Research Article
Blighia sapida Aril Consumption at DegreeNeighboring 50% do not Affect Bone Growth of Rats, from 50%, Bone growth is Negatively Affected
Ouattara Howélé1*, Meite Alassane2, Soro Soronikpoho3 and Kati-Coulibaly Séraphin4
1Ufr Biologie, Université Péléforo Gon Coulibaly de Korhogo
2Laboratoire de Nutrition Pharmacologie, Ufr Biosciences, Université Félix HOUPHOUËT BOIGNY de Cocody-Abidjan
3Laboratoire de Biochimie et Sciences des Aliments, Ufr Biosciences, Université Félix HOUPHOUËT-BOIGNY de Cocody-Abidjan
4Ufr Biosciences, Université Félix HOUPHOUËT-BOIGNY de Cocody-Abidjan
Corresponding author: Ouattara Howélé, Ufr Biologie, Université Péléforo Gon Coulibaly de Korhogo, Tel: 22507544664; Email: ouattarahowele@gmail.com
Citation: Howélé O, Alassane M, Soronikpoho S, Séraphin KC. Blighia sapida Aril Consumption at Degree Neighboring 50% do not Affect Bone Growth 02 of Rats, from 50%, Bone growth is Negatively Affected. Indian J Nutri. 2016;3(2): 144.
Copyright © 2016 Ouattara Howélé et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Indian Journal of Nutrition | ISSN: 2395-2326 | Volume: 3, Issue: 2
Submission: 17/08/2016;; Accepted: 22/09/2016; Published: 30/10/2016
Abstract
The aim of this study was to understand the effects of Blighia sapida aril level in rat's diets in bones formation and its functionalities.
Quantities of Blighia sapida aril were cooked (500 g/l) and lyophilize. Six homogeneous groups of young Albinos wistar rats were fed ad libitum with six different diet which can be distinguished by the level of Blighia sapida aril powder, control diet (0% Blighia sapida arils powder), Bs1 (6.25% Blighia sapida arils powder), Bs2 (12.5% Blighia sapida arils powder), Bs3 (25% Blighia sapida arils powder), Bs4 (50% Blighia sapida arils powder) and Bs5 (75% Blighia sapida arils powder). At the end of the experimentation, blood sample and urine were collected and analysed. Also, at the end of the experimentation thighbones and shinbones wereremoved and then bone length, thickness and volume were measured. Ash, calcium and phosphorus of the bones collected were measured after incineration. Results show that that it was no difference on the variation (p>0.05) of bone mass, bones ashes mass, bone volume, bone calcium, bone phosphorus, bone length and bone thickness between values obtained on rats fed with the control diet and that of rats fed with the experimental diets (diet Bs1, diet Bs2 and dietBs3). Nevertheless, it was observed a diminution (p<0.05) of these parameters on rats fed with diet Bs4 and diet Bs5 compared to that observed on rats fed with the control diet.
The result of this study showed that Blighia sapida aril is nutritiously rich but must be consumed with moderation in order to improve bone growth and its functionalities because of the amino-acids content of the protein parts.
Keywords: Blighia sapida aril; Diet; Albinos wistar rats; Bone formation and functionalities
Introduction
Bone was for a long time considered as an organ inertbiologically. With time and technological evolution, scientists haveunderstood that bone is an assembling of living cells endowed withvarious functions essential for the life of the entire organism. Bone grows, it is constantly renewed and is the organism support [1,2]. In bone, it can be distinguished osteoclast cells which are responsible for bone resorption [1], and osteoblast cells which are responsiblefor bone formation [2]. Bone is a weft constituted by a deposit of calcium and phosphorus. Bones contain 99% of the organism calcium and phosphorus reserves. Inside the bone, there is bone marrow where is synthetizing blood cells. Then bone marrow is the center of hematopoiesis. Calcium, vitamin D and proteins are the magic threesome of nutriments which maintained homeostasis of bone [3-5]. The necessary and sufficient condition for bone to play its role of synthesis and organ support is realized when alimentation contain allthe important nutriments which are carbohydrates, proteins, lipids,ash and vitamins [6]. Under normal circumstances, when there is no lack of food, bone nutritional needs are fulfilled [7]. Natural disasters such as flood and drought unfortunately cause lack of food which situation brings people to add to their eating patterns fruits of wild trees in order to resist. It is the case of Blighia sapida aril insertion in alimentation, in Côte d'Ivoire (1984) when there was famine [8] even if morbidity was observed when consumed unripe, and in Jamaica (2002) when there was flood [9]. This contributed to avoid a high andlong famine. Population found in Blighia sapida aril a palliative to thelack of food.
Results of previous studies talking about the impact of Blighia sapida aril consumption on young Albinos rats growth and their wellbeing showed that Blighia sapida aril could not be exclusively used as basis of nutritional source [10]. However, the consumption of Blighia sapida aril with moderation can improve growth whereas high level of its consumption provokes growth retardation of young rats and raises total cholesterol and LDL cholesterol [10].
In this work we relate the effects of Blighia sapida aril level in rats diets on bone formation and its functionalities.
Material and Methods
Material
Plant material: Blighia sapida is planted in the North and the Center of Côte d'Ivoire. Arils are consumed in these regions. Arils used for the experimentation were brought from Katiola, a town in the North center of Côte d'Ivoire, in the period of March, April, and May when they are more available. They were spread on polythene paper and exposed to sunlight for two weeks, six hours a day to get dry. In the night, they were kept on the plastic and put in a house at room temperature (25 °C-30 °C). After the drying process of the fresh aril, the dried arils were kept in plastic bags and sent to Abidjan (South of Côte d'Ivoire), the biggest town of Côte d'Ivoire, where the experimentation were made.
Animals: 36 young albino Wistar rats weighing between 49 g and 75 g were used. They were bred in the animal house of Ufr Biosciences of the University Félix HOUPHOUËT-BOIGNY of Abidjan (Côte d'Ivoire). During the breeding, rats were fed with food made by a society in Côte d'Ivoire call IVOGRAIN which is specialized in mass production of livestock food. This food is making up by crud protein matter (15%), crud fat matter (3.5%), cellulose matter (12%), mineral matter (9%), calcium (1%), phosphorus (0.9%), sodium (0.3%), vitamin A (15000 ui/kg), vitamin D3 (3000 ui/kg), vitamin E (10 mg/ kg).
Methods
Preparation of the arils: In Abidjan, the dry arils were crushed with a mortar. Each time, 500 g of Blighia sapida arils powder were cooked in one liter of water (500 g/l) using a gas stone during two hours. Arils pasta obtained were lyophilized. The powder obtained was used to formulate five kind of experimental diet call diet Bs1, diet Bs2, diet Bs3, diet Bs4 and diet Bs5. Previously, a control diet was prepared. The composition of the control diet was similar to that recommended to scientist by the American Institute of Nutrition for rapid Growth (AIN-93G) when they want to make experience with animals, particularly rats [11].
Difference between experimental diets
In experimental diets, a part of the control diet was substituted byBlighia sapida arils powder.
The degree of substitution was 6.25% for diet Bs1, 12.5% for diet Bs2, 25% for diet Bs3, 50% for diet Bs4 and 75% for diet Bs5.
Experimentation
At the beginning of the experimentation, the animals were teamedin six homogeneous young groups of rats. They were put individuallyin metabolic cages and maintained under standard laboratory conditions (temperature 25±2 °C) with dark and light cycle (12h/12h). In the metabolic cages, rats were acclimatized to this condition and fed with the control diet five days before the commencement of the experiment. After that, they were fed ad libitum with the different diets (control diet, diet Bs1, diet Bs2, diet Bs3, diet Bs4 and diet Bs5) according to the different group of animals during 28 days.
Biochemical parameters
At the end of the experimentation, blood sample was collected at the vena cava level of all animal and put in individually vacuum valve. The blood samples were centrifuged at 8000 r.p.m for 15 min to harvest the plasma which was used for the various analyses. Serum samples were used for analyses of total protein, plasmatic calcium and phosphorus using appropriate kit and an automatic chemistry analyzer (Hitachi model 902, Roche).
Also, at the end, urine of 24 hours of each animal was collected and adds up to that of the group. In each quantity obtained urinary protein, urinary calcium and urinary phosphorus were measured after been centrifuged using appropriate kit and an automatic chemistry analyzer (Hitachi model 902, Roche).
Hematological parameters
Also, blood sample was collected at the vena cava level of each animal and put in individually tube containing Ethylene Diamine Tetra Acetic (EDTA). These blood samples were used for full blood count. Then, hematological parameters including hemoglobin content, total count of red blood cells (RBC) and white blood cells (WBC), differential count of leukocytes such as granulocyte (%), lymphocyte (%), monocyte (%), hematocrit (Hct), mean cell volume (MCV), mean corpuscular hemoglobin concentration (MCHC) and platelet count were measured using an automatic hematological analyzer (Symex-kx-21N).
Bone treatment
At the end of the experimentation, after blood sample was taken, all animals were sacrificed. Then, thighbones and shinbones were removed. They were measured fresh and dry after being dry in an electric oven at 100 °C during 24 hours. Thighbones length, shinbones length and their thickness were measured with a calliper rule. These bones obtained were used to evaluate bone metabolism.
- Bone volume: Thighbones and Shinbones volume weremeasured thank to Archimedes principle. Test tube was filled with distilled water. Add bone in the distilled water, provokes it level in the tube. The difference of level between the second position and the first position constituted the bone volume.
- Determination of ash, calcium and phosphorus rate :Thighbones and Shinbones put individually in jar were incinerated in an electric oven at 6500°C during four hours [12]. Ashes obtain were collected and weighed.
Statistical analysis
The experimental results were expressed as the mean±S.E.M. Data were assessed by the method of analysis of ANOVA followed by Dunnett test [13,14]. p value of < 0.05 was considered as statistically significant.
Result
Effect of Blighia sapida aril powder level on plasmaticcalcium and phosphorus
Consumption of Blighia sapida aril powder at different level, during 28 days, does not provoke significant variation (p>0.05) on plasmatic calcium and phosphorus. Values of calcium obtained were 71.33±1.75 mg/ml for control group of rats, 72.50±4.55 mg/ml for group of rats fed with diet Bs1, 67.50±3.39 mg/ml for group of rats fed with diet Bs2, 68.33±4.93 mg/ml for group of rats fed with diet Bs3, 69.67±1.03 mg/ml for group of rats fed with diet Bs4 and 65.50±1.29 mg/ml for group of rats fed with diet Bs5. Those of phosphorus were 39.00±1.41 mg/ml for control group of rats, 36.33±3.33 mg/ml for group of rats fed with diet Bs1, 39.50±4.18 mg/ml for group of rats fed with diet Bs2, 35.83±3.19 mg/ml for group of rats fed with diet Bs3, 38.17±1.21 mg/ml for group of rats fed with diet Bs4 and 38.50±1.29 mg/ml for group of rats fed with diet Bs5.These results are shown on Figure 1.
Effect of Blighia sapida aril powder level on plasmatic protein
Plasmatic protein measured on rats fed with the different dietswere 0.62±0.04 g/l, 0.59±0.02 g/l, 0.65±0.05 g/l and 0.62±0.05 g/l for group of rats fed with diet Bs1,diet Bs2, diet Bs3, diet Bs4 and diet Bs5 respectively. These rates were not significantly different (p>0.05) from that measured on rats fed with the control diet which was 0.67±0.02 g/l. These results are shown on Figure 2.
Effect of Blighia sapida aril powder level on urinary calciumand phosphorus
Urinary calcium and phosphorus measured on rats fed with diet Bs1, diet Bs2, diet Bs3, diet Bs4 and diet Bs5 were not significantly different (p>0.05) from that measured on rats fed with the control diet.
Values of calcium obtained were 47.00±4.15 mg/ml for control group of rats, 48.00±4.05 mg/ml for group of rats fed with diet Bs1, 45.00±3.27 mg/ml for group of rats fed with diet Bs2, 46.00±3.89 mg/ ml for group of rats fed with diet Bs3, 47.00±3.95 mg/ml for group of rats fed with diet Bs4 and 44.00±3.23 mg/ml for group of rats fed with diet Bs5. Those of phosphorus were 33.00±3.22 mg/ml for control group of rats, 34.00±3.47 mg/ml for group of rats fed with diet Bs1, 35.00±3.19 mg/ml for group of rats fed with diet Bs2, 32.00±4.02 mg/ ml for group of rats fed with diet Bs3, 33.00±3.98 mg/ml for group of rats fed with diet Bs4 and 32.00±2.98 mg/ml for group of rats fed with diet Bs5. These results are shown on Figure 3.
Effect of Blighia sapida aril powder level on urinaryprotein
Urinary protein measured on rats fed with the different diets were0.16±0.05 g/l, 0.16±0.07 g/l, 0.16±0.06 g/l, 0.15±0.04 g/l, 0.13±0.04 g/l and 0.12±0.05 g/l for group of rats fed with control diet, diet Bs1, diet Bs2, diet Bs3, diet Bs4 and diet Bs5 respectively. There were any significant different of urinary rate (p>0.05) between rats fed with diet Bs1, diet Bs2 and diet Bs3 when each mean value are compared to the one obtained on rats fed with the control diet while a significant difference (p<0.05) was observed on rats fed with diet Bs4 and diet Bs5 compare to the value obtained on rats fed with the control diet. These results are shown on Figure 4.
Effect of Blighia sapida aril powder level on bone mass
The average values of bone mass measured on rats fed with diet Bs1, diet Bs2 and diet Bs3 were not significantly different (p>0.05) from that measured on rats fed with the control diet. On the other hand, the average values of bone mass obtained on rats fed with diet Bs4 and diet Bs5 were very significantly different (p<0.01) and very highly significantly different (p<0.001) respectively from that obtain on rats fed with the control diet. Investigation made indicate bone mass averages values of 1.00±0.29 g, 1.00±0.31 g, 0.99±0.30 g, 1.02±0.31 g, 0.72±0.06 g and 0.64±0.06 g for rats fed with control diet, diet Bs1, diet Bs2, diet Bs3, diet Bs4 and diet Bs5 respectively. These results are shown on Figure 5.
Effect of Blighia sapida aril powder level on thighbonesand shinbones length
Consumption of Blighia sapida aril powder at degrees of 6.25%(Bs1), 12.50% (Bs2) and 25% (Bs3), during 28 days, do not provoke significant variation (p>0.05) on thighbones length and shinbones length. On the other hand, consumption of Blighia sapida aril powder at degrees of 50% (Bs4) and 75% (Bs5) provoke respectively a very significant reduce (p<0.01) and very highly significantly reduce (p<0.001) of thighbones length and shinbones length when compared to that obtained on rats fed with the control diet. Values of average thighbones length obtained were 3.33±0.05 cm for control group of rats, 3.40±0.08 cm for group of rats fed with diet Bs1, 3.37±0.05 cm for group of rats fed with diet Bs2, 3.33±0.05 cm for group of rats fed with diet Bs3, 2.95±0.08 cm for group of rats fed with diet Bs4 and 2.05±0.10 cm for group of rats fed with diet Bs5. Those of shinbones length were 4.42±0.07 cm for control group of rats, 4.30±0.11 cm for group of rats fed with diet Bs1, 4.35±0.13 cm for group of rats fed with diet Bs2, 4.45±0.15 cm for group of rats fed with diet Bs3, 3.85±0.06 cm for group of rats fed with diet Bs4 and 3.18±0.07 cm for group of rats fed with diet Bs5. These results are shown on Figure 6.
Effect of Blighia sapida aril powder level on thighbonesand shinbones thickness
Rats fed with diets containing 6.25% of Blighia sapida aril powder(Bs1), 12.50% of Blighia sapida aril powder (Bs2), 25% of Blighia sapida aril powder (Bs3) and 50 % of Blighia sapida aril powder (Bs4) had average values of thighbones thickness and shinbones thickness which were not significantly different (p>0.05) to these values obtained on rats fed with the control diet. Only rats fed with the diet containing 75% of Blighia sapida aril powder (Bs5) had average values of thighbones thickness and shinbones thickness significantly reduced (p<0.05) compared to these values obtain on rats fed with the control diet.
Values of average thighbones thickness obtained were 0.33±0.04cm for control group of rats, 0.33±0.06 cm for group of rats fed with diet Bs1, 0.34±0.06 cm for group of rats fed with diet Bs2, 0.33±0.06 cm for group of rats fed with diet Bs3, 0.31±0.03 cm for group of rats fed with diet Bs4 and 0.24±0.04 cm for group of rats fed with diet Bs5. Those of shinbones thickness were 0.26±0.04 cm for control group of rats, 0.25±0.04 cm for group of rats fed with diet Bs1, 0.26±0.04 cm for group of rats fed with diet Bs2, 0.24±0.04 cm for group of rats fed with diet Bs3, 0.23±0.04 cm for group of rats fed with diet Bs4 and 0.18±0.05 cm for group of rats fed with diet Bs5. These results are shown on Figure 7.
Effect of Blighia sapida aril powder level on bones ashesmass
The average values of bone ashes mass of rats fed with diet Bs1, diet Bs2 and diet Bs3 were not significantly different (p>0.05) from the average value of bone ash mass of rats fed with the control diet. On the other hand, the average values of bone ashes mass obtained on rats fed with diet Bs4 and diet Bs5 were very significantly reduced (p<0.01) and very highly significantly reduced (p<0.001) than that obtained on rats fed with the control diet. Investigation made show thighbones ashes mass averages values of 0.28±0.01 g, 0.27±0.02 g, 0.27±0.02 g, 0.27±0.02 g, 0.17±0.01 g and 0.11±0.01 g for rats fed with control diet, diet Bs1, diet Bs2, diet Bs3, diet Bs4 and diet Bs5 respectively. Those of shinbones ashes mass average were 0.27±0.01 g for control group of rats, 0.27±0.01 g for group of rats fed with diet Bs1, 0.27±0.01 g for group of rats fed with diet Bs2, 0.27±0.01 g for group of rats fed with diet Bs3, 0.16±0.01 g for group of rats fed with diet Bs4 and 0.11±0.01 g for group of rats fed with diet Bs5. These results are shown on Figure 8.
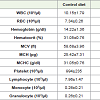
Effect of Blighia sapida aril powder level on hematological parameters
There was no significant change (p>0.05) in the number of white blood cells, granulocytes (%), lymphocytes (%), monocytes (%), the number of red blood cells, hemoglobin, hematocrit (Hct), mean cell volume (MCV), mean corpuscular hemoglobin (MCH), mean corpuscular hemoglobin concentration (MCHC) and platelet whichever the diet consumed. These results are shown on Table 1.
Discussion
The decrease of bone mass, bones ashes, calcium and phosphoruscontent on rats which consumed diets Bs4 and Bs5 compare to that of rats which consumed control diet and the other experimental diets are the consequence of bone acquiring lateness. This bone acquiring lateness can be due to the insufficiency of protein in diet Bs4 and diet Bs5 [15,16]. In fact, according to the analysis carried out by Ouattara et al. [17], Blighia sapida aril protein content is lower than that recommended by the Food Agriculture Organization [18]. Then, the mixture of control diet with Blighia sapida aril powder corresponds to a decreasing of protein content of the diet make up. This decreasing of protein content is more and more important with the augmentation of Blighia sapida aril powder proportion. The inadequacy of protein content in diet Bs4 and diet Bs5 may be very high so that it reduced osteoblasts cells activities and then bone acquiring capital [19]. All these assertions may explain the diminution of bone mass, thighbonesand shinbones length, thighbones and shinbones thickness and bones ashes mass. However, plasmatic protein measured on rats fed with the different diets did not show any difference (p>0.05) between each other. Therefore it is not the protein content of diet Bs4 and diet Bs5 wihich must be incriminated in the bone growth retardation rather than the diversity of essential amino-acids found in the protein part of Blighia sapida aril.
The fact that hematological parameters measured on rats fed withthe different diets were not significantly different (p>0.05) from eachother, show that hematopoietic factors such as iron content, vitaminB12 content, vitamin B9 content and folic acid content were sufficientto insure a normal hematopoiesis [20-22].
Conclusion
Blighia sapida aril consumption at degree neighboring 50% indaily nutritional need did not have a negative effect on bone growth of rats fed with these diets compared to the bone growth of rat fed with the control diet free from Blighia sapida aril. However, from a degree of 50% in daily nutritional need, the bone growth is negatively affected with a diminution of bone acquiring capital of rats fed with these diets. This diminution is certainly due to the weakness of essential amino-acids in Blighia sapida aril.
It will be necessary to analyze the protein fraction of Blighia sapida aril in order to know the amino-acids content of this fraction.
Acknowledgment
The author, OUATTARA Howélé, is grateful to the Unity of formation and Research (UFR) biosciences of University of Félix HOUPHOUËT-BOIGNY for permitting us to work in their laboratories.
References
- Baron R (2001) L’ostéoporose et les mécanismes moléculaires de la résorption osseuse. Médecine/Sciences 17: 1260-1269.
- Marie P (2001) Differentiation, function and control of osteoblast. Médecine/Sciences 17: 1252-1259.
- Bertrière MC (2003) Prévention nutritionnelle de l’ostéoporose. Objectif nutrition 69: 3-9.
- Amling M (2015) Clacium and vitamin D in bone metabolism : Clinical importance for fracture treatment. Unfallchirurq 118: 995-999.
- Friedman MA, Bailey AM, Rondon MJ, MC Nerry EM, Sahar ND, et al. (2016) Calcium and phosphorus-Supplemented Diet Increase Bone mass after short-term Exercise and increase Bone mass and Structural Strength after long-Term Exercise in Adult Mice. Plos One 11: e0151995.
- International Osteoporosis Foundation (2006) Une nourriture saine pour des os sains.
- Burher E (2015) La santé osseuse chez l’enfant : mesures de préventions primaires. Humann nutrition, schulstrsse 38, 8451 Kleinandelfingen.
- ngble (1986) Organisation Panamericaine de la Santé. Empoisonnement par consommation d’ackee(Blighiasapida) dans le Département du Nord, Haïti. Vol 22, No2.
- Ouattara H, Meite A, Kouame P, Kati-Coulibaly S (2016) Impact of Blighia sapida (k. Koenig) aril powder level in diet on the growth and the well-being of albino Wistar rats. African Journal of Food Sciences 10: 164-171.
- Reeves PG, Nelsen F H, Fahey GCJ (1993) Purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr 123: 1939-1951.
- Bar A, Shinder D, Sara Y, Vax E, Plavnik I (2003) Metabolism and requirements for calcium and phosphorus in the fast-growing chicken as affected by age. Br J Nutr 89: 51-61.
- Ostle B (1966) Statistics in Research. Iowa state university press. Iowa, USA 310-361.
- Woolson RF (1987) Statistical methods for the Analysis of Biomedical Data. John Wiley and Sons Inc., New York.
- Kerstetter JE, Diane E, Wall KO, OʼBrien, Donna M, et al. (2005) Meat and soy protein affect calcium homeostasis in healthy women. J Nutr 136: 1890-1895.
- Kerstetter JE, O'Brien KO, Caseria DM, Wall DE, Insogna KL (2005) The impact of dietary protein on calcium absorption and kinetic measures of bone turnover in women. J Clin Endocrinol Metab 90: 26-31.
- Ouattara H, BobeleNF, Dally T, Kati-coulibaly S (2010) Nutritional composition studies of Blighia sapida aril from Côte d’Ivoire. Journal of Applied Biosciences 32: 1989-1994.
- Food Agriculture Organisation (2003) Evolution des graisses alimentaires et évolution de leur consommation. Laboratoire de nutrition tropicale, centre ORSTOM 34032, Montpellier cedex, France.
- Alan SA, James L, Barbara Y (2009) Anatomie pathologique. Synthèse du tissu osseux. Atlas DE Wheater p.258.
- Hoffbrand AV, Herbert V (1999) Nutritionl anemia. Semin Hematol 36 (4Suppl 7): 13-23.
- Wickramasinghe SN (2006) Diagnosis of megaloblastic anemias. Blood Rev 20: 299-318.
- Carmel R (2008) How I treat Cobalamin (vitamin B12). Blood 112: 2114-2221.
- Merto CM, Wuillemin WA (2009) Diagnosis and therapy of anemia in general practice. Praxis (Bem 1994) 98: 191-199.