and economics. Biotechnol Adv 20: 491-515.
- Olaizola M (2003) Commercial development of microalgal biotechnology: From the test tube to the market place. Biomol Eng 20: 459-466.
- Morist A, Montesinos JL, Cusido JA, Godia F (2001) Recovery and treatment of Spirulina platensis cells cultured in a continuous photobioreactor to be used as food. Process Biochem 37: 535-547.
- Rabelo SF, Lemes AC, Takeuchi KP, Frata MT, Monteiro de Carvalho JC, et al. (2013) Development of cassava doughnuts enriched with Spirulina platensis biomass. Braz J Food Technol 16: 42-51.
- Becker W (2004) Microalgae in human and animal nutrition. In: Richmond A (ed) Handbook of microalgal culture: Biotechnology and applied phycology. Blackwell science, Blackwell publishing company, UK pp. 312.
- Usharani G, Saranraj P, Kanchana D (2012) Spirulina cultivation: A review. Int J Pharm Biol Arch 3: 1327-1341.
- Markou G, Georgakakis D (2011) Cultivation of filamentous cyanobacteria (blue-green algae) in agro-industrial wastes and wastewaters: A review. Appl Energy 88: 3389-3401.
- Gerde JA, Wang T, Yao L, Jung S, Johnson LA, et al. (2013) Optimizing protein isolation from defatted and non-defatted Nannochloropsis microalgae biomass. Algal Res 2: 145-153.
- Khan Z, Bhadouria P, Bisen PS (2005) Nutritional and therapeutic potential of Spirulina. Curr Pharm Biotechnol 6: 373-379.
- Bonotto S (1988) Food and chemicals from microalgae. Prog Oceanography 21: 207-215.
- Manjit Kaur SD, Ahluwalia AS (1992) Biochemical studies on Spirulina protein. In: Spirulina ETTA National symposium MCRC pp.78-84.
- Mendiola JA, Jaime L, Santoyo S, Reglero G, Cifuentes A, et al. (2007) Screening of functional compounds in supercritical fluid extracts from Spirulina platensis. Food chem 102: 1357-1367.
- Chu WL, Lim YW, Radhakrishnan AK, Lim P E (2010) Protective effect of aqueous extract from Spirulina platensis against cell death induced by free radicals. BMC Comp Altern Med 10: 53.
- Sundararaman M, Averal HI, Akbarsha MA, Subramanian G (1994) Bio-activity of marine cyanobacteria in the animal-based systems: Modulation of food intake, body weight and some hematological characters. Anal Appl Biol 125: 195-206.
- Chamorro G, Salazar M, Favila L, Bourges H (1996) Pharmacology and toxicology of Spirulina alga. Rev Invest Clin 48: 389-399.
- Anitha L, Chandralekha K (2010) Effect of supplementation of spirulina on blood glucose, glycosylated hemoglobin and lipid profile of male non-insulin dependent diabetics. Asian J Exp Biol Sci 1: 36-46.
- Sarada R, Pillai MG, Ravishankar GA (1999) Phycocyanin from Spirulina sp: Influence of processing of biomass on phycocyanin yield, analysis of efficacy of extraction methods and stability studies on phycocyanin. Process Biochem 34: 795-801.
- Mendes RL, Nobre BP, Cardoso MT, Pereira AP, Palavra AF (2003) Supercritical carbon dioxide extraction of compounds with pharmaceutical importance from microalgae. Inorg Chim Acta 356: 328-334.
- Herrero M, Martin-Alvarez PJ, Senorans FJ, Cifuentes A, Ibanez E (2004) Optimization of accelerated solvent extraction of antioxidants from Spirulina platensis microalga. Food chem 93: 417-423.
- Sajilata MG, Singhal RS, Kamat MY (2008) Fractionation of lipids and purification of a α-linolenic acid (GLA) from Spirulina platensis. Food Chem 109: 580-586.
- Rechner AR, Spencer JP, Kuhnle G, Hahn U, Rice-Evans CA (2001) Novel biomarkers of the metabolism of caffeic acid derivatives in vivo. Free Radic Biol Med 30: 1213-1222.
- Colla LM, Bertolin TE, Costa JA (2004) Fatty acids profile of Spirulina platensis grown under different temperatures and nitrogen concentrations. Z Naturforsch C 59: 55-59.
- Nuhu AA (2013) Spirulina (Arthrospira): An important source of nutritional and medicinal compounds. J Mar Biol 2013: 8.
- Dasgupta T, Banejee S, Yadav PK, Rao AR (2001) Chemomodulation of carcinogen metabolizing enzymes, antioxidant profiles and skin and forestomach papillomagenesis by Spirulina platensis. Mol Cell Biochem 226: 27-38.