Case Report
A Curious Case of Left Subclavian Vein Stent Fracture
Naukarkar V*, Rajpal K and Shetty D
Department of Radiodiagnosis, Nair Hospital, Mumbai, India
*Corresponding author: Naukarkar V, Department of Radiodiagnosis, Nair Hospital, Mumbai 400008, India, Phone: +918767207597; E-mail: naukarkarvikrant@gmail.com
Copyright: © 2023 Naukarkar V, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information: Submission: 27/02/2023; Accepted: 24/04/2023; Published: 28/04/2023
Abstract
Percutaneous transluminal interventions have become the method of choice for treating symptomatic and significant vascular occlusions. Stenting is one such method with high success and fewer complication rates. To reduce complications like stent fracture care should be taken to choose the appropriate material and design of the stent and attention should be given to eliminating factors causing external stent compression. Stent fractures in the arterial system are well described in the literature. Very few reports are available for central venous stent fracture. We discuss an interesting case of a stent fracture in the
left subclavian vein due to extrinsic compression.
Keywords: Subclavian Vein; Stent Fracture; Radiology; Case Report
Introduction
Chronic kidney disease patients on hemodialysis are prone to
central venous occlusions due to repeated procedures for vascular
access and altered hemodynamics [1,2]. Here we describe a middleaged
adult presenting with central venous occlusion, treated twice
with stenting, and suffered stent fractures.
Case Presentation
42 years old male patient suffering from chronic kidney disease
on hemodialysis via left brachial arteriovenous fistula was admitted
with complaints of swelling of the left upper limb. CT angiography
study revealed complete occlusion of the left proximal subclavian and
distal brachiocephalic vein with multiple collateral venous channels
draining into the superior vena cava. Angioplasty was done across the
occluded segment using 12 mm × 60 mm and 16 mm × 80 mm balloon
angioplasty catheters. A 20 mm × 80 mm self-expanding metallic
stent (Boston Scientific WALLSTENT-Uni™ Endoprosthesis) was
placed across the central venous stenosis from the left subclavian vein
to the superior vena cava. After the procedure, a good flow was noted
across the stent from the left subclavian vein to the brachiocephalic
into the superior vena cava. The patient had a dramatic improvement
in symptoms.
Eighteen months later the same patient presented with swelling of
bilateral upper limbs. CT venography study revealed partial occlusion
of the right proximal internal jugular, subclavian and brachiocephalic
vein, and complete occlusion of left proximal internal jugular,
subclavian and brachiocephalic vein with multiple venous collaterals
draining into superior vena cava and femoral veins. The stent was seen
in the superior vena cava, left brachiocephalic, and subclavian veins.
What caught our attention the most was 95% of stent restenosis in the
distal third part with stent fracture. Angioplasty of the restenosis was
done with 12 mm × 40 mm and 14 mm × 40 mm balloon angioplasty
catheters. A 14 mm × 60 mm (Cordis S.M.A.R.T. CONTROL™
NITINOL) stent was placed across the restenosis inside the farmer
stent. A good flow was noted across the stents from the left subclavian
vein to the brachiocephalic vein into the superior vena cava. The
patient was put on aspirin, warfarin, and apixaban and discharged.
Six months later patient again presented with a recurrence of
symptoms with worsening bilateral upper limb swelling and multiple
dilated tortuous subcutaneous vessels. CT venography revealed
complete occlusion of bilateral proximal internal jugular, subclavian
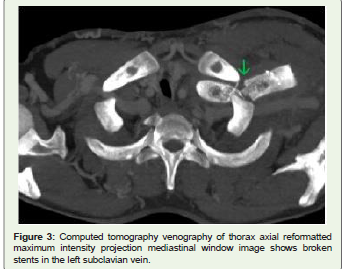
and brachiocephalic veins. To our surprise, both the stents placed in
the left subclavian and brachiocephalic veins showed fractures in the
distal third part with the medial end of the former stent indenting
the lateral wall of the superior vena cava and complete thrombosis
of the stents. The patient was due on the renal transplant list and
the ipsilateral brachial fistula was closed for hemodialysis access. It
was decided to manage the patient conservatively. He was started
on temporary peritoneal dialysis until the transplant and continued
anticoagulants and antithrombotic medications [Figure 1-4].
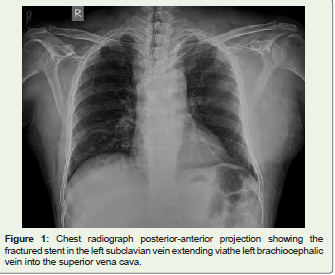
Figure 1: Chest radiograph posterior-anterior projection showing the
fractured stent in the left subclavian vein extending viathe left brachiocephalic
vein into the superior vena cava.
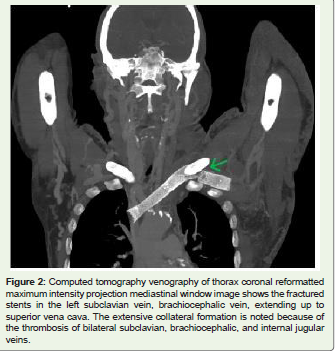
Figure 2: Computed tomography venography of thorax coronal reformatted
maximum intensity projection mediastinal window image shows the fractured
stents in the left subclavian vein, brachiocephalic vein, extending up to
superior vena cava. The extensive collateral formation is noted because of
the thrombosis of bilateral subclavian, brachiocephalic, and internal jugular
veins.
Discussion
Central venous occlusion is one of the very well-known
complications in chronic kidney disease patients on hemodialysis.
Incidence varies from 14-17%. If the occlusion is on the same side as
that of access for dialysis it needs to be treated either by surgical or
percutaneous transluminal interventions. Surgical methods carry high
morbidity and mortality and are difficult due to the position of central
veins deep in the thorax. Now a day’s intravascular interventions are
the procedure of choice for central venous thrombosis. It includes
balloon angioplasty and stent angioplasty. Both these methods
carry high success rates. Balloon angioplasty is associated with
comparatively higher recurrence. Stents are better than angioplasty
alone. However, both methods require secondary interventions most
of the time. Stenting-related complications include stent thrombosis,
stent migration, fracture, infection, and restenosis [1,2].
Sometimes a stent may get fractured. In the arterial system, it is
because of intrinsic factors like vascular pulsations. On the venous
side external compression is the main contributing factor. A stent
fracture in the left common iliac vein due to compression between the
spine and the right common iliac artery is described in the literature.
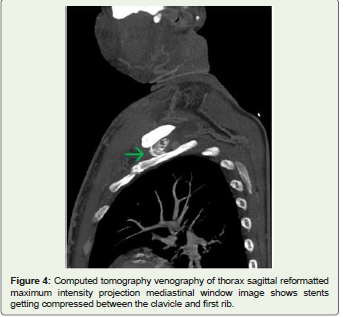
In our case, external compression between the clavicle and first rib
during arm movement may have led to repeated microtrauma and
stent fracture. Respiratory chest wall motion and aortic vascular
pulsations are contributory factors. Neointimal hyperplasia had an
additive effect and led to in-stent thrombosis. Similar findings are
also described for left brachiocephalic vein stent fracture due to
compression between the aorta and manubrium. In thoracic outlet
syndrome, Resection of the first rib is recommended before subclavian
arterial stenting for a better outcome. Whether the same can be tried
in patients with chronic kidney diseases needs to be found out [3,4].
While stenting veins over sizing is done to avoid stent migration.
Protrusion into the superior vena cava and the contra lateral venous
system should be avoided to preserve future access to hemodialysis
[5,6].
Various other factors that decide the chances of stent fracture
are design, the material of the stent, the site, and the length of the
occlusion. Wall stents are made of biomedical super alloy and have
a comparatively higher risk of recurrence of stenosis. Newer nitinolbased
stents are made up of nickel and titanium and carry a lower
risk of restenosis. The design of the stent like an open cell stent allows
more neointimal proliferation leading to in-stent thrombosis and
fracture. Closed cell stent resists this phenomenon. Self-expandable
stents offer low resistance and are more prone to collapse. Stent
which offers high radial force, for example nitinol-based stent resists
compression more as compared to Wallstent. Site of stenosis if more
proximal to the heart is more prone to stent fracture because of
cardiac pulsations. Larger the length of occlusion increase the chance
of stent collapse, kinking, and fracture. More stable venous stents that
can withstand external compression are being developed and studied
with randomized trials [7,8].
In our case, the double stent fracture was managed conservatively
because of extensive in-stent thrombosis, and closure of the ipsilateral
fistula, and the patient was on a renal transplant list. In such cases, the chances of infection and further stent migration are very less. The extensive collateral formation will eventually reduce the symptoms
[9,10].
Conclusion
Stents are safe in central venous occlusions in patients with
chronic kidney disease. Although associated with few complications
they show fewer recurrence rates. Rare complications like stent
fractures should be kept in mind while choosing the design and
material of stents and whether something can be done to avoid
external compression should be considered. Anticoagulation should
be started and continued lifelong for better outcomes.