Research Article
Comparative Efficacy and Safety of Fosfomycin and Nitrofurantoin in Acute Uncomplicated Lower Urinary Tract Infection in Young Women A Randomized, Open Label, Comparative Study
Anita K*, Razdan kaul A, Dhanesha G and Priyambada U
Department of Obstetrics and Gynaecology, Asian Institute of Medical Sciences, India
*Corresponding author: Anita K, Department of Obstetrics and Gynaecology, Asian Institute of Medical Sciences, Sector 21A, Faridabad, Haryana, India
Article Information: Submission: 02/12/2019; Accepted: 31/12/2019; Published: 03/01/2020
Copyright: © 2019 Anita K, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Aim: The aim is to evaluate the comparative efficacy and safety of fosfomycin and nitrofurantoin in acute uncomplicated lower urinary tract infection in
young women in our study cohort.
Material and methods: A randomized open label, intention to treat, comparative study was conducted over period of two years. Women were prescribed
either nitrofurantoin or fosfomycin and their final outcomes in term of clinical and bacteriological cure were compared.
Results: Both the drugs were found to be equally effective in treating UTI with comparable safety and tolerability.
Keywords
Urinary tract infection, Fosfomycin; Nitrofurantoin
Introduction
Acute uncomplicated lower Urinary Tract Infection (UTI) in an
otherwise healthy, non-pregnant woman is one of the most common
problems for which young women seek medical attention. More
than 30% of all women will experience UTI during their lifetime and
the prevalence of UTI in women is approximately 50 times higher
than in men. Antimicrobial resistance among uropathogens causing
community-acquired UTIs is increasing worldwide.
The use of fosfomycin and nitrofurantoin are returning, owing to
their broad-spectrum activity against both gram-positive and gramnegative
bacteria. Fosfomycin is a unique antibiotic that is chemically
different from any other known antibacterial agent. Nitrofurantoin is
bactericidal.
Materials and Methods
A randomized open label, intention to treat, comparative study was conducted in the department of Obstetrics and Gynecology,
Asian institute of medical sciences, Faridabad over a period of two
years from June 2017 to May 2019.The study was undertaken after
prior approval from the institutional ethics committee.
Young females diagnosed with uncomplicated UTI were
randomized to receive either 100 mg capsules of nitrofurantoin twice
a day for 7 days or a 3 gms single dose of fosfomycin. The diagnosis
of uncomplicated UTI was based on clinical history, physical
examination, urine analysis and urine culture with susceptibility
testing. The clinical and bacteriological response was compared
between the patients in the two treatment groups after ten days.
Results
70 women with uncomplicated UTI attending outpatient
department formed the subject matter for this study. 35 study subjects
were assigned to each group with one group having nitrofurantoin
treatment and other having fosfomycin. Relevant finding were recorded in specially designed pro forma including age, complaints,
past history, drugs use to treat UTI, relevant side effects and response
to drugs.
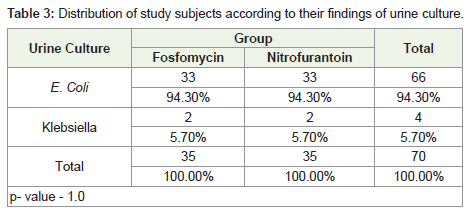
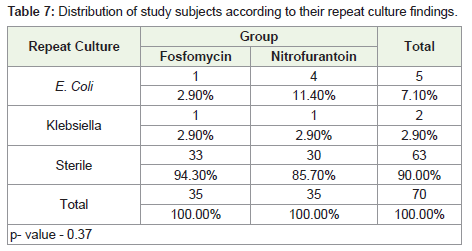
Urine culture analysis revealed growth of E. coli in 94.3% of the
patients in both the treatment groups. Rest of the patients had growth
of Klebsiella. Both the groups were similar in terms of the urine
culture growth results (p value = 1.0).
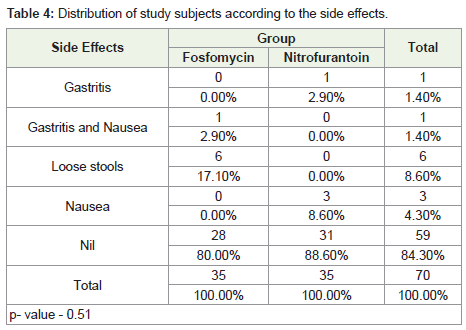
Seven patients from the Fosfomycin group (20%) reported side
effects, while only four patients in the Nitrofurantoin group.
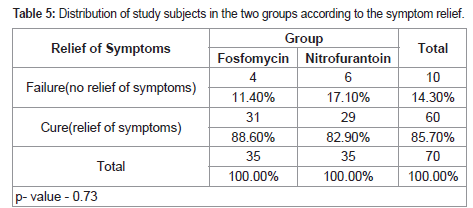
14.3% of the women had no relief of symptoms. The two study
drugs were not different statistically with respect to their relief in
symptoms (p value =0.73)
Repeat culture results revealed that 94.3% of the patients in the
Fosfomycin group had sterile urine, while 85.7% in the Nitrofurantoin
group had sterile urine. The study groups were not statistically
different with respect to their findings of repeat urine culture result
(p value = 0.37) (Table 1-8).
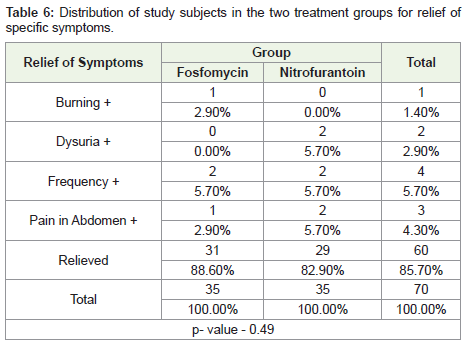
Table 6: Distribution of study subjects in the two treatment groups for relief of
specific symptoms.
In the Fosfomycin group, only two patients (5.7%) required
further treatment, while in the Nitrofurantoin group four patients
(11.4%) required further treatment. The difference is not statistically
significant (p=0.63).
Discussion
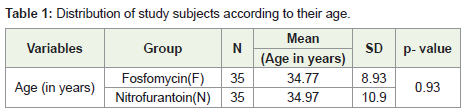
In the present study, both the treatment groups included 35
patients each. Mean age of cases in the Fosfomycin group was 34.77
± 8.93 years and of the Nitrofurantoin group was 34.97 ± 10.90 years.
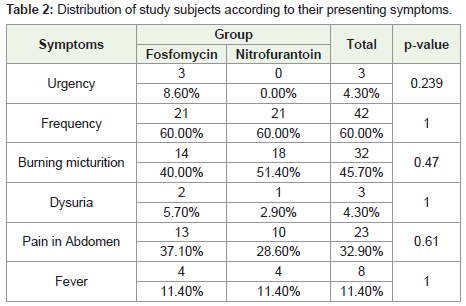
The most common complaint of the patients in our study
was frequency, reported by 60% of the patients in both the study
groups. The next common complaint was burning micturition,
which was observed in 40% in the Fosfomycin group and 51.4% in
the Nitrofurantoin group. Pain in the abdomen was the next most
common complaint in both the treatment groups. It was reported
by 37.1% in the Fosfomycin group and 28.6% in the Nitrofurantoin
group. Less common complaints were urgency, fever and dysuria.
Huttner and colleagues did not report specific clinical complaints
of their patients, but the median number of symptoms was 3, ranging
from 2 to 4 [1]. We have defined clinical improvement as cure
(complete resolution of symptoms and signs of UTI without prior
failure), failure (need for additional or change in antibiotic treatment
due to a UTI or discontinuation due to lack of efficacy or persistence
of symptoms). Stein defined clinical response as cure (elimination of
all prê therapy symptoms), improvement (most but not all symptoms
improved or absent), or failure (not improved from the initial
assessment) [2]. In both the treatment groups in their study, 87% of
the patients had symptoms for less than 48 hours.
In the present study, 74.3% of the patients in the Fosfomycin
group and 80% of the patients in the Nitrofurantoin group had 10 to
20 pus cells. Nagel et al. conducted a retrospective study to evaluate
the clinical and economic outcomes of fosfomycin compared to
matched controls for the treatment of complicated lower tract and
uncomplicated UTI [3]. In their study, 68% of the patients receiving
Fosfomycin had more than 10 pus cells, which is similar to our study.
Baseline urine culture analysis in our study revealed E. coli
growth in 94.3% of the patients in both the treatment groups. Rest
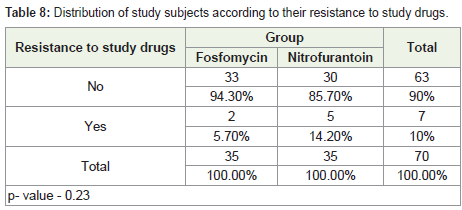
of the patients had Klebsiella growth. Furthermore, 2 patients in the
Fosfomycin group and 5 cases in the Nitrofurantoin group were
resistant to drugs. In the study by Huttner et al., of the 487 baseline
urine cultures obtained, 377 (77%) were positive, among which, E. coli (61%), Klebsiella spp (7%),
Enterococcus spp (7%), and Proteus spp (5%) predominated [1].
However, in our study only E. coli and Klebsiella predominated. In
the Nitrofurantoin group, 1% were resistant to Nitrofurantoin, 23%
resistant to co-trimoxazole, 12% resistant to fluoroquinolone and 6%
were Extended Spectrum Beta-Lactamase (ESBL) producers.
Similar to our study,in the study by Stein et al., the predominant
pretreatment uropathogen in each group was E. coli [2]. Next most
common isolates were Proteus mirabilis, Klebsiella pneumoniae, and
Staphylococcus saprophyticus.
Overall, 94% of pretreatment isolates were susceptible to
fosfomycin compared with 83% for nitrofurantoin. Of the E. coli isolates, 99% were susceptible to fosfomycin and 90% were
susceptible to Nitrofurantoin. Similarly, in our study, 94.3% isolates
were susceptible to Fosfomycin and 85.7% were susceptible to
nitrofurantoin.
In the present study, seven patients from the Fosfomycin
group (20%) reported side effects, while only four patients in
the Nitrofurantoin group (12%) reported any side effects. In the
Fosfomycin group, one reported gastritis and nausea, and six patients
reported loose stools. In the Nitrofurantoin group, one patient
reported gastritis and three reported nausea.
Whereas, in the randomized trial by Huttner, 21 of 248 (8%) and 16
of 247 (6%) in the nitrofurantoin and fosfomycin groups, respectively,
reported at least one adverse event.1 Stein and colleagues found that
the most common adverse effects related to fosfomycin treatment were
diarrhea (2.4%), vaginitis (1.8%), and nausea (0.8%) [2]. Common
side effects associated with nitrofurantoin treatment were nausea
(1.6%), vaginitis (1.6%), dizziness (0.8%), and diarrhea (0.8%). GI
adverse events were reported in 3.9% of fosfomycin recipients among
women with an uncomplicated lower UTI. Moreover, in a large noncomparative
study in 387 women and men with uncomplicated UTIs
who received single-dose fosfomycin, adverse events were reported
in 4.9 % of patients, with diarrhea, nausea and vomiting occurring in
3.1, 1.3 and 0.5 % of patients, respectively.
We observed that in the Fosfomycin group, 88.6% of the patients
reported relief of symptoms, while in the Nitrofurantoin group 82.9%
reported relief of symptoms.
Four patients in the Fosfomycin group did not report relief of
symptoms, one with burning micturition and pain in abdomen each
and two with frequency. In the Nitrofurantoin group, six patients did
not report relief of symptoms, two each of dysuria, frequency and
pain in the abdomen.
Stein et al. reported that the clinical cure rates were similar with
Fosfomycin and Nitrofurantoin at each of the follow-up assessment
[2]. Which is comparable to our study. P value 0.73 which is
statistically insignificant. At the end of the study, the overall success
(cure and improvement) rates were 80% in both the fosfomycin and
nitrofurantoin groups. Huttner et al. reported that at 28 days after
therapy completion, 171 of 244 patients (70%) receiving nitrofurantoin
had maintained clinical resolution versus 139 of 241 (58%) receiving
fosfomycin (difference, 12% [95% CI, 4%-21%]; P = 0.004) [1]. Clinical
response at the earlier point of 14 days after therapy completion also
differed significantly between groups, with 184 of 247 patients (75%)
receiving nitrofurantoin experiencing clinical resolution vs 162 of
247 (66%) receiving fosfomycin (difference, 9% [95% CI, 1%-17%]; P
= 0.03). Patients in either treatment group with early clinical failure
(due to the persistence of symptoms rather than recurrence after
initial improvement) returned for additional antibiotic therapy at the same point after inclusion (mean [SD] of 6.3 [3.8] and 6.5 [3.6] days in
the nitrofurantoin and fosfomycin groups, respectively). In our study
also, two patients in the Fosfomycin group and four patients in the
Nitrofurantoin group required further treatment with antibiotics and
the difference is not statistically significant (0.63) Similarly, another
double-blind, placebo-controlled trial by comparing a single 3-g dose
of fosfomycin vs 7 days of nitrofurantoin, 50 mg 4 times daily, in
women with clinically confirmed cystitis also found no difference in
clinical response at any time point 4 to 42 days after starting treatment
as per-protocol analyses which is in tandem to our study [4].
We found that the bacteriological cure rate in the Fosfomycin
group was 94.3%, while it was 85.7% in the Nitrofurantoin group.
Among the patients in the Fosfomycin group, one patient had E. coli and Klebsiella growth each. In the Nitrofurantoin group, four patients had growth of E. coli and one patient had Klebsiella growth.
However, contrary to our study, Huttner et al. reported that
patients receiving nitrofurantoin had significantly more bacteriologic
success: among those with positive baseline cultures, 146 of 177 (82%)
and 121 of 165 (73%) saw no recurrence on day 14 in the nitrofurantoin
and fosfomycin groups, respectively (P =.04).68 The difference
remained at day 28, when both groups saw an overall decrease in
success, with 129 of 175 (74%) and 103 of 163 (63%), respectively
(difference, 11% [95% CI, 1%-20%]; P =.04). Stein et al. reported
a statistically higher failure rate with fosfomycin 5 to 10 days after
the initial treatment dose which is in contrast to our study in which
failure rates with Fosfomycin were less compared to nitrofurantoin. 2
The bacteriologic cure rates were not statistically different at any of
the subsequent follow-up visits. The overall bacteriologic cure rate
was 60% for fosfomycin and 59% for nitrofurantoin.
Gupta et al. conducted a study which included non pregnant
women aged 18-45 years with acute uncomplicated cystitis, pyuria
and a positive urine culture [5]. The women received a single dose
of fosfomycin trometamol 3 gm (n = 20), ciprofloxacin 250 mg
twice daily for 3 days (n = 25) or nitrofurantoin 100 mg twice daily
for 7 days (n = 17), and clinical success rates and bacteriological
eradication rates were more than 90 % in all three treatment groups.
At the enrolment visit, 94 % of women had rectal colonization with
E. coli; the prevalence of rectal E. coli was significantly (p < 0.001)
reduced after treatment with fosfomycin and ciprofloxacin, but not
nitrofurantoin. This finding is also different from our study. Isolation
of two ciprofloxacin-resistant rectal E. coli strains from a single patient
occurred 10 days after completing treatment with ciprofloxacin.
Among patients receiving fosfomycin or nitrofurantoin, all isolated
rectal E. coli strains were susceptible to the study drug.
Furthermore, we observed that on subsequent urine culture after
10-14 days of treatment, only two patients in the fosfomycin group
(5.7%) had resistance to the study drugs, while in the Nitrofurantoin
group, five patients (14.2%) reported resistance.
Shah et al. found that out of 97 Multi-Drug Resistant (MDR) gram-negative urinary isolates, multi-drug resistance in E. coli was predominant (44.5%) as compared to K. pneumoniae (2%). In their study, 98% of MDR E. coli isolates were susceptible to fosfomycin and 81% to nitrofurantoin. These results coincided with a study conducted in Taiwan against MDR E. coli, where 95.5% and 75.1% isolates were susceptible to fosfomycin and nitrofurantoin respectively which is similar to our study. Bano et al. concluded maximum susceptibility to fosfomycin (100%) and nitrofurantoin (100%) against K. pneumoniae in a study conducted in Pakistan.
Few limitations of this study. First, it was conducted as an openlabel study, which might have introduced some level of measurement bias. However, interestingly Fosfomycin which was given as a single dose relieved symptoms in a higher number of patients as compared to Nitrofurantoin. Huttner et al. commented that openlabel design may influence the patient reported clinical outcomes [1]. A perception of greater treatment intensity (twice a day dose for 7 days) can differentially reduce the frequency of clinical failure in the nitrofurantoin as compared to that in the fosfomycin group, second, clinical response was patient-reported and could have introduced a bias. Lastly, this was a single center study. Epidemiology of uncomplicated UTI might vary at other geographical locations, where the clinical and bacteriological response to Fosfomycin and Nitrofurantoin might change. So the results of the study might not be applicable to other geographical locations. Also, different dosages of Nitrofurantoin are used in different parts of the world. For instance, 100 mg thrice a day is the most frequently used dose in Europe. So it
is not clear at this point that the clinical and bacteriological efficacy is dose-dependent.
Conclusion
Both drugs appear to be equally effective, safe and well tolerated
in young female with acute uncomplicated UTI.
References
Citation
Anita K, Razdan kaul A, Dhanesha G and Priyambada U. Comparative Efficacy and Safety of Fosfomycin and Nitrofurantoin in Acute Uncomplicated Lower Urinary Tract Infection in Young Women A Randomized, Open Label, Comparative Study. Indian J Gynecol. 2019;1(1): 101.