Review Article
Novel Therapeutic Advancements in Type 2 Diabetes
Jain A and Nandave M*
Department of Pharmacology, Delhi Pharmaceutical Sciences and Research University, New Delhi, India
*Corresponding author: Nandave M, Department of Pharmacology, Delhi Pharmaceutical Sciences and Research University (DPSRU), Mehrauli-Badarpur Road, Pushp Vihar, Sector 3, New Delhi 110017, India, E-mail: mukeshnandave@gmail.com
Copyright: © 2020 Nandave M, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 13/11/2020; Accepted: 07/12/2020; Published: 09/12/2020
Abstract
Type 2 diabetes mellitus (T2DM) is a metabolic disorder of the endocrine system and its prevalence has increased at an accentuating
rate in the past years. Its growing prevalence is a matter of concern as it is one of the major contributing factors for the mortality
and morbidity due to cardiovascular diseases and related ailments. Current treatments involve the use of sulfonylureas, metformin,
d-phenylalanine analogs, thiazolidinediones, α-glucosidase inhibitors, GLP-1 agonists, DPP-4 inhibitors and SGLT-2 inhibitors as mono
or combination therapy. However, the current treatments are accompanied with undesirable side effects that could be reduced through
novel advancements. Moreover, not only the prevalence but the factors that trigger the development of T2DM are also increasing
which brings about the need to develop new drugs in order to treat the same. In this review, authors have discussed some recent novel
advancements in the current treatment, novel drugs that are at different stages of discovery and development for the treatment of T2DM
and novel targets aiming which we could develop new drugs with enhanced therapeutic efficacy and reduced side effects. Additionally,
the authors have attempted to discuss the other novel therapeutic uses of the currently available drugs.
Keywords
Re-purposing of the drug; Off-label use of drug.; GLP-1; DPP-4
Introduction
Diabetes mellitus is a cluster of metabolic disorders characterized by hyperglycaemia. Decreased insulin secretion and insulin action could be the reason for the development of this disease associated with the endocrine system. Symptoms of this disease include polyuria, polyphagia, polydypsia, weight loss, blurred vision and sometimes may lead to impairment of growth or increased susceptibility towards certain infections. Uncontrolled diabetes may also lead to ketoacidosis and non-ketonic hyperosmolar syndrome.
Types of diabetes and its prevalence:
There are mainly two types of diabetes- Type 1 & Type 2: Type 1 diabetes is an autoimmune disorder and caused by auto immune destruction of β-cells leading to marked reduction in insulin production or could be idiopathic, where the etiology is not knownType 2 diabetes/diabetes mellitus (T2DM) is more prevalent than
type 1 diabetes and is caused mainly due to insulin resistance in cells
or relative deficiency of insulin secretion that is inadequate to perform
its function. This abnormality of insulin secretion and action leads to
hyperglycaemia and is underlying for several years before showing
any symptoms in the patient. Age, obesity and lack of exercise are
often contributing factors for this disease [1]
According to the Diabetes Atlas (Ninth Edition 2019) published
by Indian Diabetes Federation (IDF), there are currently 463 million
people suffering with diabetes and the number is going to accentuate
to 578 million and 700 million by 2030 and 2045 respectively. India was found to be home to 72 million of adults with diabetes, being the
second largest worldwide. In the South East Asia region, India has the
largest diabetes mortality rate with more than 1 million deaths due to
diabetes and related complications [2].
Inculcation of some habits for a healthy lifestyle is a valuable
preventive measure, but what about those who already have this
disease. There are so many factors that develop this disease and
therefore, we need drugs that target those factors. Additional factors
that could be the reason for development or advancement of this
disease are being discovered at a remarkably rapid rate, and unknown
factors that may be reason are yet to be discovered. This leads us to
the need to develop new drugs for the same. Moreover, a combination
of drugs that targets over one cause at a time is being researched and
developed to meet the needs of patients. Thus, in this article, we will
discuss the drugs that are now available to treat T2DM, drugs that
FDA has recently approved, and novel drugs that are being developed.
Target receptors and novel drugs for T2DM:
• Sulfonylureas: They are being used since very long to treat T2DM.
Carbutamide was the first drug under this class but was withdrawn
due to its side effects on bone marrow [3]. The first generation
drugs under this class were tolbutamide and chlorpropamide which
posed cardiovascular risks in patients and showed incidence of
hypoglycaemia. Overcoming all the side effects, second generation
drugs like gliclazide, glipizide, glibenclamide and glimepiride are now
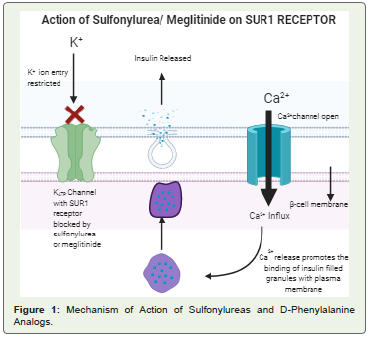
being used to successfully treat T2DM [3] (Figure 1).• Meglitinide or D-phenylalanine analogs: These include insulin
secretagogues like Repaglinide and Nateglinide that are used to
control post-prandial hyperglycaemia without causing hypoglycaemia
between the meals in diabetic patients [3,4]. Mitiglinide is a new
addition in this class. Both classes have similar mechanism of action
that is explained in the figure given below [5] (Figure 1).
• Metformin: Metformin (N, N’- dimethyl biguanide) is used worldwide as a drug for the treatment of T2DM [6]. It shows its
antihyperglycaemic effect by increasing the insulin sensitivity of
peripheral tissue and inhibiting hepatic gluconeogenesis [7]. It also
inhibits the mitochondrial complex 1 that results in disruption of
cAMP and protein kinase A signaling in response to glucagon [8].
There is now evidence that metformin could be used for the treatment
of other related diseases like neurodegenerative diseases, cancer and
cardiovascular diseases and also in the treatment of Polycystic Ovarian
Syndrome [9,10]. Thus, research and trials are being conducted to
understand these novel uses of metformin. Given below are the novel
advancements in metformin.
Metformin in the treatment of Cancer:
The cross talk (interaction between signaling pathways forming
complex networks) between heptahelical G-Protein Coupled
Receptor (GPCR) signaling system and insulin/insulin-like. Growth
Factor-1 (IGF-1) receptor plays a significant role in the regulation
of various physiological functions and a number of anomalous
processes like cardiovascular and renal abnormalities in obesity,
T2DM and metabolic syndrome. Thus, these abnormalities could
be considered as a trigger for the growth of cancer cells. GPCRS and
similarly derived agonists have been found as autocrine/paracrine
growth factors for solid tumors in pancreas, breasts, colon and
prostate gland. At cellular level, insulin coordinates with the agonists
of GPCR in stimulating mitotic signaling, DNA synthesis as well as
proliferation of pancreatic cancer cells.This crosstalk is dependent on the mammalian Target of
Rapamycin (mTOR) complex one (m TORC 1) that is inhibited
by AMPK at multiple steps which leads to the disruption in the
crosstalk between the two receptors, thus leading to inhibition of
growth of cancerous pancreatic cells. Metformin has the ability to
stimulate the AMPK, thus, could take part in inhibiting the crosstalk.
In recent studies, metformin showed a significant reduction in the
growth of pancreatic cells heterografted in nu/nu mice. Also, recent
epidemiological studies show that metformin can be attributed for
reducing the risk of pancreatic, breast, colon as well as prostate
cancer as it prevented the growth of breast and p53-/- colon cancer
cells in animal models. Thus, metformin is being considered as a
new therapeutic agent for treating the cancers mentioned above
[11].
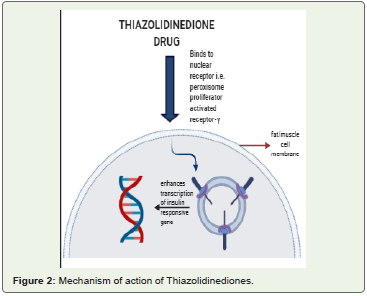
• Thiazolidinedione(PPARγ agonists): PPARγ is the factor involved in regulating the expression of genes involved in glucose and lipid metabolism. Thiazolidinediones, being agonists of this receptor help in regulation of insulin sensitivity, manage the uptake and storage of glucose and lipid in the body [12] (Figure 2). They also show antiinflammatory and anti-atherosclerotic actions [13]. Unfortunately, when these drugs are used alone, they cause undesirable side effects, such as weight gain, edema and anaemia [14].
Troglitazone was the first drug under this class that was soon
withdrawn due to occurrence of severe hepatotoxicity in patients
[15,16]. Rosiglitazone has also been withdrawn from India and Europe
as it posed increased risk of myocardial infarction in patients [17].
Pioglitazone was withdrawn from India due to incidence of bladder
cancer in patients consuming it but is now sold with a boxed warning [18]. Lobeglitazone is being used in the treatment of T2DM and it has
also shown promising results in clinical trials for its effectiveness in
the treatment of non-alcoholic fatty liver disease in diabetic patients
[19].
Drugs Currently in Clinical Trial (Table 1):
• Saroglitazar Mg- It is a dual PPAR α/γ agonist with greater effect
on the α-subtype. It was approved in 2013 in India for the treatment
of diabetic dyslipidaemia and hypertriglyceridaemia in patients not
receiving any statin therapy for the same. In 2020, it was approved to
be used as an ad-on to metformin in the treatment of T2DM and also
for non-cirrhotic non-alcoholic steatohepatitis in India [20].Novel Advancements (Table 1):
• Benzylidene Thiazolidinediones Derivatives as partial PPARγ
agonists: The already available thiazolidinediones (TZDs) are full
agonists of PPARγ receptors and are bound to the TZD nucleus
through hydrogen bonding. Aforementioned, these full agonists
show various side effects and thus, new agonists are being researched
that have the same or better therapeutic effects with fewer side effects.One such class serving the purpose mentioned above is of novel
5-Benzylidene Thiazolidin- 2,4dione Derivatives (BTZD). This novel
class of compounds is selective agonists of PPARγ receptor with partial
binding affinity because these derivatives bind through hydrophobic
bonds with the ligand binding pocket in the TZD nucleus. BTZD
have been formed by making different substitutions at the nitrogen
atom present in the TZD nucleus. Eleven derivatives were formed by
this procedure and were tested on GAL-4 PPAR chimeric receptors
and were expressed in temporarily transfected HepG2 cells [13].
Among these 11 derivatives, only 1a and 1i were shown to have weak
to moderate activity towards PPARγ at a concentration of 5 μM and
25 μM. Further five analogues of 1a and nine analogues of 1i were
tested using the same procedure for their activity and among these,
only 3a showed the desired activity.
Thus, 1a, 1i and 3a were discovered as partial agonists of PPARγ
and their activity was found to be weak to moderate using Surface Plasma Resonance. Pioglitazone 1a, 1i and 3a were administered
in Streptozotocin-Nicotinamide (STZ-NA) induced diabetic rat
at a dose of 36mg/kg and the rat models showed improvement
in hyperglycaemia and prevention in the increase of HbA1c
in comparison to the rats being given placebo. Moreover, the
histopathological damage to tissues in pancreas and liver caused by
STZ-NA induced diabetes was also restored in the ones receiving the
drug dosage. 1a, 1i and 3a are found to be cytotoxic at a concentration
of 100Μ but not at their effective dose concentration. Based on these
observations, the scientists have found this new class of substituted
thiazolidinediones with partial affinity as potent drugs for T2DM
[13].
• INT131: Diaryl sulfonamides are a novel chemical class that acts
as selective PPARγ Modulators (SPPARMγ). Studies for its structure
activity relationship and pharmacological profile led to the selection
of INT131 as a potent partial agonist under this class. INT131 is a
non-thiazolidinedione SPPARγM that binds to the same binding
pocket as thiazolidinediones but has unique binding site and interacts
with the receptors at sites different from the interaction sites of TZDs.
This distinction in binding leads to alternative conformational change
in PPARγ, thus, leading to difference in gene transcription caused
by INT131 and TZDs. This enables INT131 to retain its anti-diabetic
activity and have minimal side effects, if any. INT131 has higher
affinity for PPARγ than rosiglitazone and pioglitazone. Moreover,
it has greater than 1000 fold selectivity for PPARγ over PPARα and
PPARδ. INT131 has successfully completed pre-clinical studies and
has proved to be more potent than the TZDs full agonists in lowering
serum glucose, insulin, and triglycerides and also improved glucose
tolerance. It also increased the levels of adipokine adinopectin and
improved the level of adinopectin, otherwise reduced in obesity
and T2DM and acted as a mediator for insulin sensitivity and antiinflammatory
effects of PPARγ. It was well tolerated by animals in preclinical
studies without depicting any toxicity and also demonstrated
lower risk of carcinogenicity in comparison to full PPARγ and dual
PPARγ/α agonists. It was also well tolerated in patients in Phase 1 and
2a clinical trials and showed efficient anti-diabetic activity which led
to the conduction of its Phase 2b trials in which it is being compared
with pioglitazone to test its SPPARγM activity [21].
• α- Glucosidase Inhibitors: α-Glucosidases are a group of
carbohydrate digesting enzymes. The α-glucosidase inhibitors act by
competitively inhibiting these enzymes as well as α-amylase so that
the absorption of sugars from the gut is delayed [22]. This delay grants
the body time to produce enough amount of insulin to digest the
carbohydrates ingested by the person [17]. However, a recent clinical
study conducted on healthy subjects has shown that the medicinal
effects of α-glucosidase inhibitors may also be based on metabolic
effects of colonic starch fermentation [23]. They decrease both post
prandial hyperglycaemia and hyperinsulinaemia and may increase
the sensitivity towards insulin [22]. They have also been found to
reduce the stress on pancreatic β-cells by promoting the release of
incretins such as GLP-1 [17,22].
Drugs under this class that are sold in the market are- Acarbose,
Voglibose and Miglitol. Among these, Acarbose is the most widely
prescribed α-glucosidase inhibitor [24]. The already present α-glucosidase inhibitors are known to produce gastrointestinal side
effects and have low efficacy with high IC50 value [25]. Thus, there
is a need to search novel compounds that could overcome these
problems. Some of the novel compounds are discussed below-
• (R)-4-Fluorophenyl-1H-1,2,3Triazolebromide: This chemical compound and its derivatives (8a-n) were synthesized using commercially available S-ethyl lactate via modified Chiron approach. Screening of all of the above mentioned compound and its derivatives depicted effective inhibition of α-glucosidase enzyme with lesser IC50 value in comparison to the standard Acarbose. Saccharomyces cerevisiae α-glucosidase was used to conduct the molecular docking study of this novel compound and its derivatives. The studies showed that 1, 2, 3-triazole ring in (R)-4-fluorophenyl-1H-1, 2, 3-triazole derivatives was responsible for the inhibitory activity towards α-glucosidase by this novel compound and fluorine is the best substituent on its phenyl ring in order to carry out the same [26].
• Substituted 3-aryl coumarins derivatives: 3-arylcoumarin belongs to the class of naturally occurring compounds- arylcoumarin. Its derivatives were synthesized using microwave radiation heating. The derivatives 11, 17 & 35 formed by various substitutions in
3-arylcoumarin are of our interest as they show anti-diabetic activity
through their inhibitory effect on α-glucosidase enzyme during in vitro studies. During in vivo hypoglycaemic assay, these derivatives
reduced the glucose levels in Streptozotocin (STZ) diabetic mice
compared to healthy mice. Further chronic experiment of these three
derivatives led to the observation that derivatives 17 & 35 were more
effective than 11 in preventing body weight gain in hyperglycaemic
mice and they also reduced postprandial hyperglycaemia in STZmice.
Thus, based on all the experiments conducted, 17 & 35 were
found out to be equipotent to glibenclamide in their anti-diabetic
effect. Keeping all the factors in mind, the derivative 35 was concluded
as the potent novel compound that offered potential drug design
concept and anti-diabetic activity [27].
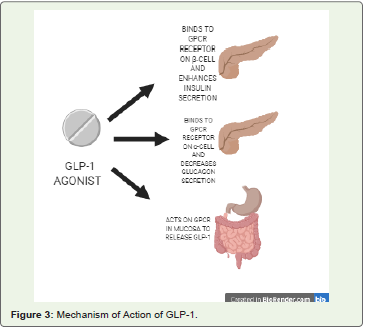
• Glucagon-like Peptide- 1 (GLP-1) analogues: GLP-1 receptors are a member of class P family of G-protein coupled receptor that are
expressed in β-cell of pancreas, various cell types of gastrointestinal
tract and neurons present in the nervous system [28]. These receptors
secrete an incretin hormone named GLP-1 after the intake of glucose
that promotes insulin secretion from pancreatic β-cells and decreases
the secretion of glucagon from pancreatic α-cells. Besides this, it
also delays gastric emptying, prevents β-cell apoptosis or promotes
β-cell proliferation as well as reduces appetite [29]. It has also been
observed to exhibit other pharmacological functions like increased
cognitive activity, decrease in cardiovascular risks etc. [37]. The
glucose dependent action of GLP-1 seems promising in avoidance of the unwanted weight gain [30]. Although, a major problem posed
by this incretin is its short half life of (2-3 min) and the degradation
action of dipeptidyl peptidase-4(DPP-4) enzyme on it [31]. Thus,
GLP-1 analogs were developed that have prolonged action in in vivo
conditions and show resistance towards degradation by DPP-4
enzyme [29] (Figure 3).
Drugs Approved by FDA (Table 1):
a. Bydureon BCise- It is an injectable suspension prepared by Astra Zeneca. It has to be administered once a week and was approved
by the FDA in 2017. This new formation of bydureon is an incretin
mimetic to improve glycaemic control and has the added benefit
of weight loss. Unlike other agonists available, it has a microsphere
delivery system that ensures consistent release of adequate amount
of exenatide. However, in its clinical trials, it was found out that it
produces hypoglycaemia if co-administered with insulin. It may also
produce itching or nodules at the site of injection [32].b. Rybelus (Semaglutide) Tablets-: FDA approved this drug on
September 20, 2019. The company that proposed this drug is Novo
Nordisk. It is an oral GLP-1 receptor agonist [33].
c. Ozempic (Semaglutide) Injection-: It was approved by FDA
on December 5, 2017, and was prepared by Novo Nordisk. It is a
weekly GLP-1 analogue [33].
d. Adlyxin (Lixisenatide) Injection-: It was approved on July 27,
2016, and its parent company is Sanofi. It needs to be administered
once daily and is prandial GLP-1 agonist [33].
e. Soliqua (100/33-Insulin glargine and lixisenatide) injection-: Sanofi made this combination of drugs. It is prescribed to be used as
adjunct to diet and exercise in order to enhance glycaemic control. It
was approved by FDA on November 21,2016 [33].
Drugs That Are Currently in Clinical Trial (Table 1):
a. LY2944876: This drug was in Phase 2 of the clinical trial in
2016. The company that is conducting its trial is Eli Lily. This drug
is given through injection under the skin. It is an oxyntomodulin
analog. It belongs to the class of antihyperglycaemic and obesity
therapy [34].b. LY3298176 (Tirzepatide): b. LY3298176 (Tirzepatide): This novel drug belongs to a new
class of dual receptor agonists that target GLP-1 as well as Gastric
Inhibitory Peptide (GIP). This drug is a fatty acid modified peptide.
It is required to be given subcutaneously once a week. Even a clinical
trial was conducted to compare its therapeutic effects to placebo
and Dulaglutide (https://clinicaltrials.gov/ no. - NCT02759107).
The results of this clinical trial came out to be very promising as
it showed effective glycaemic control and improvement in body
weight. Moreover, its chronic administration helped in decreasing
body weight and food intake. The side effects of this drug are mostly
gastrointestinal, such as vomiting, diarrhoea, abdominal distension
etc. However, all these side-effects are dose dependent with mild to
moderate severity [28].
Novel Advancements (Table 1):
a. Novel GLP-1 gene delivery complex- GLP-1 receptor agonists
have come out as a potent class of drugs for the treatment of T2DM.
However, their short half-life (<5 min.) and rapid clearance rate
limits their therapeutic efficacy [31]. To deal with this shortcoming,
scientists have developed oral delivery of therapeutic DNA by
antibody. In this, human IgG1 (hIgG1) – Fc-Arg/ pDNA complex
was prepared by electrostatic complexation of expression plasmid
and various ratios of the positively modified Fc fragments of an
antibody which targets FcRn receptor (a neonatal receptor). The biodistribution
and anti-diabetic action of the complex was examined
either in Balb/c mice or Lepdb/db mice.GLP-1 gene delivery system on the cationic hIgG1-Fc-9Arg
showed increased half-life, less immunoactivity and superior bioactivity
than simple GLP-1 analogs. Thus, this delivery system could
be a favourable approach for using GLP-1 [35].
b. Abextide 2-Exendin-4: is a peptide that is extracted from the
saliva of Gila monster and 53% of its amino acid sequence is similar
to the mammalian GLP-1 and shows resistance to degradation
by DPP-4 enzyme. Though commercially available exenatide
overcomes almost all the drawbacks of GLP-1, it requires twice-daily
administration due to its rapid clearance rate. In order to overcome
this problem, the molecular size of this protein drug was increased by
using albumin as a drug carrier. Albiglutide, an exendin-4-albumin
analog is a fusion protein that has been made using this approach
and is very effective but the fusion protein is hard to prepare and
store. Thus, there emerged a need to synthesize analogs of exendin-4
that bind covalently with albumin and have a greater half-life. Using
Evans Blue dye, they initially prepared Abextide that has similar in
vivo function as exendin-4 in healthy BALB/c mice and in diabetic
model of rodents. However it was prone to hydrolyzation which
made it lose its therapeutic efficacy. Thus, they synthesized a more
stable maleimide modified Evans Blue dye to bind with Cys-40 in
order to make Abextide 2 that was stable in both powder and solution
forms at room temperature and it was found to be more effective than
Albiglutide in decreasing glucose levels in db/db mice [37].
• Dipeptidyl Peptidase-4(DPP-4) Inhibitors (Table 1): DPP-4 is a protease enzyme that is made up of 766 amino acids. Its
degradation action on GLP-1 leads to decrease in the concentration
of these incretins in the body due to which there is a rise in blood
sugar level. Thus, inhibitors of DPP-4 enzyme were considered as a
novel treatment for T2DM. DPP-4 inhibitors are orally active drugs
whose bioavailability is greater than 80% when taken orally and are
capable of reducing the release of DPP-4 enzyme by more than 90%
for about 24 hours. They are also capable of inducing the release of
GLP-1 and GIP, because of which the load on pancreatic β-cells to
secrete insulin for glucose utilization is reduced. Thus, they help in
preventing oxidative stress in pancreatic β-cell, which is one of the
main reasons for β-cell apoptosis in patients with T2DM. They help
in enhancing the β-cell activity and this is evident as the release of
insulin is increased. They are capable of reducing the HbA1c by 0.7
to 0.8% and play a major role in controlling fasting and post-prandial
hyperglycaemia. They also reduce the secretion of glucagon which
ultimately prevents hepatic glucose production in patients with
T2DM [37]. Sitagliptin, Vildagliptin and Saxagliptin are some FDA
approved drugs under this class that are being used for the treatment
of T2DM [17].
Drugs under Development (Table 1):
a. Galangin: Skeletal muscles are responsible for about 40-50%
insulin dependent glucose uptake. However, insulin resistance due
to T2DM leads to abnormal glucose levels and reduction in the
mass of the skeletal muscles in our body. There is no combination
or mono therapy that targets improvement in skeletal muscle health
along with restoring normal glucose levels. It has been shown that
exercise leads to reduction of DPP-4 enzyme levels in our body and
this leads to improved skeletal muscle mass. Thus, it was concluded that the inhibition of DPP-4 could lead to improvement in skeletal
muscle health as well as restore glucose homeostasis. For the same
purpose, using molecular docking simulations the efficacy of a
natural flavanoid named galangin (3, 5, 7 trihydroxyflavone) has been
studied and it has shown to bind to the DPP-4 enzyme in order to
inhibit it. In vitro studies showed that galangin inhibits DPP-4 in a
dose dependent manner.Sulforhodamine B (SRB) assay was used to demonstrate its effect
on rat L6 skeletal muscle cells. Both alone and in combination with
insulin, galangin demonstrated proliferation of differentiated skeletal
muscle cells and also reduced glucose levels in skeletal muscles better
than when the cells were treated alone with insulin. Reduction in
glucose levels in skeletal muscles also indicates overall reduction in
plasma glucose levels as skeletal muscles constitute major part of the
body. Thus, galangin seems a promising novel compound for the
treatment of T2DM along with improving the skeletal muscle health
in body affected due to T2DM [38].
b. ZY15557: It is a competitive inhibitor of DPP-4 enzyme and
has a longer half-life than the currently available DPP-4 inhibitors.
It is highly selective for DPP-4 and relatively less effective against
DPP-8 and DPP-9. In vitro ZY15557 inhibited serum DPP-4 activity
in rat, mice, dog, monkey and humans. In C57 mice and Wistar rats,
it even increased the of GLP-1 and insulin levels. In db/db mice it also
reduced the extremely high level of insulin and the level of glycated
hemoglobin (HbA1c). In the hyperlipidaemia phenotype of db/
db mice, it reduced hepatic cholesterol accompanied with decrease
in hepatic oxidative stress. Thus, it is concluded that ZY15557 is an
effective competitive inhibitor of DPP-4 enzyme with accentuated
half-life and sustained action [39].
c. Evogliptin: It is a piperazine derivative and competitively
inhibits the DPP-4 enzyme. In preclinical studies, it has shown to
improve the insulin sensitivity in high fat diet- fed mice, increased
the neogenesis and multiplication of β-cell in Streptozotocin induced
diabetic mice and delayed the inception of diabetes in young leptin
receptor deficient db/db mice. Based on these promising results in
animal studies, evogliptin is now under clinical development and
received its first approval in South Korea in October 2015 for glucose
control in diabetic patients [40]. It acts in a dose dependent manner
and has a sustained action [41].
• Sodium-glucose co-transporter 2(SGLT2) Inhibitor: Kidneys are known to have a major role in maintaining glucose homeostasis
through gluconeogenesis as well as glucose reabsorption. SGLT-2
plays a major role in reabsorbing glucose from the glomerular filtrate
and is responsible for the reabsorbing about 90% of the filtered renal
glucose through the brush border cells in the proximal convoluted
tubule.
Phlorizin is a naturally occurring glucoside obtained from the
root bark of fruit trees and is responsible for the origin of the SGLT2
inhibitors. In 1950s, a study was conducted in which phlorizin blocked
the transport of glucose in several tissues like that of kidney and small
intestine. The reason for this was its inhibitory action on SGLT2and
it was even able to induce glycosuria without posing the risk of
hypoglycaemia. However, it could not be formulated into a drug due to its poor bioavailability and inhibitory action on Glucose Transporter.
But all this gave us a novel approach to create SGLT2 inhibitors that
have an insulin independent action. O-glucosidase derivatives of
phlorizin were not formulated into drugs due to their poor selectivity
towards SGLT2 and poor bioavailability. C-glucosidase derivatives of
phlorizin include Dapagliflozin, Empagliflozin and Canagliflozin that
have shown promising results in clinical trials and are now circulated
in market as drugs for T2DM [42].
Drugs approved by FDA (Table 1):
Steglatro (Ertugliflozin) tablets and Segluromet (Ertugliflozin and metformin HCl): These oral hypoglycemic drugs were approved in December 2017 and were clinically tested by the company Merck and Co. [33].Drugs under clinical trial (Table 1):
Remogliflozin Etabonate (RE) (100 mg, twice a daily tablet): It is a potent and selective inhibitor of SGLT-2 with a short half-life. After successfully completing its Phase one & two clinical trial, the drug showed effective reduction in HbA1c, fasting and post prandial glucose levels, systolic and diastolic pressure and weight loss in T2DM patients with uncontrolled hyperglycaemia in Phase 3 trial. RE is a prodrug that is de-esterified into Remogliflozin by non-specific esterases in the gastro-intestinal tract. Based on the results of Phase 3 clinical trials, RE(100 mg tablet) has been approved for adequate glycaemic control, together with diet and exercise in adults aged 18 years or above with T2DM by the Health Regulatory Authority of India [43].• Combination Drugs Targeting DPP-4 AND SGLT-2 Receptors: These combination drugs have been developed to achieve effective glycaemic control accompanied by weight loss. This combination therapy is more effective in lowering blood glucose levels and HbA1c than DDP-4 or SGLT-2 inhibitors (DPP4i and sglt2i) alone. However, this combination is therapeutically effective only when SGLT2i are combined or added to DPP-4i, not vice-versa [44].
Drugs approved by FDA (Table 1):
a. Glyxambi: It was approved by FDA on 2nd February, 2015. It
is a fixed dose combination of 10 or 25 mg empagliflozin combined
with 5 mg Linagliptin in a tablet once daily to be given as an addition
to exercise and diet in order to enhance glycaemic control type 2
diabetic patients [45].b. Qtern Tablets: This is an oral hypoglycemic combination of
dapagliflozin (acts on SGLT-2 protein) and saxagliptin (acts on DPP-
4). The company that proposed this combination and conducted its
clinical trial is Astra zeneca. FDA approved this drug in February
2017 [33].
c. Qternmet XR: It is a modified version of the Qtern tablet that
is also made and tested by the same parent company i.e. Astra Zeneca.
This oral combination drug is available in the form of an extendedrelease
tablet and has metformin in addition to dapagliflozin and
saxagliptin. It was approved by the FDA in December, 2019 [33].
• Compounds and Receptors that are being put to clinical trials for testing their potency as Hypoglycaemic Drugs (Table 1): a. SIRTUIN-1 receptor as a novel target: SIRTUIN-1(SIRT-1) is a NAD+ dependent class 3 histone deacetylase. It regulates the glucose and lipid metabolism, reduces the oxidative stress in cells, positively regulates the secretion of insulin from β cells in pancreas and also aids in modulation of insulin signaling in metabolic pathways. However, high calorie intake, insulin resistance and high glucose tolerance in cells for a long period of time slows down the regulation of SIRT-1. Based on several trials performed in mice models, it has been found that Calorie Restriction (CR) without malnutrition could help in the activation of SIRT1, which can then regulate the glucose metabolism in patients with T2DM. However, the CR that we need requires a very strict diet for a very long time which is difficult for a person to follow. Thus, researchers are working on designing drugs that could target positive SIRT1 regulation in T2DM patients [46].
b. Obeticholic Acid as a novel therapeutic compound: Farsenoid X receptor is a nuclear hormone receptor involved in regulating the lipid and glucose metabolism. Obeticholic acid (OCA) is a semisynthetic derivative of chenodeoxycholic acid and in a clinical trial; it has shown the ability to enhance the insulin sensitivity of cells and even reduced the indicators of insulin inflammation in patients suffering with non-alcoholic fatty liver disease and T2DM. In animal models, OCA has caused decrease in hepatic steatosis and insulin resistance [47].
c. G-Protein Coupled Receptor 40 (GPR40) as a novel target: GPR40 is expressed in β-cell of pancreas and even plays a role in
stimulating Glucose Dependent Insulin Secretion (GDIS) in these
cells when stimulated by extracellular Free Fatty Acids (FFAs).
GPR40 is expressed in other tissues as well but to a lesser extent. Its
role in GDIS has been supported by various pre-clinical studies in
which its removal or reduced expression decreased insulin secretion
from pancreatic β-cells and its overexpression improved GDIS in
both wild-type and diabetic rats. Notably, antagonist GW1100 was
able to inhibit the GPR40 induced GDIS from MNI6 cells in mice.
It is also expressed in the entero endocrine cells and triggers the
secretion of GLP-1 and GIP. As we have enough evidence from preclinical
and clinical studies, GPR40 synthetic selective agonists- TAK-
875 that mimic the structure of FFAs and a full agonist (stimulates the
secretion of GLP-1 and GIP as well) - AM837 have been synthesized
and these two are currently being tested in humans. These agonists
successfully accentuate insulin secretion from pancreatic β-cells,
help in restoring metabolic homeostasis and improve glucose
tolerance. However, the β-cell oxidation caused by chronic exposure
to synthetic FFAs and the receptor’s role in insulin sensitivity is still
being debated. However, as no study conducted has shown the risk
of hypoglycemia, this novel target and its agonists seem a promising
therapeutic tool for the treatment of T2DM [48].
Conclusion
With the above literature review, we further conclude that there
is a far more potential for research in the advancements of numerous
targets for the treatment of T2DM.
Acknowledgement
Authors would like to thank Ms. Megha Sahu and Mr. Ajay Kumar Pal for aiding us in structuring and reviewing the manuscript.
References
Citation
Jain A, Nandave M. Novel Therapeutic Advancements in Type 2 Diabetes. Indian J Cardio Biol Clin Sci. 2020;6(1): 112.