Case Report
Tacrolimus Induced Leukoencephalopathy in Liver Transplant Recipient at Therapeutic Blood Levels
Mrinal M, Acharya UV*, Sudarshan R, Pramesh R, Patil PG, Bhavana G and Nigam U
Manipal Hospital, Bangalore, India
Corresponding author: Acharya UV, Consultant Radiologist, Manipal Hospital, 98, Old airport road, Bangalore, India, E-mail: Ullasva77@gmail.com
Citation:Mrinal M, Acharya UV, Sudarshan R, Pramesh R, Patil PG, et al. Tacrolimus Induced Leukoencephalopathy in Liver Transplant Recipient at Therapeutic Blood Levels. Indian J Appl Radiol. 2018;4(1): 125.
Copyright ©2018 Mrinal M, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Indian Journal of Applied Radiology | Volume: 4, Issue: 1
Submission: 25/05/2018; Accepted: 26/07/2018; Published: 31/07/2018
Abstract
Among the various Central Nervous System (CNS) complications of liver transplantation, immunosuppressant induced neurotoxicity constitutes a rare but important proportion. Cyclosporine-A and tacrolimus are two of the main immunosuppressive agents that have revolutionized transplant medicine. Neurotoxicity due to these agents is the major cause of seizures and headache occurring in post liver transplant patients. The signal changes on Magnetic Resonance Imaging (MRI) typically resemble Posterior Reversible Encephalopathy Syndrome (PRES) but can have heterogeneous often challenging imaging features. Prompt diagnosis and dose reduction/replacement of the immunosuppressant can reverse the symptoms and lead to improved survival of liver transplant patients. We report one such rare case of leukoencephalopathy which showed atypical PRES like signal changes and aseptic meningitis on MRI at therapeutic blood levels.
Keywords: Liver transplant; Tacrolimus; Immunosuppressant induced neurotoxicity; MRI; PRES
Key Messages:
Introduction
Neurological complications are common after organ transplantation and are associated with significant morbidity. Approximately one-third of transplant recipients experience neurologic alterations and is most commonly attributable to use of immunosuppressive drugs [1,2], because they are characterized by a narrow therapeutic index [3].
Tacrolimus (Tac) and Cyclosporine A (CsA) act as the cornerstones of the majority of immunosuppressive protocols to prevent graft rejection. They act as Calcineurin inhibitors thus inhibiting T-lymphocyte signal transduction and interleukin-2 (IL-2) transcription [4,5].
There are varying reports of neurotoxicity related to tacrolimus ranging from 7 to 32% in Solid Organ Transplants (SOT) [6]. MRI signal changes are often similar to that of PRES and occur in 0.5-5% of SOT recipients [7]. Less common but nevertheless important imaging findings include atypical PRES like changes and aseptic meningitis, knowledge of which is very essential for the right management decisions.
Early identification of drug-induced neurotoxicity in transplanted patients is important, not only because of patient’s poor clinical status but because it is reversible with dose decrement or replacement of drug. Blood levels of tacrolimus should be routinely monitored. However, neurotoxicity can occur within normal therapeutic range also [8]. Hence MRI imaging plays a vital role in management of such patients with suspected encephalopathy.
Case History
A 37 years old male with a history of deceased donor liver transplantation done in a tertiary institute in south India for hepatitis B associated cirrhosis, presented to our centre after 3 months of transplantation surgery with decreased sleep, emotional disturbances, headache and vomiting. The patient denied history of seizures, cognitive changes, episodic confusion, diplopia, dysarthria or fever.
On physical examination-vitals were normal. Blood pressure was not elevated. Lab parameters showed normal Liver Function Tests (LFT), Gamma-Glutamyl Transpeptidase (GGT), serum potassium, serum sodium and serum creatinine.
Patient was receiving tacrolimus as part of his immunosuppressive regimen since three months. The tacrolimus trough level was 10.5 ng/ml (Therapeutic range 5.0 - 15.0 ng/ml).
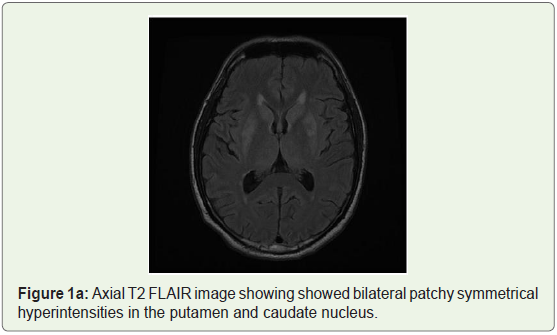
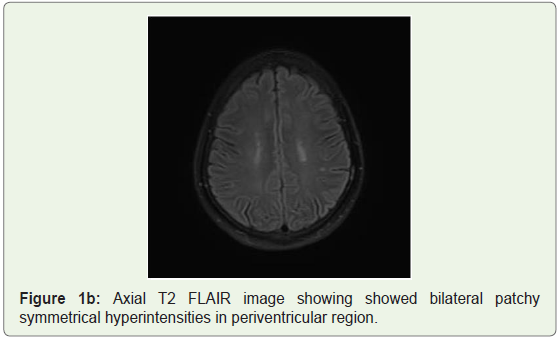
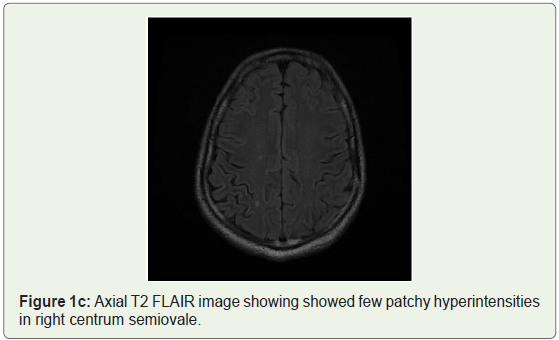
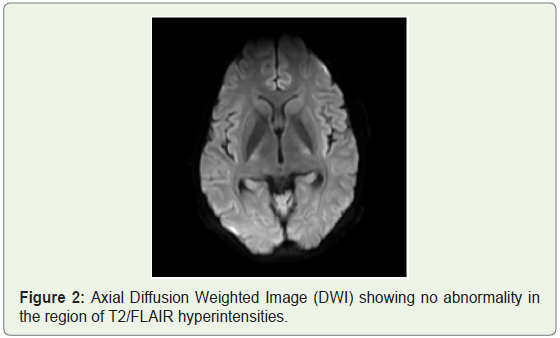
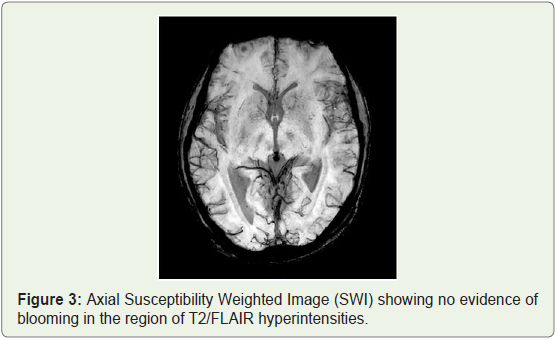
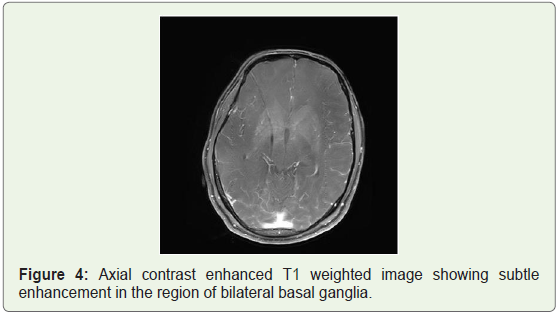
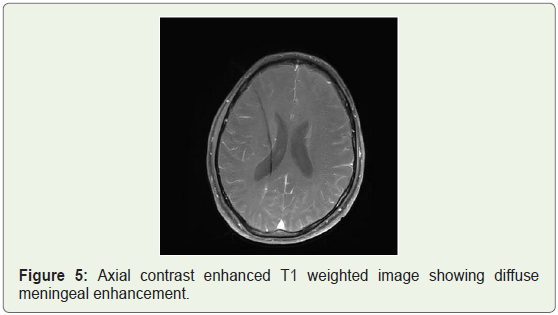
MRI brain was done which showed bilateral patchy symmetrical T2/Flair (fluid attenuation inversion recovery) hyperintensities in the putamen and caudate nucleus (Figure 1a), periventricular region and centrum semiovale (Figure 1b and Figure 1c). Diffusion Weighted Imaging (DWI) and Susceptibility Weighted Imaging (SWI) were normal (Figure 2 and Figure 3). On post contrast images, subtle enhancement seen in the region of bilateral basal ganglia with diffuse meningeal enhancement (Figure 4 and Figure 5).
Based on these findings, differential diagnosis that was considered included 1) Toxic encephalopathy with aseptic meningitis (immunosuppressant induced toxicity); 2) Metabolic encephalopathy and 3) Infections such as cryptococcal meningitis.
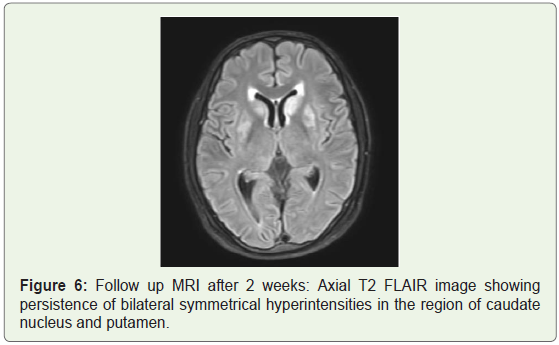
Lumbar puncture was then performed and showed no Cerebrospinal Fluid (CSF) abnormalities. Metabolic work up showed normal blood sugar, ammonia, urea and electrolyte levels. Final diagnosis of tacrolimus induced leukoencephalopathy was made. Repeat tacrolimus levels were still within therapeutic range. Tacrolimus was replaced with cyclosporine. Patient gradually showed improvement of symptoms over the next few days. Follow up plain MRI was done after two weeks which showed persistence of signal changes in the basal ganglia (Figure 6). However patient improved clinically with resolution of headache.
Discussion
Organ transplantation has become a practical and effective option for patients with acute and/or chronic irreversible organ disease. Tacrolimus and cyclosporin A are calcineurin inhibitors used to prevent rejection following allogenic solid organ transplantation.
Calcineurin is expressed in several areas of the brain: cerebral cortex, striatum, substantia nigra, cerebellum and hippocampus [4]. Calcineurin is the only calcium-activated phosphatase in the brain and a major regulator of key proteins essential for synaptic transmission and neuronal excitability, involved in memory and synaptic plasticity [5].
Tacrolimus is a macrolide calcineurin inhibitor. It reduces peptidylprolyl isomerase activity by binding to the immunophilin FKBP12 (FK506 binding protein), creating a new complex. This FKBP12-FK506 complex interacts with and inhibits calcineurin, thus inhibiting calcium-dependent events, such as T-lymphocyte signal transduction, interleukin-2 gene transcription, nitric oxide synthase activation, cell degranulation and apoptosis [9].
Neurological complications in transplant patients treated with tacrolimus have been well documented, especially after liver transplantation [6,10]. Early calcineurin inhibitor-induced neurotoxicity is considered when neurological symptoms occur within 4 weeks after transplantation [7]. Neurotoxicity can occur both at therapeutic and at high CsA or Tac levels [8]. Sometimes, neurotoxicity can be only indirectly inferred from the resolution of the clinical symptoms when treatment is discontinued. Neurotoxic effects can manifest either in the central or in the peripheral nervous system.
The major central neurotoxic effect of calcineurin inhibitors is Posterior Reversible Leukoencephalopathy Syndrome (PRES). Other major side effects include akinetic mutism, toxic encephalopathy, seizures. The minor central effects include insomnia, visual symptoms, headache, tremor, paraesthesia and mood changes; they are more frequent than major effects, occurring in almost 40% of transplant patients.
Peripheral toxicity occurs weeks to months after starting immunosuppressive treatment. Both the nerve and the muscle may be involved [9]. Axonal and demyelinating neuropathy has been reported. The more severe forms have been observed during Tac therapy, such as multifocal demyelinating neuropathy resembling Chronic Inflammatory Demyelinating Neuropathy (CIDP).
The physiopathological mechanism causing neurotoxicity of tacrolimus has not been clarified. Certain authors suggested that factors such as hypocholesterolaemia or hypomagnesaemia may predispose, as is well known for cyclosporin A [11]. No such factors were identified in the present case.
In previous reports with histopathologic analysis of tacrolimus leukoencephalopathy: the white matter was described to show small areas of demyelination, which were associated with veins and with sparing of axons in these areas [12,13]. Philippe Grimbert, et al. reported a case of Tacrolimus (FK506)-induced severe and late encephalopathy in a renal transplant recipient and identified significant tac levels in the CSF indicating that the molecule can cross the blood-brain barrier and probably attaches to myelin which is rich in lipids, permitting tac to exert a direct toxic effect [14].
MRI should be performed early in the diagnostic evaluation of patients on calcineurin inhibitors who develop significant neuropsychiatric events [15]. Imaging findings are similar to pattern of PRES with MRI showing typical distribution in the posterior regions of the white matter of the brain. Involvement of periventricular and supraventricular white matter with diffusion restriction is more commonly seen with immunosuppressant induced toxicity as compared to more cortical-subcortical pattern of involvement in PRES with diffusion restriction being less common [16].
A large amount of data has suggested that TAC induced encephalopathy is not a homogenous entity: indeed, is often presented with atypical and potentially misleading imaging characteristics not always corresponding to PRES, and includes reversible deep white matter edema and laminar cortical infarcts with hemorrhagic transformation, associated with distal vasospasms on Magnetic Resonance Angiography (MRA) [17].
Our case presented with atypical PRES like features with involvement of deep white matter and basal ganglia which can be attributed to disruption of blood brain barrier and non-specific demyelination as seen with PRES.
Another unique finding seen in our case was aseptic meningitis. There are 2 major proposed mechanisms for drug induced aseptic meningitis which includes direct irritation of the meninges by the drug and immunological type III/ IV hypersensitivity to the drug [18]. As tacrolimus itself is an immunosuppressant, aseptic meningitis can be attributable to blood-brain barrier disruption and direct meningeal irritation by the drug.
Clinical recovery after stoppage of the culprit drug is usually complete; however MRI signal changes may persist on follow up imaging as was seen in our case [19].
Conclusion
We conclude that leukoencephalopathy is a serious complication of tacrolimus use that can be completely reversible after dose decrement. Although radiologically, a predilection for the posterior cerebral white matter has been reported, uncommon presentations such as atypical PRES and aseptic meningitis should be kept in mind. As neurotoxicity can occur both at therapeutic and at high drug levels of calcineurin inhibitors, therapeutic drug monitoring cannot predict all adverse events associated with these immunosuppressive drugs. Hence, post-transplant patients with neurological complaints should be investigated with MRI at the earliest.
References
- Zivković SA, Abdel-Hamid H (2010) Neurologic manifestations of transplant complications. Neurol Clin 28: 235-251.
- Zivkovic S (2007) Neuroimaging and neurologic complications after organ transplantation. J Neuroimaging 17: 110-123.
- Dhar R, Human T (2011) Central nervous system complications after transplantation. Neurol Clin 29: 943-972.
- Asai A, Qiu Jh, Narita Y, Chi S, Saito N, et al. (1999) High level calcineurin activity predisposes neuronal cells to apoptosis. J Biol Chem 274: 34450-34458.
- Baumgärtel K, Mansuy IM (2012) Neural functions of calcineurin in synaptic plasticity and memory. Learn Mem 19: 375-384.
- Wu Q, Marescaux C, Wolff V, Jeung MY, Kessler R, et al. (2010) Tacrolimus-associated posterior reversible encephalopathy syndrome after solid organ transplantation. Eur Neurol 64: 169-177.
- Barbas AS, Rege AS, Castleberry AW, Gommer J, Ellis MJ, et al. (2013) Posterior reversible encephalopathy syndrome independently associated with tacrolimus and sirolimus after multivisceral transplantation. Am J Transplant 13: 808-810.
- Lucey MR, Kolars JC, Merion RM, Campbell DA, Aldrich M, et al. (1990) Cyclosporin toxicity at therapeutic blood levels and cytochrome P-450 IIIA. Lancet 335: 11-15.
- Sklar EM (2006) Post-transplant neurotoxicity: what role do calcineurin inhibitors actually play? AJNR Am J Neuroradiol 27: 1602-1603.
- Eidelman BH, Abu-Elmagd K, Wilson J, Fung JJ, Alessiani M, et al. (1991) Neurologic complications of FK 506. Transplant Proc 23: 3175-3178.
- Mueller AR, Platz KP, Bechstein WO, Schattenfroh N, Stoltenburg-Didinger G, et al. (1994) Neurotoxicity after orthotopic liver transplantation. A comparison between cyclosporine and FK506. Transplantation 58: 155-170.
- Small SL, Fukui MB, Bramblett GT, Eidelman BH (1996) Immunosupression-induced leucoencephalopathy from tacrolimus (FK506). Ann Neurol 40: 575-580.
- Thyagarajan GK, Cobanoglu A, Johnston W (1997) FK506-induced fulminant leukoencephalopathy after single-lung transplantation. Ann Thorac Surg 64: 1461-1464.
- Grimbert P, Azema C, Pastural M, Dhamane D, Remy P, et al. (1999) Tacrolimus (FK506)-induced severe and late encephalopathy in a renal transplant recipient. Nephrol Dial Transplant 14: 2489-2491.
- Koh KN, Park M, Kim BE, Im HJ, Seo JJ (2010) Early central nervous system complications after allogeneic hematopoietic stem cell transplantation in children. Korean J Hematol 45: 164-170.
- Truwit CL, Denaro CP, Lake JR, DeMarco T (1991) MR imaging of reversible cyclosporin A-induced neurotoxicity. AJNR Am J Neuroradiol 12: 651-659.
- Wu Q, Marescaux C, Qin X, Kessler R, Yang J (2014) Heterogeneity of radiological spectrum in tacrolimus-associated encephalopathy after lung transplantation. Behavioural Neurology, pp: 9.
- Jolles S, Sewell WA, Leighton C (2000) Drug-induced aseptic meningitis: diagnosis and management. Drug Saf 22: 215-226.
- Schuuring J, Wesseling P, Verrips A (2003) Severe tacrolimus leukoencephalopathy after liver transplantation. AJNR Am J Neuroradiol 24: 2085-2088.