Case Report
Multiparametric MR Evaluation of an Unusual Case of Periprostatic Leiomyoma with Bizarre Nuclei
Gupta A1,2*, Satapathy AK1and Mohapatra SSG1
1Department of Radiodiagnosis, Institute of Medical Sciences and SUM Hospital, Siksha ‘O’ Anusandhan University, Bhubaneswar, Odisha, India
2Department of Radiodiagnosis, MGS Hospital, Punjabi Bagh, New Delhi, India
2Department of Radiodiagnosis, MGS Hospital, Punjabi Bagh, New Delhi, India
*Corresponding author:Abhinav Gupta, Department of Radiodiagnosis, Institute of Medical Sciences and SUM Hospital, Siksha ‘O’ Anusandhan University, Bhubaneswar, Odisha, India. E-mail: abhinav491@gmail.com
Copyright:© 2024 Gupta A, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article Information:Submission: 21/02/2024; Accepted: 15/03/2024; Published: 19/03/2024
Abstract
A middle-aged patient with perineal pain and frequent urination had a periprostatic mass detected via Transrectal Ultrasound (TRUS). The serum prostatic surface antigen (PSA) level was within normal range. Multiparametric MRI (mp-MRI) revealed a mildly enlarged prostate. No abnormal focal or diffuse T2W
hypointense lesions were detected in the prostate. A well-defined periprostatic mass measuring 27 x 18.4 x 22.9 mm, exhibiting T1W/T2W hypointense signals, was identified. The mass closely abutted prostatic apex, anteroinferior capsule, anterior rectal wall, and left puborectalis sling, without invasion or
infiltration. DWI indicated no diffusion restriction. The dynamic contrast-enhanced MRI (DCE-MRI) showed slow continuous enhancement within the mass, potentially excluding malignancy. Additionally, absence of T2W hypointense lesions in transition-zone ruled out stromal nodules of BPH. Furthermore,
absence of such lesions in the transition/ and or peripheral-zone, along with an intact prostatic-capsule, excluded prostatic malignancy with extracapsular extension. The absence of T2W hyperintense lesions in peripheral-zone of prostate excluded STUMP. The lack of irregular margins, invasion, or infiltration
into surrounding structures, along with absence of diffusion restriction in DWI, and the lack of early hyperintensity in DCE-MRI collectively rules out the likelihood of various malignant mesenchymal tumors. TRUS-guided biopsy for histopathological examination (HPE) supported by immunohistochemistry
(IHC), conclusively identified the mass as a periprostatic Leiomyoma with Bizarre Nuclei (LBN). LBN is extremely rare and carries a potential for malignant transformation. In conclusion, mp-MRI stands as a valuable modality for characterizing a periprostatic mass, facilitating differentiation between benign and
malignant lesions. However, a definitive diagnosis requires HPE and IHC.
Keywords:Multiparametric MRI; Prostatic Mesenchymal Tumors; Periprostatic Mass; Leiomyoma with Bizarre Nuclei
Introduction
The presence of a periprostatic mass presents a diagnostic
challenge, as it can closely resemble primary prostatic diseases,
especially malignancies [1-3]. About 60-70% of prostatic malignancies
manifest as hypoechoic focal lesions in Transrectal
Ultrasound (TRUS) relative to normal peripheral zone, while 30-40%
are isoechoic and may thus go unnoticed[4].Multiparametric MRI
(mp-MRI) is currently the leading imaging modality for detecting
and characterizing various prostatic lesions due to its exceptional
sensitivity and specificity [5-7]. While certain peri-prostatic masses
exhibit characteristic imaging features, histopathological evaluation
(HPE) is essential for a precise diagnosis [2,8,9].
Case Report
We report an unusual case of periprostatic mass in a patient aged
47-years with perineal pain and frequent urination for the past three
months. There was no history of fever, urinary retention, hematuria,
or ejaculatory impairment. Digital rectal examination revealed a
palpable firm to hard nodular mass anterior to the rectum. Serum
prostate-specific antigen (PSA) level was 0.9 ng/ml [normal <4.00
ng/ml]. The patient exhibited normal renal and liver functions, as
evidenced by laboratory results, and urinalysis was unremarkable.
The TRUS indicated prostate volume of approximately 33 cc
and revealed a hypoechoic mass measuring 2.5 cm x 2.3 cm that
extended beyond the prostate. There was no discernible distortion of
the prostatic capsule. In the mp-MRI using 1.5 T scanner, a mildly
enlarged prostate was identified with a volume of 32 cc. No abnormal
focal or diffuse T2W hypointense lesions were detected in the
transition and / or peripheral zone. Prostate margins were delineated
as a thin rim of low signal intensity, indicating intact prostatic capsule.
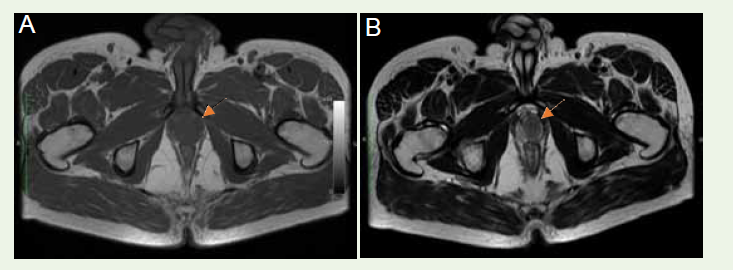
Remarkably a well-defined periprostatic mass, measuring 27 x 18.4 x
22.9 mm, was delineated. The mass closely abutted the anteroinferior
capsule and displayed hypointense signals in both T1W and T2W
sequences [Figure 1A], [Figure 1B]. Its proximity to the apex of the prostate was observed without any infiltration into parenchyma.
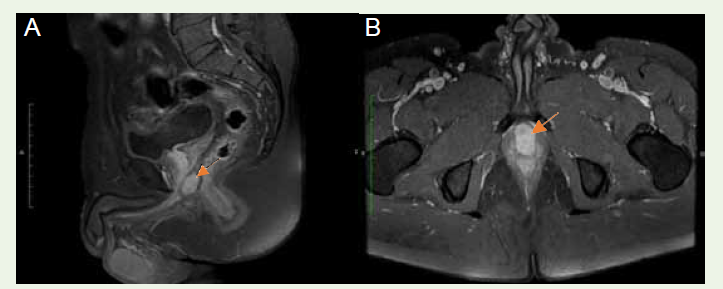
Additionally, the mass was noted to abut the anterior rectal wall
and the left puborectalis sling [Figure 2A], [Figure 2B]. The seminal vesicles, urinary bladder, rectum, and neurovascular bundle exhibited
no invasion or infiltration. There was an absence of ascites or pelvic
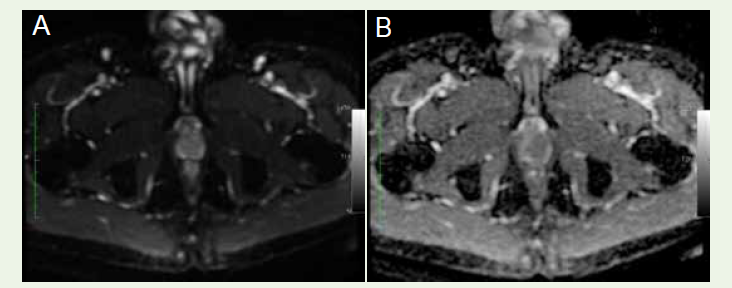
lymphadenopathy. DWI and ADC mapping revealed no diffusion
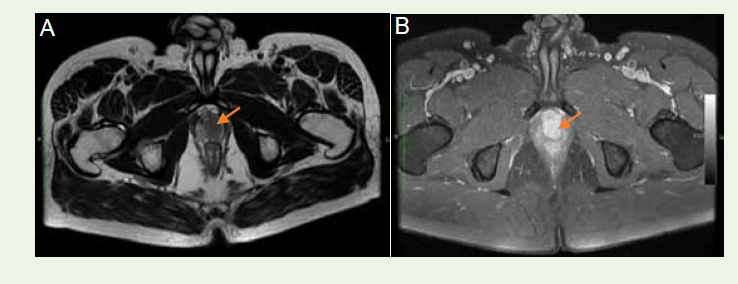
restriction [Figure 3A], [Figure 3B]. The DCE-MRI demonstrated
a slow, continuous, homogenous contrast enhancement within the
mass [Figure 4A], [Figure 4B].
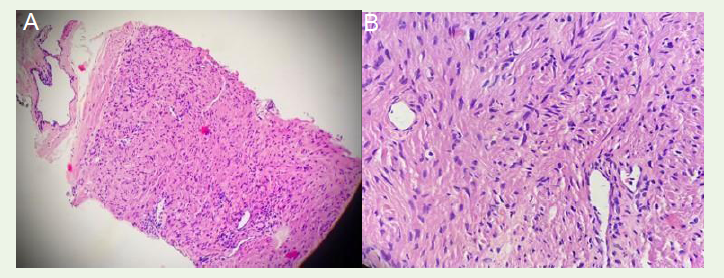
TRUS guided biopsy for HPE of the mass showed an encapsulated
tumor of spindle cells arranged in fascicles with elongated blunt
nuclei, mild nuclear atypia, minimal mitosis (1/10 HPF), absence
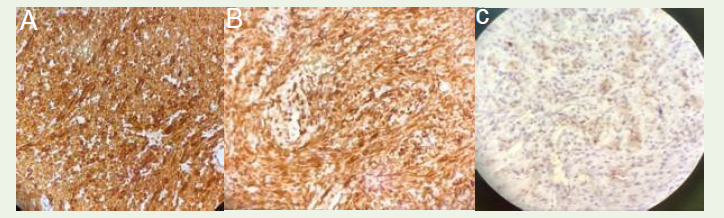
of necrosis, invasion, or glandular element [Figure 5A], [Figure 5B] Immunohistochemistry (IHC) revealed positive expressions for
desmin [Figure 6A] smooth muscle actin (SMA) [Figure 6B] and a
low Ki-67 nuclear protein [Figure 6C]. On the contrary, the staining
was negative for cluster of differentiation 34 and C-kit. The results
of HPE and IHC staining collectively confirmed the diagnosis of a
periprostatic Leiomyoma with Bizarre Nuclei (LBN).
Discussion
In the present case, the mp-MRI detected a slightly enlarged
prostate with normal T1W and T2W signals. Prostate margins were
well delineated as a thin rim of low signal intensity, indicating intact
prostatic capsule. Remarkably, a well-defined periprostatic mass was
discovered adjacent to the prostate apex, exhibiting hypointense
signals in both T1W and T2W sequences. Significantly, no signs
of invasion or infiltration into the prostatic parenchyma, seminal
vesicle, neurovascular bundle, urinary bladder, rectum, or any other
pelvic structure were observed.
The absence of T2W hypointense lesions in the transition zone of
prostate ruled out possibility of stromal nodules of benign prostatic
hyperplasia in the present case.
It is noteworthy that about 70% of prostatic malignancies occur
within peripheral zone and manifest as a reasonably well defined
T2W hypointensity, along with low ADC signals in the DWI,
typically demonstrating early hyperintensity on DCE-MRI [6,7].
Therefore, the absence T2W hypointense lesions in the transition/
and or peripheral zones, along with an intact capsule, and normal
PSA level, perhaps excludes prostatic malignancy with extracapsular
extension in the present case [1,2,10].
In the current case, the DWI of the periprostatic mass revealed
no diffusion restriction, with normal ADC values. DCE-MRI
demonstrated a slow continuous wash-in, a feature linked with
benign masses. This contrasts with the fast wash-in, leading to
early hyperintensity, followed by fast wash-out with a reduction in
enhancement, which is a hallmark of malignant masses [1].
The potential identification of this mass as a stromal tumor of
uncertain malignant potential (STUMP) was contemplated. STUMPs
typically manifest in the peripheral zone of the prostate at the base,
presenting as a well-defined mass with a combination of solid and
cystic components, exhibiting a distinctive high T2W signal intensity.
DWI with ADC mapping often reveals moderately restricted
diffusion, the degree of which depends on the proportion of cystic and
solid components [2,3,8]. Nevertheless, in the present case, the mass
was located adjacent to the prostatic apex and exhibited hypointense
signals on T2W imaging, indicating absence of cystic component.
Figure 2:(A)T1W Sagittal and (B) T1W Axial contrast images show periprostatic mass (arrow) abutting the apex of prostate without invasion of parenchyma and abutting the anterior rectal wall.
Figure 3:(A) Diffusion Weighted Imaging and (B) Apparent Diffusion Coefficient Map shows no diffusion restriction in the mass.
Figure 4:Dynamic contrast enhanced MRI: Axial T1W images show (A) Hypointense mass before administration of the contrast (arrow)(B) Post-contrast homogenous enhancement in the mass (arrow)
Figure 5:TRUS Biopsy from periprostatic mass shows (A) An encapsulated tumor having spindle cells arranged in fascicles, whorls & intersecting thin walled ectatic vessels. (B) Tumor cells have elongated blunt nuclei with mild nuclear atypia, eosinophilic bipolar cytoplasm, with minimal mitosis (1/10 HPF). No necrosis, invasion, or glandular elements.
Prostatic stromal sarcomas are primarily solid tumors. They were
also considered in the differential diagnosis. They typically manifest
as large masses invading adjacent structures. On MRI, they appear
as hypointense regions on T1W images and exhibit heterogeneous
hyperintensity on T2W images, indicative of underlying areas of
internal necrosis, hemorrhage, and cystic degeneration. DWI and
ADC mapping reveal varying degrees of restricted diffusion, based
on the cellular composition of the mass [2,3,8]. However, absence
of irregular margins of the mass, lack of invasion or infiltration into
surrounding structures, and hypointense signals on T2W imaging in
the current case, argues against the likelihood of prostatic stromal
sarcoma.
The evaluation also considered malignant non-stromal
mesenchymal tumors of prostate, including leiomyosarcoma,
rhabdomyosarcoma, synovial sarcoma, and gastrointestinal stromal
tumors (GIST). These tumors are rare and pose a diagnostic
challenge. On MRI, they typically appear as hypointense regions
on T1W images but display heterogeneous hyperintensity on T2W
images, indicating underlying hemorrhage, internal necrosis, and
local infiltration [2,3,8]. However, absence of irregular margins of the
mass, lack of invasion or infiltration into surrounding structures, and
the T2W hypointense signals, collectively diminish their possibility
in the present case.
Prostatic solitary fibrous tumors (SFT) originate from
periprostatic soft tissue, initially presenting as a predominantly
fibrous benign mass with potential for malignancy. They were also
considered in the differential diagnosis. On MRI, SFT typically
appears hypointense on T1W images and exhibits heterogenicity on
T2W images [2,3,8], showing a distinctive “chocolate chip cookie”
appearance. This results from low-intensity foci in T2-weighted
images, attributed to collagen content and low cellularity. As vascular
tumors, SFT show robust enhancement [11]. However, in the present
case, dynamic contrast enhanced study did not reveal pronounced
enhancement within the mass.
GIST manifesting as a periprostatic mass typically arises at the
anorectal junction or in the perirectal or periprostatic soft tissue
potentially compressing and invading the prostate. On MRI, they
typically appear as a well-defined lobulated mass with heterogenous
T2 hyperintense signals, intermediate to low T1 signal intensity, and
irregular enhancement. DWI and ADC mapping usually show marked
diffusion restriction [2,3]. In the present case, the periprostatic mass
marked by its non-invasiveness, hypointense T2-weighted signals,
slow homogeneous enhancement pattern, with a normal anorectal
junction diminishes the likelihood of it being a gastrointestinal
stromal tumor.
Prostatic leiomyomas are uncommon. They typically arise from
the central prostate towards the apex. They originate from the smooth
muscle elements within the prostate’s stroma, capsule, or mullerian
remnants [2]. In the present case, distinctive mp-MRI characteristics
including well-defined margins, proximity to the prostatic apex,
the absence of infiltration or invasion into surrounding structures,
typical low signal intensity on T2W images, and the absence of early
enhancement in dynamic contrast-enhanced MRI, collectively point
towards a benign tumor, likely a leiomyoma [2,3,8,12].
TRUS guided biopsy of the mass for histopathological
examination revealed an encapsulated tumor comprising spindle cells
arranged in fascicles with elongated blunt nuclei, mild nuclear atypia,
a low mitotic rate (1/10 high-power fields [HPF]), and an absence
of internal necrosis, invasion, or glandular elements consistent with
the diagnosis of Leiomyoma with Bizarre Nuclei (LBN), previously
termed symplastic leiomyoma [13,14]. Immunohistochemically, it
differed from STUMP and GIST by its smooth muscle actin (SMA)
(+), desmin (+), CD34 (-), and C-kit (-) staining pattern [2,8,9].
LBN represents a subgroup of leiomyomas, a category
infrequently reported and primarily documented in uterine
leiomyomata [14]. The World Health Organization has characterized
LBN as a leiomyoma demonstrating focal or diffuse nuclear
atypia, with or without increased mitosis typically averaging 1–2
mitoses/10 HPF, occasionally reaching up to 7–8 mitoses/10 HPFs,
but not exceeding 10 mitoses/HPF [14]. Distinguishing LBN from
leiomyosarcomas is crucial; the latter exhibits marked cellular atypia,
≥10 mitoses/10 HPF, hyperchromatic nuclei with moderate to severe
nuclear pleomorphism, and tumor cell necrosis [14]. However,
LBN may represent a precancerous stage of leiomyosarcoma, given
the subsequent risk of malignant transformation [15-17]. Hence,
some experts recommend radical surgery [15] while others advocate
conservative management and close follow-up [16].
To conclude, mp-MRI stands as a valuable modality for
identifying, localizing, and characterizing periprostatic masses,
facilitating the differentiation between benign and malignant lesions.
However, for a conclusive diagnosis, histopathologic examination
(HPE) and immunohistochemistry (IHC) remain indispensable.
Acknowledgements
The authors gratefully acknowledge the Department of Pathology
at Institute of Medical Sciences, SUM Hospital for their invaluable
contribution in providing histopathology and immunohistochemistry
images for this publication.
References
Citation
Gupta A, Satapathy AK, Mohapatra SSG.. Multiparametric MR Evaluation of an Unusual Case of Periprostatic Leiomyoma with Bizarre Nuclei. Indian J Appl Radiol. 2024;10(1): 192.